Abstract
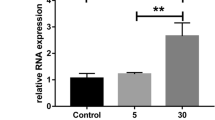
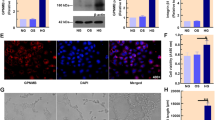
Retinal microangiopathies, such as neovascularization and preretinal and vitreous hemorrhages, are the primary pathological features of diabetic retinopathy (DR). These conditions can worsen visual impairment and may result in blindness. Furthermore, multiple metabolic pathways are associated with microangiopathy in DR. However, the specific underlying pathological mechanisms remain unclear. Several studies have demonstrated the important role of G protein-coupled receptor 124 (Gpr124) in cerebral vascular endothelial cells, but its effect on the retinal endothelium has not been elucidated. In this study, we found that Gpr124 is expressed in both pathological retinal fibrous vascular membranes of DR patients and retinal blood vessels of mice, with elevated protein expression specifically observed in the retinas of DR model mice. Furthermore, Gpr124 expression was elevated after high-glucose treatment of human retinal microvascular endothelial cells (HRMECs). Inhibition of Gpr124 expression affected the high glucose-induced proliferation, migration, and tube-forming ability of HRMECs. We concluded that Gpr124 expression was upregulated in DR and promoted HRMECs angiogenesis in a high-glucose environment. This finding may help to elucidate the pathogenesis of DR and provide a critical research basis for identifying effective treatments.






Similar content being viewed by others
References
Cheung, N., Mitchell, P., & Wong, T. Y. (2010). Diabetic retinopathy. Lancet, 376, 124–136.
Teo, Z. L., Tham, Y. C., Yu, M., Chee, M. L., Rim, T. H., Cheung, N., Bikbov, M. M., Wang, Y. X., Tang, Y., Lu, Y., & Wong, I. Y. (2021). Global prevalence of diabetic retinopathy and projection of burden through 2045. Ophthalmology, 128, 1580–1591.
Hu, Z., Su, Y., **e, P., Chen, L., Ji, J., Feng, T., Wu, S., Liang, K., & Liu, Q. (2019). OCT angiography-based monitoring of neovascular regression on fibrovascular membrane after preoperative intravitreal conbercept injection. Graefes Archive for Clinical and Experimental Ophthalmology, 257, 1611–1619.
Tamaki, K., Usui-Ouchi, A., Murakami, A., & Ebihara, N. (2016). Fibrocytes and fibrovascular membrane formation in proliferative diabetic retinopathy. Investigative Ophthalmology & Visual Science, 57, 4999.
Wong, T. Y., Cheung, C. M. G., Larsen, M., Sharma, S., & Simó, R. (2016). Diabetic retinopathy. Nature Reviews Disease Primers, 2, 16012.
Gonzalez-Cortes, J. H., Martinez-Pacheco, V. A., Gonzalez-Cantu, J. E., Bilgic, A., de Ribot, F. M., Sudhalkar, A., Mohamed-Hamsho, J., Kodjikian, L., & Mathis, T. (2022). Current treatments and innovations in diabetic retinopathy and diabetic macular edema. Pharmaceutics, 15, 122.
Sorour, O. A., Levine, E. S., Baumal, C. R., Elnahry, A. G., Braun, P., Girgis, J., & Waheed, N. K. (2022). Persistent diabetic macular edema: Definition, incidence, biomarkers and treatment methods. Survey of Ophthalmology, 68(2), 147–174.
Brownlee, M. (2001). Biochemistry and molecular cell biology of diabetic complications. Nature, 414, 813–820.
Syrovatkina, V., Alegre, K. O., Dey, R., & Huang, X.-Y. (2016). Regulation, signaling, and physiological functions of G-proteins. Journal of Molecular Biology, 428, 3850–3868.
Bar-Shavit, R., Maoz, M., Kancharla, A., Nag, J. K., Agranovich, D., Grisaru-Granovsky, S., & Uziely, B. (2016). G Protein-coupled receptors in cancer. International Journal of Molecular Sciences, 17, 1320.
Sun, L., & Ye, R. D. (2012). Role of G protein-coupled receptors in inflammation. Acta Pharmacologica Sinica, 33, 342–350.
