Abstract
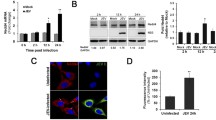
Japanese encephalitis virus (JEV) is a flavivirus transmitted by mosquitoes that causes severe encephalitis in humans and animals. It has been suggested that AXL, a transmembrane protein, can promote the replication of various flaviviruses, such as dengue (DENV), Zika (ZIKV), and West Nile (WNV) viruses. However, the effect of AXL on JEV infection has not yet been determined. In the present study, we demonstrate that AXL is down-regulated after JEV infection in the late stage. JEV NS2B-3 protein specifically interacted with AXL, and promoted AXL degradation through the ubiquitin–proteasome pathway. AXL-degradation increased cell apoptosis by disrupting phosphatidylinositol 3-kinase (PI3K)/Akt signal transduction. In addition, the degradation of AXL promoted JEV release to supernatant, whereas the virus in the cell lysates decreased. The supplementation of AXL ligand Gas6 inhibited the JEV-mediated degradation of AXL. Altogether, we discover a new function of NS2B-3 during the process of JEV replication, and provide a new insight into the interactions between JEV and cell hosts.






Similar content being viewed by others
References
Aguirre S, Maestre AM, Pagni S, Patel JR, Savage T, Gutman D, Maringer K, Bernal-Rubio D, Shabman RS, Simon V, Rodriguez-Madoz JR, Mulder LC, Barber GN, Fernandez-Sesma A (2012) DENV inhibits type I IFN production in infected cells by cleaving human STING. PLoS Pathog 8:e0002934
Anderson MR, Kashanchi F, Jacobson S (2016) Exosomes in viral disease. Neurotherapeutics 13:535–546
Barrows NJ, Campos RK, Liao KC, Prasanth KR, Soto-Acosta R, Yeh SC, Schott-Lerner G, Pompon J, Sessions OM, Bradrick SS, Garcia-Blanco MA (2018) Biochemistry and molecular biology of flaviviruses. Chem Rev 118:4448–4482
Bhattacharyya S, Zagorska A, Lew ED, Shrestha B, Rothlin CV, Naughton J, Diamond MS, Lemke G, Young JA (2013) Enveloped viruses disable innate immune responses in dendritic cells by direct activation of TAM receptors. Cell Host Microbe 14:136–147
Brazil DP, Yang ZZ, Hemmings BA (2004) Advances in protein kinase B signalling: AKTion on multiple fronts. Trends Biochem Sci 29:233–242
Campbell GL, Hills SL, Fischer M, Jacobson JA, Hoke CH, Hombach JM, Marfin AA, Solomon T, Tsai TF, Tsu VD, Ginsburg AS (2011) Estimated global incidence of Japanese encephalitis: a systematic review. Bull World Health Organ 89:766–774
Caobi A, Nair M, Raymond AD (2020) Extracellular vesicles in the pathogenesis of viral infections in humans. Viruses 12:1200
Chen YH, Du W, Hagemeijer MC, Takvorian PM, Pau C, Cali A, Brantner CA, Stempinski ES, Connelly PS, Ma HC, Jiang P, Wimmer E, Altan-Bonnet G, Altan-Bonnet N (2015) Phosphatidylserine vesicles enable efficient en bloc transmission of enteroviruses. Cell 160:619–630
Chen S, Wu Z, Wang M, Cheng A (2017) Innate immune evasion mediated by flaviviridae non-structural proteins. Viruses 9:291
Chen J, Yang YF, Yang Y, Zou P, Chen J, He Y, Shui SL, Cui YR, Bai R, Liang YJ, Hu Y, Jiang B, Lu L, Zhang X, Liu J, Xu J (2018) AXL promotes Zika virus infection in astrocytes by antagonizing type I interferon signalling. Nat Microbiol 3:302–309
Ding Q, Gaska JM, Douam F, Wei L, Kim D, Balev M, Heller B, Ploss A (2018) Species-specific disruption of STING-dependent antiviral cellular defenses by the Zika virus NS2B3 protease. Proc Natl Acad Sci USA 115:E6310–E6318
