Abstract
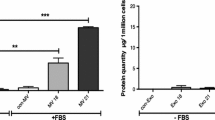
During the last stages of wound healing, myofibroblasts differentiate mainly from fibroblasts. Myofibroblasts from normal skin wounds (Wmyo) can communicate with its surrounding using secreted factors. They also have the capacity to produce microvesicles (MVs), a type of extracellular vesicles, as mediators of intercellular communication. MVs cargo are potentially capable of regulating the behavior of targeted cells and tissues. The aim of this study is to evaluate the effect of Wmyo-derived MVs on dermal fibroblasts and to determine the responsible signaling molecule. Microvesicles were obtained from culture media of myofibroblasts and characterized using protein quantification, dynamic light scattering and transmission electron microscopy. Uptake of fluorescent MVs in fibroblasts was assessed by flow cytometry. Cytokines concentrations were quantified in MV samples by a multiplex ELISA. Different concentration of MVs or a selected cytokine were used as treatments over fibroblasts culture for 5 days. Following the treatments, parameters linked to the extracellular matrix were studied. Lastly, the selected cytokine was neutralized within MVs before evaluating collagen production. We showed that Wmyo derived-MVs were internalized by dermal fibroblasts. Cytokine array analysis revealed that a large amount of placental growth factor 1 (PLGF-1) (0.88 ± 0.63 pg/μg proteins in MVs) could be detected in MVs samples. Cutaneous fibroblasts treated with MVs or PLGF-1 showed significantly stimulated procollagen I level production (Fold change of 1.80 ± 0.18 and 2.07 ± 0.18, respectively). Finally, the neutralization of PLGF-1 in MVs significantly inhibited the production of procollagen I by fibroblasts. Our study shows that Wmyo derived-MVs are involved in intercellular communication by stimulating collagen production by fibroblasts during wound healing. This effect is possibly attained through PLGF-1 signalling. These findings represent a promising opportunity to gain insight into how MVs and Wmyo may mediate the healing of a skin wound.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code Availability
Not applicable.
Abbreviations
- Wmyo:
-
Myofibroblasts from normal skin wounds
- MVs:
-
microvesicles
- PLGF-1:
-
Placental growth factor 1
- ECM:
-
Extracellular matrix
- TGFß1:
-
Transforming growth factor-ß
- α-SMA:
-
Alpha-Smooth Muscle Actin
- A2M:
-
Alpha-2-macroglobulin
- MMPs:
-
Matrix metalloproteinases
- DME:
-
Dulbecco’s Modified Eagle’s medium
- FBS:
-
Fetal bovine serum
- RFP-670:
-
Red Fluorescent Protein
- PBS:
-
Phosphate-buffered saline
- DLS:
-
Dynamic light scattering
- PLGF-1-NAb:
-
PLGF-1 neutralizing antibodies
- LTA:
-
Lymphotoxin alpha/ tumor necrosis beta
References
Adini A, Kornaga T, Firoozbakht F, Benjamin LE (2002) Placental growth factor is a survival factor for tumor endothelial cells and macrophages. Cancer Res 62(10):2749–2752
Bainbridge, P. (2013). Wound healing and the role of fibroblasts. J Wound Care, 22(8), 407-408, 410-412. https://doi.org/10.12968/jowc.2013.22.8.407
Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M (2008) Growth factors and cytokines in wound healing. Wound Repair Regen 16(5):585–601. https://doi.org/10.1111/j.1524-475X.2008.00410.x
Baum J, Duffy HS (2011) Fibroblasts and Myofibroblasts: what are we talking about? J Cardiovasc Pharmacol 57(4):376–379. https://doi.org/10.1097/FJC.0b013e3182116e39