Zalewska, M., Siara, M., & Sajewicz, W. (2014). G protein-coupled receptors: Abnormalities in signal transmission, disease states and pharmacotherapy. Acta Poloniae Pharmaceutica, 71, 229–243.
De Francesco, E. M., Sotgia, F., Clarke, R. B., Lisanti, M. P., & Maggiolini, M. (2017). G Protein-coupled receptors at the crossroad between physiologic and pathologic angiogenesis: Old paradigms and emerging concepts. International Journal of Molecular Sciences, 18, 2713.
Carson-Walter, E. B., Watkins, D. N., Nanda, A., Vogelstein, B., Kinzler, K. W., & St Croix, B. (2001). Cell surface tumor endothelial markers are conserved in mice and humans. Cancer Research, 61, 6649–6655.
Wang, Y., Cho, S.-G., Wu, X., Siwko, S., & Liu, M. (2014). G-Protein coupled receptor 124 (GPR124) in endothelial cells regulates vascular endothelial growth factor (VEGF)-induced tumor angiogenesis. CMM, 14, 543–554.
Chang, J., Mancuso, M. R., Maier, C., et al. (2017). Gpr124 is essential for blood–brain barrier integrity in central nervous system disease. Nature Medicine, 23, 450–460.
Dejana, E., & Nyqvist, D. (2010). News from the brain: The GPR124 orphan receptor directs brain-specific angiogenesis. Science Translational Medicine. https://doi.org/10.1126/scitranslmed.3001793
Kuhnert, F., Mancuso, M. R., Shamloo, A., et al. (2010). Essential regulation of CNS angiogenesis by the orphan G protein-coupled receptor GPR124. Science, 330, 985–989.
Zhou, Y., & Nathans, J. (2014). Gpr124 controls CNS angiogenesis and blood-brain barrier integrity by promoting ligand-specific canonical wnt signaling. Developmental Cell, 31, 248–256.
Choi, J. S. Y., Haan, J. B., & Sharma, A. (2022). Animal models of diabetes-associated vascular diseases: An update on available models and experimental analysis. British Journal of Pharmacology, 179, 748–769.
Pitale, P. M., & Gorbatyuk, M. S. (2022). Diabetic retinopathy: from animal models to cellular signaling. IJMS, 23, 1487.
Ai, L. Q. Y., Yuan, R. D., Chen, X., Liu, Y. J., Liu, W. Y., Zhu, J. Y., Zhang, Z., Yan, J., Chen, C. L., Lin, S., & Ye, J. (2020). Retinal blood vessel-origin yes-associated protein (YAP) governs astrocytic maturation via leukaemia inhibitory factor (LIF). Cell Proliferation, 53, e12757.
Jian, L., Mei, Y., **ng, C., & Rongdi, Y. (2021). Haem relieves hyperoxia-mediated inhibition of HMEC-1 cell proliferation, migration and angiogenesis by inhibiting BACH1 expression. BMC Ophthalmology, 21, 104.
Du, H., Li, C. H., Gao, R. B., Tan, Y., Wang, B., Peng, Y., Yang, N., Ning, Y. L., Li, P., Zhao, Y., & Zhou, Y. G. (2024). Inhibition of the interaction between microglial adenosine 2A receptor and NLRP3 inflammasome attenuates neuroinflammation posttraumatic brain injury. CNS Neuroscience & Therapeutics, 30, e14408.
Moritz, C. P. (2017). Tubulin or not tubulin: Heading toward total protein staining as loading control in western blots. Proteomics, 17, 1600189.
Colella, A. D., Chegenii, N., Tea, M. N., Gibbins, I. L., Williams, K. A., & Chataway, T. K. (2012). Comparison of stain-free gels with traditional immunoblot loading control methodology. Analytical Biochemistry, 430, 108–110.
Noda, M., Vallon, M., & Kuo, C. J. (2016). The Wnt7’s Tale: A story of an orphan who finds her tie to a famous family. Cancer Science, 107, 576–582.
Zhang, C., Wang, H., Li, P., Hong, C., Chen, A., Qiu, Y., Zeng, A., Zhou, Y., Hu, B., & Li, Y. (2021). Mfsd2a overexpression alleviates vascular dysfunction in diabetic retinopathy. Pharmacological Research, 171, 105755.
