Abstract
Introduction
Long-acting injectable (LAI) antipsychotic use may reduce healthcare resource utilization compared with oral antipsychotic use by improving adherence and reducing dosing frequency. Our goal was to examine treatment patterns, healthcare utilization, and costs among recently diagnosed schizophrenia patients receiving oral versus LAI antipsychotics.
Methods
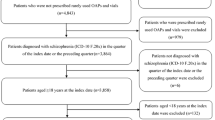
The MarketScan Multi-state Medicaid database was used to identify schizophrenia patients aged ≥ 18 years who received an LAI or oral antipsychotic between January 1, 2011 and December 31, 2014. Primary outcomes included treatment patterns such as adherence (measured as proportion of days covered-PDC), persistence, discontinuation, switching, and healthcare resource utilization and costs. Propensity score matching (PSM) was used to control for differences in baseline characteristics between the cohorts. Outcomes were assessed over a 12-month post-index period and compared between treatment cohorts.
Results
After PSM, 2302 patients were included in each of the LAI and oral antipsychotics cohorts. There were no differences in PDC or therapy switching between the two cohorts. Compared with the oral cohort, patients receiving LAIs had lower discontinuation rates (46.1 vs. 61.6%, p < 0.001), fewer inpatient admissions (0.5 vs. 0.9, p < 0.001), hospital days (3.9 vs. 6.5, p < 0.001), and ER visits (2.4 vs. 2.9, p = 0.007), and a higher number of prescription fills (29.5 vs. 25.3, p < 0.001). Patients prescribed LAIs had lower monthly inpatient ($US4007 vs. 8769, p < 0.001) and ER visits costs ($682 vs. 891, p < 0.001) but higher monthly medication costs ($10,713 vs. $655, p < 0.001) compared with the oral cohort over the 12-month post-index period. Overall, both cohorts had similar total medical costs (LAI vs. oral: $24,988 vs. 23,887, p = 0.354) during the follow-up period.
Conclusion
Patients receiving LAIs were more likely to remain on medication compared with the oral group, which may account for reduced inpatient admissions. Hospitalization cost reductions offset the higher costs of LAI medications, resulting in no increase in total healthcare costs relative to oral antipsychotic use.
Funding
Alkermes Inc.





Similar content being viewed by others
References
Tandon R, Nasrallah HA, Keshavan MS. Schizophrenia, “just the facts” 4. Clinical features and conceptualization. Schizophr Res. 2009;110(1–3):1–23.
Schizophrenia. National Institute of Mental Health Web site. http://www.nimh.nih.gov/health/statistics/prevalence/schizophrenia.shtml. Updated November 2017. Accessed Feb 22 2018.
** H, Mosweu I. The societal cost of schizophrenia: a systematic review. Pharmacoeconomics. 2017;35(1):25–42.
Millier A, Schmidt U, Angermeyer MC, et al. Humanistic burden in schizophrenia: a literature review. J Psychiatr Res. 2014;54:85–93.
Cloutier M, Aigbogun MS, Guerin A, et al. The economic burden of schizophrenia in the US in 2013. J Clin Psychiatry. 2016;77(6):764–71.
Wyatt RJ. Neuroleptics and the natural course of schizophrenia. Schizophr Bull. 1991;17(2):325–51.
Leucht S, Tardy M, Komossa K, et al. Antipsychotic drugs versus placebo for relapse prevention in schizophrenia: a systematic review and meta-analysis. Lancet. 2012;379:2063–71.
Lacro JP, Dunn LB, Dolder CR, Leckband SG, Jeste DV. Prevalence of and risk factors for medication nonadherence in patients with schizophrenia: a comprehensive review of recent literature. J Clin Psychiatry. 2002;63:892–909.
Velligan DI, Wang M, Diamond P, et al. Relationships among subjective and objective measures of adherence to oral antipsychotic medications. Psychiatr Serv. 2007;58(9):1187–92.
Pinikahana J, Happell B, Taylor M, Keks NA. Exploring the complexity of compliance in schizophrenia. Issues Ment Health Nurs. 2002;23(5):513–28.
Cramer JA, Rosenheck R. Compliance with medication regimens for mental and physical disorders. Psychiatr Serv. 1998;49(2):196–201.
Sullivan G, Wells KB, Morgenstern H, Leake B. Identifying modifiable risk factors for rehospitalization: a case-control study of seriously mentally ill persons in Mississippi. Am J Psychiatry. 1995;152(12):1749–56.
Ascher-Svanum H, Faries DE, Zhu B, Ernst FR, Swartz MS, Swanson JW. Medication adherence and long-term functional outcomes in the treatment of schizophrenia in usual care. J Clin Psychiatry. 2006;67(3):453–60.