Fan YC, Liang JJ, Chen JM, Lin JW, Chen YY, Su KH, Lin CC, Tu WC, Chiou MT, Ou SC, Chang GJ, Lin YL, Chiou SS (2019) NS2B/NS3 mutations enhance the infectivity of genotype I Japanese encephalitis virus in amplifying hosts. PLoS Pathog 15:e1007992
Filgueira L, Lannes N (2019) Review of emerging Japanese encephalitis virus: new aspects and concepts about entry into the brain and inter-cellular spreading. Pathogens 8:111
Gordon TB, Hayward JA, Marsh GA, Baker ML, Tachedjian G (2019) Host and viral proteins modulating ebola and marburg virus egress. Viruses 11:25
Hamel R, Dejarnac O, Wichit S, Ekchariyawat P, Neyret A, Luplertlop N, Perera-Lecoin M, Surasombatpattana P, Talignani L, Thomas F, Cao-Lormeau VM, Choumet V, Briant L, Despres P, Amara A, Yssel H, Misse D (2015) Biology of Zika virus infection in human skin cells. J Virol 89:8880–8896
Hastings AK, Yockey LJ, Jagger BW, Hwang J, Uraki R, Gaitsch HF, Parnell LA, Cao B, Mysorekar IU, Rothlin CV, Fikrig E, Diamond MS, Iwasaki A (2017) TAM receptors are not required for Zika virus infection in mice. Cell Rep 19:558–568
Hastings AK, Hastings K, Uraki R, Hwang J, Gaitsch H, Dhaliwal K, Williamson E, Fikrig E (2019) Loss of the TAM receptor Axl ameliorates severe zika virus pathogenesis and reduces apoptosis in microglia. iScience 13:339–350
Kumar A, Jovel J, Lopez-Orozco J, Limonta D, Airo AM, Hou S, Stryapunina I, Fibke C, Moore RB, Hobman TC (2018) Human Sertoli cells support high levels of Zika virus replication and persistence. Sci Rep 8:5477
Lauter M, Weber A, Torka R (2019) Targeting of the AXL receptor tyrosine kinase by small molecule inhibitor leads to AXL cell surface accumulation by impairing the ubiquitin-dependent receptor degradation. Cell Commun Signal 17:59
Lei J, Hansen G, Nitsche C, Klein CD, Zhang L, Hilgenfeld R (2016) Crystal structure of Zika virus NS2B-NS3 protease in complex with a boronate inhibitor. Science 353:503–505
Lemke G (2013) Biology of the TAM receptors. Cold Spring Harb Perspect Biol 5:a009076
Lemke G (2019) How macrophages deal with death. Nat Rev Immunol 19:539–549
Lemke G, Rothlin CV (2008) Immunobiology of the TAM receptors. Nat Rev Immunol 8:327–336
Lennemann NJ, Coyne CB (2017) Dengue and Zika viruses subvert reticulophagy by NS2B3-mediated cleavage of FAM134B. Autophagy 13:322–332
Li M, Ablan SD, Miao C, Zheng YM, Fuller MS, Rennert PD, Maury W, Johnson MC, Freed EO, Liu SL (2014) TIM-family proteins inhibit HIV-1 release. Proc Natl Acad Sci USA 111:E3699–E3707
Li M, Waheed AA, Yu J, Zeng C, Chen HY, Zheng YM, Feizpour A, Reinhard BM, Gummuluru S, Lin S, Freed EO, Liu SL (2019) TIM-mediated inhibition of HIV-1 release is antagonized by Nef but potentiated by SERINC proteins. Proc Natl Acad Sci USA 116:5705–5714
Li M, Ye J, Zhao G, Hong G, Hu X, Cao K, Wu Y, Lu Z (2019) Gas6 attenuates lipopolysaccharideinduced TNFalpha expression and apoptosis in H9C2 cells through NFkappaB and MAPK inhibition via the Axl/PI3K/Akt pathway. Int J Mol Med 44:982–994
Li W, Li N, Dai S, Hou G, Guo K, Chen X, Yi C, Liu W, Deng F, Wu Y, Cao X (2019) Zika virus circumvents host innate immunity by targeting the adaptor proteins MAVS and MITA. FASEB J 33:9929–9944
Liu H, Zhang L, Sun J, Chen W, Li S, Wang Q, Yu H, **a Z, ** X, Wang C (2017) Endoplasmic reticulum protein SCAP inhibits dengue virus NS2B3 protease by suppressing its K27-linked polyubiquitylation. J Virol 91:e02234-e2316
