Abstract
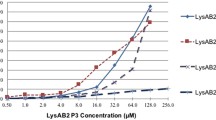
Antimicrobial agents targeting peptidoglycan have shown successful results in eliminating bacteria with high selective toxicity. Bacteriophage encoded endolysin as an alternative antibiotics is a peptidoglycan degrading enzyme with a low rate of resistance. Here, the engineered endolysin was developed to defeat multiple drug-resistant (MDR) Acinetobacter baumannii. First, putative endolysin PA90 was predicted by genome analysis of isolated Pseudomonas phage PBPA. The His-tagged PA90 was purified from BL21(DE3) pLysS and tested for the enzymatic activity using Gram-negative pathogens known for having a high antibiotic resistance rate including A. baumannii. Since the measured activity of PA90 was low, probably due to the outer membrane, cell-penetrating peptide (CPP) DS4.3 was introduced at the N-terminus of PA90 to aid access to its substrate. This engineered endolysin, DS-PA90, completely killed A. baumannii at 0.25 µM, at which concentration PA90 could only eliminate less than one log in CFU/ml. Additionally, DS-PA90 has tolerance to NaCl, where the ∼50% of activity could be maintained in the presence of 150 mM NaCl, and stable activity was also observed with changes in pH or temperature. Even MDR A. baumannii strains were highly susceptible to DS-PA90 treatment: five out of nine strains were entirely killed and four strains were reduced by 3–4 log in CFU/ml. Consequently, DS-PA90 could protect waxworm from A. baumannii-induced death by ∼70% for ATCC 17978 or ∼44% for MDR strain 1656-2 infection. Collectively, our data suggest that CPP-fused endolysin can be an effective antibacterial agent against Gram-negative pathogens regardless of antibiotics resistance mechanisms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Azeredo, J. and Sutherland, I.W. 2008. The use of phages for the removal of infectious biofilms. Curr. Pharm. Biotechnol. 9, 261–266.
Baaijens, J.A., Aabidine, A.Z.E., Rivals, E., and Schönhuth, A. 2017. De novo assembly of viral quasispecies using overlap graphs. Genome Res. 27, 835–848.
Bhargava, K., Nath, G., Bhargava, A., Aseri, G.K., and Jain, N. 2021. Phage therapeutics: from promises to practices and prospectives. Appl. Microbiol. Biotechnol. 105, 9047–9067.
Blasco, L., Bleriot, I., González de Aledo, M., Fernández-García, L., Pacios, O., Oliveira, H., López, M., Ortiz-Cartagena, C., Fernández-Cuenca, F., Pascual, Á., et al. 2022. Development of an anti-Acinetobacter baumannii biofilm phage cocktail: genomic adaptation to the host. Antimicrob. Agents Chemother. 66, e0192321.
Chen, H., Battalapalli, D., Draz, M.S., Zhang, P., and Ruan, Z. 2021. The application of cell-penetrating-peptides in antibacterial agents. Curr. Med. Chem. 28, 5896–5925.
Cheng, Q., Nelson, D., Zhu, S., and Fischetti, V.A. 2005. Removal of group B streptococci colonizing the vagina and oropharynx of mice with a bacteriophage lytic enzyme. Antimicrob. Agents Chemother. 49, 111–117.
Dams, D. and Briers, Y. 2019. Enzybiotics: Enzyme-Based Antibacterials as Therapeutics. In Labrou, N. (eds), Therapeutic Enzymes: Function and Clinical Implications. Advances in Experimental Medicine and Biology, vol. 1148, pp. 233–253. Springer, Singapore.
De Maesschalck, V., Gutiérrez, D., Paeshuyse, J., Lavigne, R., and Briers, Y. 2020. Advanced engineering of third-generation lysins and formulation strategies for clinical applications. Crit. Rev. Microbiol. 46, 548–564.
Derakhshankhah, H. and Jafari, S. 2018. Cell penetrating peptides: a concise review with emphasis on biomedical applications. Biomed. Pharmacother. 108, 1090–1096.
