Abstract
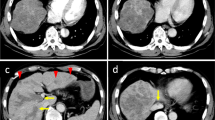
We report a 46-year-old male patient with functional liver damage due to hepatitis B virus infection. A 12 cm hepatocellular carcinoma (HCC) in the left lobe and portal venous tumor thrombosis (PVTT) with vp4 (portal vein tumor thrombosis in the main trunk) were detected by computed tomography (CT). He underwent hepatic arterial infusion chemotherapy (HAIC) with cisplatin 100 mg for HCC and received radiation therapy (39 Gy/13 Fr) for PVTT with vp4. Follow-up CT showed reduction of HCC and reduced PVTT volume after 1 month of treatment. He then initiated lenvatinib therapy at 12 mg/day. One month later, follow-up CT showed no change in HCC size and a reduction in PVTT volume. Two months after initiating lenvatinib, follow-up CT showed no change in HCC, but further reduction in contrast effect and volume of PVTT. Three months after HAIC, he underwent drug-eluting-bead transcatheter arterial chemoembolization (DEB-TACE) with 100 mg of cisplatin (CDDP) for the HCC. After DEB-TACE, he received 12 mg/day with 5-days-on/2-days-off due to vomiting. One month after DEB-TACE, blood evaluation showed decreased tumor markers, and CT revealed that the HCC had grown slightly with no change in PVTT. Five months after HAIC, he underwent DEB-TACE with 100 mg of cisplatin for the HCC. A total of 150 days have passed since the start of lenvatinib treatment, and his Child–Pugh A status has been maintained.



Similar content being viewed by others
References
Matsui J, Funahashi Y, Uenaka T, et al. Multi-kinase inhibitor E7080 suppresses lymph node and lung metastases of human mammary breast tumor MDA-MB-231 via inhibition of vascular endothelial growth factor-receptor (VEGF-R) 2 and VEGF-R3 kinase. Clin Cancer Res. 2008;14(17):5459–65.
Matsui J, Yamamoto Y, Funahashi Y, et al. E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition. Int J Cancer. 2008;122(3):664–71.
Tohyama O, Matsui J, Kodama K, et al. Antitumor activity of lenvatinib (e7080): an angiogenesis inhibitor that targets multiple receptor tyrosine kinases in preclinical human thyroid cancer models. J Thyroid Res. 2014;2014:638747.
Yamamoto Y, Matsui J, Matsushima T, et al. Lenvatinib, an angiogenesis inhibitor targeting VEGFR/FGFR, shows broad antitumor activity in human tumor xenograft models associated with microvessel density and pericyte coverage. Vasc Cell. 2014;6:18.
Kudo M, Finn RS, Qin S, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391(10126):1163–73.
Fu**o H, Kimura T, Aikata H, et al. Role of 3-D conformal radiotherapy for major portal vein tumor thrombosis combined with hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma. Hepatol Res. 2015;45(6):607–17.
Fujii T, Takayasu K, Muramatsu Y, et al. Hepatocellular carcinoma with portal tumor thrombus: analysis of factors determining prognosis. Jpn J Clin Oncol. 1993;23(2):105–9.
Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378–90.
Yoshida K, Hirokawa T, Moriyasu F, et al. Arterial-phase contrast-enhanced ultrasonography for evaluating anti-angiogenesis treatment: a pilot study. World J Gastroenterol. 2011;17(8):1045–50.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Kazuaki Chayama has received honoraria from Bristol-Myers Squibb, MSD K.K., AbbVie Inc., Gilead Sciences, Dainippon Sumitomo Pharma and Mitsubishi Tanabe and research funding from Dainippon Sumitomo Pharma, MSD K.K, AbbVie Inc, Eisai, Toray, Otsuka, Chugai, Takeda, Roche, Mitsubishi Tanabe and Bristol-Byers Squibb. Michio Imamura has received research funding from Bristol-Myers Squibb. Other authors declare no competing interests.
Human rights
All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kosaka, Y., Kawaoka, T., Aikata, H. et al. A case of advanced HCC treated with lenvatinib after hepatic arterial infusion chemotherapy combined with radiation therapy treatment for portal vein tumor thrombosis in the main trunk. Clin J Gastroenterol 13, 839–843 (2020). https://doi.org/10.1007/s12328-020-01093-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-020-01093-9