Martins Sde T, Silveira GF, Alves LR, Duarte dos Santos CN, Bordignon J (2012) Dendritic cell apoptosis and the pathogenesis of dengue. Viruses 4:2736–2753
Meertens L, Carnec X, Lecoin MP, Ramdasi R, Guivel-Benhassine F, Lew E, Lemke G, Schwartz O, Amara A (2012) The TIM and TAM families of phosphatidylserine receptors mediate dengue virus entry. Cell Host Microbe 12:544–557
Morizono K, **e Y, Olafsen T, Lee B, Dasgupta A, Wu AM, Chen IS (2011) The soluble serum protein Gas6 bridges virion envelope phosphatidylserine to the TAM receptor tyrosine kinase Axl to mediate viral entry. Cell Host Microbe 9:286–298
Morrison J, Aguirre S, Fernandez-Sesma A (2012) Innate immunity evasion by Dengue virus. Viruses 4:397–413
Neufeldt CJ, Cortese M, Acosta EG, Bartenschlager R (2018) Rewiring cellular networks by members of the Flaviviridae family. Nat Rev Microbiol 16:125–142
Okamoto T, Suzuki T, Kusakabe S, Tokunaga M, Hirano J, Miyata Y, Matsuura Y (2017) Regulation of apoptosis during flavivirus infection. Viruses 9:243
Retallack H, Di Lullo E, Arias C, Knopp KA, Laurie MT, Sandoval-Espinosa C, Mancia Leon WR, Krencik R, Ullian EM, Spatazza J, Pollen AA, Mandel-Brehm C, Nowakowski TJ, Kriegstein AR, DeRisi JL (2016) Zika virus cell tropism in the develo** human brain and inhibition by azithromycin. Proc Natl Acad Sci USA 113:14408–14413
Richard AS, Shim BS, Kwon YC, Zhang R, Otsuka Y, Schmitt K, Berri F, Diamond MS, Choe H (2017) AXL-dependent infection of human fetal endothelial cells distinguishes Zika virus from other pathogenic flaviviruses. Proc Natl Acad Sci USA 114:2024–2029
Rodriguez-Madoz JR, Belicha-Villanueva A, Bernal-Rubio D, Ashour J, Ayllon J, Fernandez-Sesma A (2010) Inhibition of the type I interferon response in human dendritic cells by dengue virus infection requires a catalytically active NS2B3 complex. J Virol 84:9760–9774
Rothlin CV, Ghosh S, Zuniga EI, Oldstone MB, Lemke G (2007) TAM receptors are pleiotropic inhibitors of the innate immune response. Cell 131:1124–1136
Rothlin CV, Carrera-Silva EA, Bosurgi L, Ghosh S (2015) TAM receptor signaling in immune homeostasis. Annu Rev Immunol 33:355–391
Samuel MA, Morrey JD, Diamond MS (2007) Caspase 3-dependent cell death of neurons contributes to the pathogenesis of West Nile virus encephalitis. J Virol 81:2614–2623
Savidis G, McDougall WM, Meraner P, Perreira JM, Portmann JM, Trincucci G, John SP, Aker AM, Renzette N, Robbins DR, Guo Z, Green S, Kowalik TF, Brass AL (2016) Identification of Zika virus and dengue virus dependency factors using functional genomics. Cell Rep 16:232–246
Servet-Delprat C, Vidalain PO, Azocar O, Le Deist F, Fischer A, Rabourdin-Combe C (2000) Consequences of Fas-mediated human dendritic cell apoptosis induced by measles virus. J Virol 74:4387–4393
Slon Campos JL, Mongkolsapaya J, Screaton GR (2018) The immune response against flaviviruses. Nat Immunol 19:1189–1198
Solomon T (2006) Control of Japanese encephalitis–within our grasp? N Engl J Med 355:869–871
Strange DP, Jiyarom B, Pourhabibi Zarandi N, **e X, Baker C, Sadri-Ardekani H, Shi PY, Verma S (2019) Axl promotes zika virus entry and modulates the antiviral state of human sertoli cells. Bio 10:e01372-19
Tabata T, Petitt M, Puerta-Guardo H, Michlmayr D, Wang C, Fang-Hoover J, Harris E, Pereira L (2016) Zika virus targets different primary human placental cells, suggesting two routes for vertical transmission. Cell Host Microbe 20:155–166