Drexelius, M., Reinhardt, A., Grabeck, J., Cronenberg, T., Nitsche, F., Huesgen, P.F., Maier, B., and Neundorf, I. 2021. Multistep optimization of a cell-penetrating peptide towards its antimicrobial activity. Biochem. J. 478, 63–78.
Fenton, M., Ross, P., McAuliffe, O., O’Mahony, J., and Coffey, A. 2010. Recombinant bacteriophage lysins as antibacterials. Bioeng. Bugs 1, 9–16.
Fischetti, V.A. 2010. Bacteriophage endolysins: a novel anti-infective to control Gram-positive pathogens. Int. J. Med. Microbiol. 300, 357–362.
Frimodt-Møller, J., Campion, C., Nielsen, P.E., and Løbner-Olesen, A. 2022. Translocation of non-lytic antimicrobial peptides and bacteria penetrating peptides across the inner membrane of the bacterial envelope. Curr. Genet. 68, 83–90.
Hong, H.W., Kim, Y.D., Jang, J., Kim, M.S., Song, M., and Myung, H. 2022. Combination effect of engineered endolysin EC340 with antibiotics. Front. Microbiol. 13, 821936.
Jeong, J.H., Kim, K., Lim, D., Jeong, K., Hong, Y., Nguyen, V.H., Kim, T.H., Ryu, S., Lim, J.A., Kim, J.I., et al. 2014. Anti-tumoral effect of the mitochondrial target domain of Noxa delivered by an engineered Salmonella Typhimurium. PLoS ONE 9, e80050.
John, C.M., Li, M., Feng, D., and Jarvis, G.A. 2019. Cationic cell-penetrating peptide is bactericidal against Neisseria gonorrhoeae. J. Antimicrob. Chemother. 74, 3245–3251.
Kelley, L.A. and Sternberg, M.J.E. 2009. Protein structure prediction on the Web: a case study using the Phyre server. Nat. Protoc. 4, 363–371.
Kim, M.S., Kim, Y.D., Hong, S.S., Park, K., Ko, K.S., and Myung, H. 2015. Phage-encoded colanic acid-degrading enzyme permits lytic phage infection of a capsule-forming resistant mutant Escherichia coli strain. Appl. Environ. Microbiol. 81, 900–909.
Lai, W.C.B., Chen, X., Ho, M.K.Y., **a, J., and Leung, S.S.Y. 2020. Bacteriophage-derived endolysins to target Gram-negative bacteria. Int. J. Pharm. 589, 119833.
Lee, S.H., Kim, D.W., Kim, H.R., Woo, S.J., Kim, S.M., Jo, H.S., Jeon, S.G., Cho, S.W., Park, J.H., Won, M.H., et al. 2012. Anti-inflammatory effects of Tat-Annexin protein on ovalbumin-induced airway inflammation in a mouse model of asthma. Biochem. Biophys. Res. Commun. 417, 1024–1029.
Lee, C.R., Lee, J.H., Park, M., Park, K.S., Bae, I.K., Kim, Y.B., Cha, C.J., Jeong, B.C., and Lee, S.H. 2017. Biology of Acinetobacter baumannii: pathogenesis, antibiotic resistance mechanisms, and prospective treatment options. Front. Cell. Infect. Microbiol. 7, 55.
Lin, M.F. and Lan, C.Y. 2014. Antimicrobial resistance in Acinetobacter baumannii: from bench to bedside. World J. Clin. Cases 2, 787–814.
Loeffler, J.M., Nelson, D., and Fischetti, V.A. 2001. Rapid killing of Streptococcus pneumoniae with a bacteriophage cell wall hydrolase. Science 294, 2170–2172.
Meile, S., Du, J., Dunne, M., Kilcher, S., and Loessner, M.J. 2022. Engineering therapeutic phages for enhanced antibacterial efficacy. Curr. Opin. Virol. 52, 182–191.