Offord S, Lin J, Mirski D, Wong B. Impact of early nonadherence to oral antipsychotics on clinical and economic outcomes among patients with schizophrenia. Adv Ther. 2013;30(3):286–97.
Dilla T, Ciudad A, Alvarez M. Systematic review of the economic aspects of nonadherence to antipsychotic medication in patients with schizophrenia. Patient Prefer Adher. 2013;7:275–84.
Sun SX, Liu GG, Christensen DB, Fu AZ. Review and analysis of hospitalization costs associated with antipsychotic nonadherence in the treatment of schizophrenia in the US. Curr Med Res Opin. 2007;23:2305–12.
Davis JM, Matalon L, Watanabe MD, Blake L, Metalon L. Depot antipsychotic drugs. Place in therapy. Drugs. 1994;47(5):741–73.
Johnson DA. Historical perspective on antipsychotic long-acting injections. Br J Psychiatry Suppl. 2009;52:S7–12.
Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754–69.
Lin J, Wong B, Offord S, Mirski D. Healthcare cost reductions associated with the use of LAI formulations of antipsychotic medications versus oral among patients with schizophrenia. J Behav Health Serv Res. 2013;40(3):355–66.
Lafeuille MH, Laliberté-Auger F, Lefebvre P, et al. Impact of atypical long-acting injectable versus oral antipsychotics on rehospitalization rates and emergency room visits among relapsed schizophrenia patients: a retrospective database analysis. BMC Psychiatry. 2013;13:221.
Offord S, Wong B, Mirski D, et al. Healthcare resource usage of schizophrenia patients initiating long-acting injectable antipsychotics vs. oral. J Med Econ. 2013;16:231–9.
Pilon D, Tandon N, Lafeuille MH, et al. Treatment patterns, healthcare resource utilization, and spending in medicaid beneficiaries initiating second-generation long-acting injectable agents versus oral atypical antipsychotics. Clin Ther. 2017;39(10):1972–85.
West JC, Marcus SC, Wilk J, et al. Use of depot antipsychotic medications for medication nonadherence in schizophrenia. Schizophr Bull. 2008;34(5):995–1001.
Heres S. Long-acting injectable antipsychotics: an underutilized treatment option. J Clin Psychiatry. 2014;75(11):1263–5.
Heres S, Hamann J, Kissling W, Leucht S. Attitudes of psychiatrists toward antipsychotic depot medication. J Clin Psychiatry. 2006;67(12):1948–53.
Hansen L. The Truven Health MarketScan Databases for life sciences researchers. Truven Health Analytics. 2017. https://truvenhealth.com/Portals/0/Assets/2017-MarketScan-Databases-Life-Sciences-Researchers-WP.pdf. Accessed Sept 19 2017.
Lehman AF, Lieberman JA, Dixon LB, et al. Practice guideline for the treatment of patients with schizophrenia. Psychiatry Online. 2004. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/schizophrenia.pdf. Accessed Feb 19 2018.
Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130–9.
Charlson ME, Charlson RE, Peterson JC, et al. The Charlson comorbidity index is adapted to predict costs of chronic disease in primary care patients. J Clin Epidemiol. 2008;61(12):1234–40.
Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
Von Korff M, Wagner EH, Saunders K. A chronic disease score from automated pharmacy data. J Clin Epidemiol. 1992;45(2):197–203.
Joshi K, Pan X, Wang R, Yang E, Benson C. Healthcare resource utilization of second-generation long-acting injectable antipsychotics in schizophrenia: risperidone versus paliperidone palmitate. Curr Med Res Opin. 2016;32(11):1873–81.
Campagna EJ, Muser E, Parks J, Morrato EH. Methodological considerations in estimating adherence and persistence for a long-acting injectable medication. J Manag Care Spec Pharm. 2014;20(7):756–66.
Risperdal Consta (risperidone) long-acting injection [package insert]. Janssen. 2017. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/RISPERDAL+CONSTA-pi.pdf. Accessed Feb 15 2018.
Abilify Maintena (aripiprazole) for extended-release injectable suspension, for intramuscular use [package insert]. Otsuka Pharmaceuticals. 2017. https://www.otsuka-us.com/media/static/Abilify-M-PI.pdf?_ga=2.155269362.833124413.1518720548-834450404.1518720548. Accessed Feb 15 2018.
Zyprexa Relprevv (olanzapine) for extended-release injectable suspension [package insert]. Lilly. 2018. http://pi.lilly.com/us/zyprexa_relprevv.pdf. Accessed Feb 15 2018.
Fluphenazine Decanoate Injection, USP [package insert]. APP Pharmaceuticals, LLC. https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/071413s019lbl.pdf. Accessed Apr 12 2018.