Wang J, Li X, Gu J, Fan Y, Zhao P, Cao R, Chen P (2015) The A66G back mutation in NS2A of JEV SA14-14-2 strain contributes to production of NS1’ protein and the secreted NS1’ can be used for diagnostic biomarker for virulent virus infection. Infect Genet Evol 36:116–125
Wang ZY, Wang Z, Zhen ZD, Feng KH, Guo J, Gao N, Fan DY, Han DS, Wang PG, An J (2017) Axl is not an indispensable factor for Zika virus infection in mice. J Gen Virol 98:2061–2068
Wang ZY, Zhen ZD, Fan DY, Qin CF, Han DS, Zhou HN, Wang PG, An J (2020) Axl deficiency promotes the neuroinvasion of Japanese encephalitis virus by enhancing IL-1alpha production from pyroptotic macrophages. J Virol 94:e00602-e620
Wells MF, Salick MR, Wiskow O, Ho DJ, Worringer KA, Ihry RJ, Kommineni S, Bilican B, Klim JR, Hill EJ, Kane LT, Ye C, Kaykas A, Eggan K (2016) GeneticAblation of AXL does not protect human neural progenitor cells and cerebral organoids from Zika Virus infection. Cell Stem Cell 19:703–708
Wu Y, Liu Q, Zhou J, **e W, Chen C, Wang Z, Yang H, Cui J (2017) Zika virus evades interferon-mediated antiviral response through the co-operation of multiple nonstructural proteins in vitro. Cell Discov 3:17006
**ng H, Xu S, Jia F, Yang Y, Xu C, Qin C, Shi L (2020) Zika NS2B is a crucial factor recruiting NS3 to the ER and activating its protease activity. Virus Res 275:197793
Yang TC, Shiu SL, Chuang PH, Lin YJ, Wan L, Lan YC, Lin CW (2009) Japanese encephalitis virus NS2B-NS3 protease induces caspase 3 activation and mitochondria-mediated apoptosis in human medulloblastoma cells. Virus Res 143:77–85
Zhou W, Woodson M, Neupane B, Bai F, Sherman MB, Choi KH, Neelakanta G, Sultana H (2018) Exosomes serve as novel modes of tick-borne flavivirus transmission from arthropod to human cells and facilitates dissemination of viral RNA and proteins to the vertebrate neuronal cells. PLoS Pathog 14:e1006764
Zhou W, Woodson M, Sherman MB, Neelakanta G, Sultana H (2019) Exosomes mediate Zika virus transmission through SMPD3 neutral Sphingomyelinase in cortical neurons. Emerg Microbes Infect 8:307–326
Zizzo G, Hilliard BA, Monestier M, Cohen PL (2012) Efficient clearance of early apoptotic cells by human macrophages requires M2c polarization and MerTK induction. J Immunol 189:3508–3520
Acknowledgements
This work was carried out with support of grants from the National Key Research and Development Plan of China (Grant No. 2016YFD0500402), the National Natural Science Foundation of China (Grant No. 31772756), and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Contributions
SDX and RBC designed the experiments. SDX, ZJL, XMY, and TTL carried out the experiments. SDX and RBC analyzed the data. JHP and YD provided constructive suggestions. SDX and RBC wrote the paper. SDX and RBC checked and finalized the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Animal and Human Rights Statement
The isolation experiments of primary peritoneal macrophages cells were conducted under the guidelines of the regional Animal Ethics Committee and the rules for experimental animals of Nan**g Agricultural University.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
**e, S., Liang, Z., Yang, X. et al. Japanese Encephalitis Virus NS2B-3 Protein Complex Promotes Cell Apoptosis and Viral Particle Release by Down-Regulating the Expression of AXL. Virol. Sin. 36, 1503–1519 (2021). https://doi.org/10.1007/s12250-021-00442-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12250-021-00442-3