Nelson, D., Loomis, L., and Fischetti, V.A. 2001. Prevention and elimination of upper respiratory colonization of mice by group A streptococci by using a bacteriophage lytic enzyme. Proc. Natl. Acad. Sci. USA 98, 4107–4112.
Oikawa, K., Islam, M.M., Horii, Y., Yoshizumi, T., and Numata, K. 2018. Screening of a cell-penetrating peptide library in Escherichia coli: relationship between cell penetration efficiency and cytotoxicity. ACS Omega 3, 16489–16499.
Pagano, M., Martins, A.F., and Barth, A.L. 2016. Mobile genetic elements related to carbapenem resistance in Acinetobacter baumannii. Braz. J. Microbiol. 47, 785–792.
Park, J.Y., Kim, S., Kim, S.M., Cha, S.H., Lim, S.K., and Kim, J. 2011. Complete genome sequence of multidrug-resistant Acinetobacter baumannii strain 1656-2, which forms sturdy biofilm. J. Bacteriol. 193, 6393–6394.
Quevillon, E., Silventoinen, V., Pillai, S., Harte, N., Mulder, N., Apweiler, R., and Lopez, R. 2005. InterProScan: protein domains identifier. Nucleic Acids Res. 33, W116–W120.
Rüter, C. 2022. Delivery of antibiotics by cell-penetrating peptides to kill intracellular pathogens. Methods Mol. Biol. 2383, 335–345.
Sadeghian, I., Heidari, R., Sadeghian, S., Raee, M.J., and Negahdaripour, M. 2022. Potential of cell-penetrating peptides (CPPs) in delivery of antiviral therapeutics and vaccines. Eur. J. Pharm. Sci. 169, 106094.
Schmelcher, M. and Loessner, M.J. 2021. Bacteriophage endolysins — extending their application to tissues and the bloodstream. Curr. Opin. Biotechnol. 68, 51–59.
Seijsing, J., Sobieraj, A.M., Keller, N., Shen, Y., Zinkernagel, A.S., Loessner, M.J., and Schmelcher, M. 2018. Improved biodistribution and extended serum half-life of a bacteriophage endolysin by albumin binding domain fusion. Front. Microbiol. 9, 2927.
Sisakhtpour, B., Mirzaei, A., Karbasizadeh, V., Hosseini, N., Shabani, M., and Moghim, S. 2022. The characteristic and potential therapeutic effect of isolated multidrug-resistant Acinetobacter baumannii lytic phage. Ann. Clin. Microbiol. Antimicrob. 21, 1.
Sobieraj, A.M., Huemer, M., Zinsli, L.V., Meile, S., Keller, A.P., Röhrig, C., Eichenseher, F., Shen, Y., Zinkernagel, A.S., Loessner, M.J., et al. 2020. Engineering of long-circulating peptidoglycan hydrolases enables efficient treatment of systemic Staphylococcus aureus infection. mBio 11, e01781–20.
Walsh, L., Johnson, C.N., Hill, C., and Ross, R.P. 2021. Efficacy of phage- and bacteriocin-based therapies in combatting nosocomial MRSA infections. Front. Mol. Biosci. 8, 654038.
Xuan, G., Lin, H., and Wang, J. 2022. Expression of a phage-encoded Gp21 protein protects Pseudomonas aeruginosa against phage infection. J. Virol. 96, e0176921.
Acknowledgements
This work was supported by the National Research Foundation of Korea grant funded by the Korean government (2021R1F1A1060072 and 2019M3E5D5066666), and by the Hankuk University of Foreign Studies Research Fund (of 2022).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors have no conflict of interest to report.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lim, J., Jang, J., Myung, H. et al. Eradication of drug-resistant Acinetobacter baumannii by cell-penetrating peptide fused endolysin. J Microbiol. 60, 859–866 (2022). https://doi.org/10.1007/s12275-022-2107-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-022-2107-y