Burger D, Montezano AC, Nishigaki N, He Y, Carter A, Touyz RM (2011) Endothelial microparticle formation by angiotensin II is mediated via Ang II receptor type I/NADPH oxidase/ rho kinase pathways targeted to lipid rafts. Arterioscler Thromb Vasc Biol 31(8):1898–1907. https://doi.org/10.1161/atvbaha.110.222703
Carmeliet, P., Moons, L., Luttun, A., Vincenti, V., Compernolle, V., De Mol, M., ... Persico, M. G. (2001). Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med, 7(5), 575–583. doi:https://doi.org/10.1038/87904
Chen L, Chen R, Kemper S, Brigstock DR (2018) Pathways of production and delivery of hepatocyte exosomes. J Cell Commun Signal 12(1):343–357. https://doi.org/10.1007/s12079-017-0421-7
Choi DS, Kim DK, Kim YK, Gho YS (2015) Proteomics of extracellular vesicles: Exosomes and ectosomes. Mass Spectrom Rev 34(4):474–490. https://doi.org/10.1002/mas.21420
Colombo M, Raposo G, Thery C (2014) Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol 30:255–289. https://doi.org/10.1146/annurev-cellbio-101512-122326
Costa Verdera H, Gitz-Francois JJ, Schiffelers RM, Vader P (2017) Cellular uptake of extracellular vesicles is mediated by clathrin-independent endocytosis and macropinocytosis. J Control Release 266:100–108
Cutroneo KR (2003) How is type I procollagen synthesis regulated at the gene level during tissue fibrosis. J Cell Biochem 90(1):1–5. https://doi.org/10.1002/jcb.10599
Darby IA, Laverdet B, Bonte F, Desmouliere A (2014) Fibroblasts and myofibroblasts in wound healing. Clin Cosmet Investig Dermatol 7:301–311. https://doi.org/10.2147/CCID.S50046
Desmouliere A, Geinoz A, Gabbiani F, Gabbiani G (1993) Transforming growth factor-beta 1 induces alpha-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 122(1):103–111
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9(6):669–676. https://doi.org/10.1038/nm0603-669
Fitzgerald W, Freeman ML, Lederman MM, Vasilieva E, Romero R, Margolis L (2018) A system of cytokines encapsulated in ExtraCellular vesicles. Sci Rep 8(1):8973. https://doi.org/10.1038/s41598-018-27190-x
Freitas-Andrade M, Carmeliet P, Charlebois C, Stanimirovic DB, Moreno MJ (2012) PlGF knockout delays brain vessel growth and maturation upon systemic hypoxic challenge. J Cereb Blood Flow Metab 32(4):663–675. https://doi.org/10.1038/jcbfm.2011.167
Germain L, Jean A, Auger FA, Garrel DR (1994) Human wound healing fibroblasts have greater contractile properties than dermal fibroblasts. J Surg Res 57(2):268–273. https://doi.org/10.1006/jsre.1994.1143
Grotendorst GR, Rahmanie H, Duncan MR (2004) Combinatorial signaling pathways determine fibroblast proliferation and myofibroblast differentiation. FASEB J 18(3):469–479
Hattori K, Heissig B, Wu Y, Dias S, Tejada R, Ferris B, Hicklin DJ, Zhu Z, Bohlen P, Witte L, Hendrikx J, Hackett NR, Crystal RG, Moore MAS, Werb Z, Lyden D, Rafii S (2002) Placental growth factor reconstitutes hematopoiesis by recruiting VEGFR1(+) stem cells from bone-marrow microenvironment. Nat Med 8(8):841–849. https://doi.org/10.1038/nm740
Hinz B (2016) Myofibroblasts. Exp Eye Res 142:56–70. https://doi.org/10.1016/j.exer.2015.07.009
Hinz B, Phan SH, Thannickal VJ, Galli A, Bochaton-Piallat ML, Gabbiani G (2007) The Myofibroblast: one function, multiple origins. Am J Pathol 170(6):1807–1816. https://doi.org/10.2353/ajpath.2007.070112
Hromada C, Mühleder S, Grillari J, Redl H, Holnthoner W (2017) Endothelial extracellular vesicles—promises and challenges. Front Physiol 8. https://doi.org/10.3389/fphys.2017.00275