Haloperidol Injection, USP [package insert]. Pfizer Labs. https://www.pfizer.com/files/products/uspi_haloperidol.pdf. Accessed Apr 12 2018.
Invega Sustenna (paliperidone palmitate) extended-release injectable suspension, for intramuscular use [package insert]. Janssen. 2017. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/INVEGA+SUSTENNA-pi.pdf. Accessed Feb 15 2018.
Crawford M, Church J, Akin B. CPI detailed report data for December 2015. Bureau of Labor Statistics website. https://www.bls.gov/cpi/cpid1412.pdf. Published 2015. Accessed Feb 15 2018.
Austin PC. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun Stat Simul Comput. 2009;38(6):1228–34.
Austin PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med. 2009;28(25):3083–107.
Pilon D, Joshi K, Tandon N, Lafeuille MH, Kamstra RL, Emond B, Lefebvre P. Treatment patterns in Medicaid patients with schizophrenia initiated on a first-or second-generation long-acting injectable versus oral antipsychotic. Patient Prefer Adherence. 2017;11:619.
Greene M, Yan T, Chang E, et al. Medication adherence and discontinuation of long-acting injectable versus oral antipsychotics in patients with schizophrenia or bipolar disorder. J Med Econ. 2018;21(2):127–34.
Brissos S, Veguilla MR, Taylor D, Balanzá-Martinez V. The role of long-acting injectable antipsychotics in schizophrenia: a critical appraisal. Ther Adv Psychopharmacol. 2014;4(5):198–219.
Ascher-Svanum H, Zhu B, Faries DE, Lacro JP, Dolder CR, Peng X. Adherence and persistence to typical and atypical antipsychotics in the naturalistic treatment of patients with schizophrenia. Patient Prefer Adherence. 2008;2:67–77.
Baser O, **e L, Pesa J, Durkin M. Healthcare utilization and costs of Veterans Health Administration patients with schizophrenia treated with paliperidone palmitate long-acting injection or oral atypical antipsychotics. J Med Econ. 2015;18(5):357–65.
Taipale, H., Mehtälä, J., Tanskanen, A., & Tiihonen, J. (2017). Comparative effectiveness of antipsychotic drugs for rehospitalization in schizophrenia—a nationwide study with 20-year follow-up. Schizophr Bull.
Tiihonen J, Mittendorfer-Rutz E, Majak M, Mehtälä J, Hoti F, Jedenius E, Taipale H. Real-world effectiveness of antipsychotic treatments in a nationwide cohort of 29 823 patients with schizophrenia. JAMA Psychiatry. 2017;74(7):686–93.
Wu EQ, Birnbaum HG, Shi L, et al. The economic burden of schizophrenia in the United States in 2002. J Clin Pyschiatry. 2005;66(9):1122–9.
Acknowledgements
Funding
This study and the accompanying article processing charges were funded by Alkermes Inc. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analyses.
Medical Writing and/or Editorial Assistance
Editorial support was provided by Michael Moriarty of STATinMED Research.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Prior Presentations
This study was presented at the following conferences: the US Psychiatric and Mental Health Congress (USPC) New Orleans, LA, USA (Sep 16–19, 2017) and Academy Managed Care Pharmacy (AMCP) Nexus, Dallas, TX, USA (Oct 16–19, 2017).
Disclosure Statement
Ankit Shah is a full-time employee and a minor shareholder of Alkermes, Inc. Mugdha Gore was employed at Alkermes, Inc. when the study was conducted. Mugdha Gore is currently the President & CEO of Avalon Health Solutions, Inc and Founder of Samsara Healthcare Inc. Lin **e is a full-time employee of STATinMED Research which is a paid consultant to Alkermes, Inc. Furaha Kariburyo is a full-time employee of STATinMED Research which is a paid consultant to Alkermes, Inc. Qisu Zhang is a full-time employee of STATinMED Research which is a paid consultant to Alkermes, Inc.
Compliance with Ethics Guidelines
This article is based on retrospective administrative claims data and does not contain any studies with human participants or animals performed by any of the authors
Data Availability
The datasets generated and/or analyzed during the current study are not publicly available due to a data licensing agreement with Truven Health Analytics.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced digital content
To view enhanced digital content for this article go to https://doi.org/10.6084/m9.figshare.7028381.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shah, A., **e, L., Kariburyo, F. et al. Treatment Patterns, Healthcare Resource Utilization and Costs Among Schizophrenia Patients Treated with Long-Acting Injectable Versus Oral Antipsychotics. Adv Ther 35, 1994–2014 (2018). https://doi.org/10.1007/s12325-018-0786-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-018-0786-x