Abstract
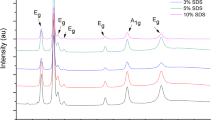
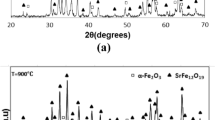
Nano- α-Al2O3 particles were synthesized by a sol–gel method using aqueous solutions of aluminum isopropoxide and 0⋅5 M aluminum nitrate. Sodium dodecylbenzenesulfonate (SDBS) was used as surfactant stabilizing agents. The solution was stirred for 48 h at 60 ∘C. The microstructural observation showed that surfactant with 48 h stirring time nanoscale α-Al2O3 powder was produced at 1200 ∘C in the range of 20–30 nm in the presence of SDBS as a surfactant with 48 h stirring time. Temperature reduction of α-Al2O3 formation has been achieved by introducing fluoride in the alumina precursor. The effects of ZnF2 additive and milling on the phase transformation as well as micrograph of the prepared α-Al2O3 particles were investigated. The samples were characterized by different techniques such as X-ray diffraction, thermogravimetry analysis, differential scanning calorimetry, Fourier transform infrared spectra, scanning electron microscopy and transmission electron microscopy. Results indicated that using zinc fluoride (ZnF2) additive accompanied by milling led to retardation of the transformation temperature and modification of the alumina particle shape. The finest size for nonagglomerated nano-plate-like α-Al2O3 particles (15–20 nm) was achieved at 950 ∘C.












Similar content being viewed by others
References
Arami H, Mazlomi M, Khalifehzadeh R and Sarnezhaad S K 2008, J. Alloys Compd. 461, 551.
Hyuk-Joon J and Kug J 1999, J. Colloid Interface Sci. 211, 110.
Karagedov G R and Lyakhov N Z 1999, Nanostruct. Mater. 11, 559.
Li J, Pan J, **ang C, Ge Q and Guo J 2006, Ceram. Int. 32, 587.
Li J, Wu Y, Pan Y, Liu W and Guo J 2007, Ceram. Int. 33, 919.
Liu L, Zhang L and An 2005, J. Am. Ceram. 88, 2559.
Ming G M, Ying J Z and Zi L X 2007, Mater. Lett. 6, 1812.
Mirjalili F, Hasmaliza M and Chuah L 2010, J. Ceram. Int. 36, 1253.
Mirjalili F, Hasmaliza M and Chuah L 2011, J. Ceram. Silikaty 55, 378.
Park Y K, Tadd E H, Zubris M and Tannenbaum R 2005, Mater. Res. Bull. 40, 1506.
Saito Y, Takei T, Hayashi S, Yasumori A and Okada K 1998, J. Am. Ceram. 81/8, 2197.
Trunov S, Schoenitz M A, Zhu M and Dreizin M 2005, Combust. Flame 140, 310.
Wang H, Gao L, Li W and Li Q 1999, Nanostruct. Mater. 11, 1263.
Wang Y, Suryanarayana C and An L 2005, J. Am. Cream. Soc. 88, 780.
Wang D G, Guo F, Chen J F, Liu H and Zhang Z 2006, Chem. Eng. J. 121, 109.
Wen H L and Yen F S 2000, J. Cryst. Growth 208, 696.
Wu Y Q, Zhang Y F, Huang X X and Guo J K 2001, Ceram. Int. 27, 265.
Xue L A and Chen I W 1992, J. Mater. Sci. Lett. 11, 443.
Yang R, Yen F, Lin S and Chen C 2007, J. Cryst. Growth 299, 429.
Yong C C and Wang J 2001, J. Am. Ceram. 84, 1225.
Acknowledgement
The authors would like to express their gratitude to University Putra Malaysia, Ministry of Science, Technology and Innovation, Government of Malaysia, for funding and supporting this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mirjalili, F. Effect of fluoride additives on production and characterization of nano-α-Al2O3 particles. Bull Mater Sci 37, 1709–1716 (2014). https://doi.org/10.1007/s12034-014-0734-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-014-0734-6