Huang P, Bi J, Owen GR, Chen W, Rokka A, Koivisto L, Heino J, Häkkinen L, Larjava H (2015) Keratinocyte microvesicles regulate the expression of multiple genes in dermal fibroblasts. J Invest Dermatol 135(12):3051–3059. https://doi.org/10.1038/jid.2015.320
Kendall RT, Feghali-Bostwick CA (2014) Fibroblasts in fibrosis: novel roles and mediators. Front Pharmacol 5:123. https://doi.org/10.3389/fphar.2014.00123
Laberge A, Arif S, Moulin VJ (2018) Microvesicles: intercellular messengers in cutaneous wound healing. J Cell Physiol 233(8):5550–5563. https://doi.org/10.1002/jcp.26426
Laberge A, Ayoub A, Arif S, Larochelle S, Garnier A, Moulin VJ (2019) α-2-macroglobulin induces the shedding of microvesicles from cutaneous wound myofibroblasts. J Cell Physiol 234(7):11369–11379. https://doi.org/10.1002/jcp.27794
Liang CC, Park AY, Guan JL (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2(2):329–333. https://doi.org/10.1038/nprot.2007.30
Mehrnaz Gharaee-Kermani SHP (2004) Role of fibroblasts and Myofibroblasts in idiopathic pulmonary fibrosis. In: Lynch JP (ed) Idiopathic pulmonary fibrosis. CRC Press, Boca Raton, pp 507–561
Merjaneh M, Langlois A, Larochelle S, Cloutier CB, Ricard-Blum S, Moulin VJ (2017) Pro-angiogenic capacities of microvesicles produced by skin wound myofibroblasts. Angiogenesis 20(3):385–398. https://doi.org/10.1007/s10456-017-9554-9
Moulin V, Castilloux G, Jean A, Garrel DR, Auger FA, Germain L (1996) In vitro models to study wound healing fibroblasts. Burns 22(5):359–362
Moulin V, Castilloux G, Auger FA, Garrel D, O'Connor-McCourt MD, Germain L (1998) Modulated response to cytokines of human wound healing myofibroblasts compared to dermal fibroblasts. Exp Cell Res 238(1):283–293. https://doi.org/10.1006/excr.1997.3827
Moulin V, Garrel D, Auger FA, O'Connor-McCourt M, Castilloux G, Germain L (1999) What's new in human wound-healing myofibroblasts? Curr Top Pathol 93:123–133
Moulin V, Auger FA, Garrel D, Germain L (2000) Role of wound healing myofibroblasts on re-epithelialization of human skin. Burns 26(1):3–12
Moulin VJ, Mayrand D, Messier H, Martinez MC, Lopez-Valle CA, Genest H (2010) Shedding of microparticles by myofibroblasts as mediator of cellular cross-talk during normal wound healing. J Cell Physiol 225(3):734–740. https://doi.org/10.1002/jcp.22268
Moulin V, Bellemare J, Bergeron D, Genest H, Roy M, Lopez-Vallé C (2012) Myofibroblasts and interactions with other cells: contribution of the tissue engineering. In: Dupuytren’s disease and related Hyperproliferative disorders: principles, research, and clinical perspectives. Springer Berlin Heidelberg, Berlin, pp 69–75
Nagy JA, Dvorak AM, Dvorak HF (2003) VEGF-A(164/165) and PlGF: roles in angiogenesis and arteriogenesis. Trends Cardiovasc Med 13(5):169–175
Parham, P. (2005). T-cell mediated immunity. In The immune system (2nd ed.) (pp. 172). New York: Garland science
Parolini, I., Federici, C., Raggi, C., Lugini, L., Palleschi, S., De Milito, A., ... Fais, S. (2009). Microenvironmental pH is a key factor for exosome traffic in tumor cells. J Biol Chem, 284(49), 34211–34222. https://doi.org/10.1074/jbc.M109.041152
Postlethwaite AE, Shigemitsu H, Kanangat S (2004) Cellular origins of fibroblasts: possible implications for organ fibrosis in systemic sclerosis. Curr Opin Rheumatol 16(6):733–738
Raposo G, Stoorvogel W (2013) Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 200(4):373–383. https://doi.org/10.1083/jcb.201211138
Roy H, Bhardwaj S, Yla-Herttuala S (2006) Biology of vascular endothelial growth factors. FEBS Lett 580(12):2879–2887. https://doi.org/10.1016/j.febslet.2006.03.087
Schonherr E, Hausser HJ (2000) Extracellular matrix and cytokines: a functional unit. Dev Immunol 7(2–4):89–101
Shaw TJ, Martin P (2009) Wound repair at a glance. Journal of Cell Science 122(18):3209–3213. https://doi.org/10.1242/jcs.031187
Stunova A, Vistejnova L (2018) Dermal fibroblasts-a heterogeneous population with regulatory function in wound healing. Cytokine Growth Factor Rev 39:137–150. https://doi.org/10.1016/j.cytogfr.2018.01.003
Sun C, Feng SB, Cao ZW, Bei JJ, Chen Q, Zhao WB, Xu XJ, Zhou Z, Yu ZP, Hu HY (2017) Up-regulated expression of matrix Metalloproteinases in endothelial cells mediates platelet microvesicle-induced angiogenesis. Cell Physiol Biochem 41(6):2319–2332. https://doi.org/10.1159/000475651
Szatanek R, Baran J, Siedlar M, Baj-Krzyworzeka M (2015) Isolation of extracellular vesicles: determining the correct approach (review). Int J Mol Med 36(1):11–17. https://doi.org/10.3892/ijmm.2015.2194
Takeo M, Lee W, Ito M (2015) Wound healing and skin regeneration. Cold Spring Harb Perspect Med 5(1):a023267. https://doi.org/10.1101/cshperspect.a023267
Trinh NT, Yamashita T, Tu TC, Kato T, Ohneda K, Sato F, Ohneda O (2016) Microvesicles enhance the mobility of human diabetic adipose tissue-derived mesenchymal stem cells in vitro and improve wound healing in vivo. Biochem Biophys Res Commun 473(4):1111–1118. https://doi.org/10.1016/j.bbrc.2016.04.025
Werner S, Grose R (2003) Regulation of wound healing by growth factors and cytokines. Physiol Rev, 83(3), 835–870. Retrieved from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12843410, http://physrev.physiology.org/content/physrev/83/3/835.full.pdf
Xue, M., & Jackson, C. J. (2015). Extracellular matrix reorganization during wound healing and its impact on abnormal scarring. In Adv Wound Care (New Rochelle) (Vol. 4, pp. 119-136)
Yanez-Mo M, Siljander PR, Andreu Z, Zavec AB, Borras FE, Buzas EI, ... De Wever O (2015) Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles, 4, 27066. doi:https://doi.org/10.3402/jev.v4.27066
Acknowledgements
The authors thank Caroline Gilbert from the CHU of the Université Laval for the use of the Nanosizer® apparatus, Richard Janvier from IBIS of the Université Laval for the use of their TEM microscope and, Annie Karakeussian-Rimbaud from the CHUM for the handling of the multiplex ELISA device.
Funding
This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) (RGPIN2014–04404); les Fonds de recherche du Québec-Santé (FRQS) (Research Centre funding grant); the Quebec Cell, Tissue and Gene Therapy Network–ThéCell (a thematic network supported by FRQS).
Author information
Authors and Affiliations
Contributions
S.A. performed the experiments and wrote the manuscript, S.L conceived the transduction of Wmyo with a fluorescent protein, V.J.M conducted the project and wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethics approval
All procedures involving patients were reviewed and approved by the Research Ethical Committee of the Centre Hospitalier Universitaire (CHU) de Québec-Université Laval, 2014–04404.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arif, S., Larochelle, S. & Moulin, V.J. PLGF-1 contained in normal wound myofibroblast-derived microvesicles stimulated collagen production by dermal fibroblasts. J. Cell Commun. Signal. 14, 427–438 (2020). https://doi.org/10.1007/s12079-020-00572-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-020-00572-5