Vallon, M., Rohde, F., Janssen, K.-P., & Essler, M. (2010). Tumor endothelial marker 5 expression in endothelial cells during capillary morphogenesis is induced by the small GTPase Rac and mediates contact inhibition of cell proliferation. Experimental Cell Research, 316, 412–421.
Cullen, M., Elzarrad, M. K., Seaman, S., et al. (2011). GPR124, an orphan G protein-coupled receptor, is required for CNS-specific vascularization and establishment of the blood–brain barrier. Proceedings of the National academy of Sciences of the United States of America, 108, 5759–5764.
Vallon, M., & Essler, M. (2006). Proteolytically processed soluble tumor endothelial marker (TEM) 5 mediates endothelial cell survival during angiogenesis by linking integrin alpha(v)beta3 to glycosaminoglycans. Journal of Biological Chemistry, 281, 34179–34188.
Liu, K., Gao, X., Hu, C., Gui, Y., Gui, S., Ni, Q., Tao, L., & Jiang, Z. (2022). Capsaicin ameliorates diabetic retinopathy by inhibiting poldip2-induced oxidative stress. Redox Biology, 56, 102460.
Sacks, D. B., Arnold, M., Bakris, G. L., Bruns, D. E., Horvath, A. R., Lernmark, Å., Metzger, B. E., Nathan, D. M., & Kirkman, M. S. (2023). Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Diabetes Care, 46, e151–e199.
Bandello, F., Corvi, F., La Spina, C., et al. (2016). Outcomes of intravitreal anti-VEGF therapy in eyes with both neovascular age-related macular degeneration and diabetic retinopathy. British Journal of Ophthalmology, 100, 1611–1616.
Ciulla, T. A., Pollack, J. S., & Williams, D. F. (2021). Visual acuity outcomes and anti-VEGF therapy intensity in diabetic macular oedema: A real-world analysis of 28 658 patient eyes. British Journal of Ophthalmology, 105, 216–221.
Tian, R., Liu, Z., Zhang, H., Fang, X., Wang, C., Qi, S., Cheng, Y., & Su, G. (2015). Investigation of the regulation of roundabout4 by hypoxia-inducible factor-1α in microvascular endothelial cells. Investigative Ophthalmology & Visual Science, 56, 2586–2594.
Cho, C., Smallwood, P. M., & Nathans, J. (2017). Reck and Gpr124 are essential receptor cofactors for Wnt7a/Wnt7b-specific signaling in mammalian CNS angiogenesis and blood-brain barrier regulation. Neuron, 95, 1221–1225.
Ta, S., Rong, X., Guo, Z. N., **, H., Zhang, P., Li, F., Li, Z., Lin, L., Zheng, C., Qingquan, G., Zhang, Y., Liu, W., Yang, Yi., & Chang, J. (2021). Variants of WNT7A and GPR124 are associated with hemorrhagic transformation following intravenous thrombolysis in ischemic stroke. CNS Neuroscience & Therapeutics, 27, 71–81.
Vallon, M., Yuki, K., Nguyen, T. D., Chang, J., Yuan, J., Siepe, D., Miao, Y., Essler, M., Makoto Noda, K., Garcia, C., & Kuo, C. J. (2018). A RECK-WNT7 receptor-ligand interaction enables isoform-specific regulation of Wnt bioavailability. Cell Reports, 25, 339-349.e9.
Tan, W., Xu, H., Chen, B., Duan, T., Liu, K., & Zou, J. (2022). Wnt inhibitory 1 ameliorates neovascularization and attenuates photoreceptor injury in an oxygen-induced retinopathy mouse model. BioFactors, 48, 683–698.
Zou, J., Tan, W., Liu, K., Chen, B., Duan, T., & Xu, H. (2022). Wnt inhibitory factor 1 ameliorated diabetic retinopathy through the AMPK/mTOR pathway-mediated mitochondrial function. The FASEB Journal. https://doi.org/10.1096/fj.202200366RR
Funding
This study was supported by grants from the Natural Science Foundation of Chongqing CSTC (CSTB2023NSCQ-MSX0238).
Author information
Authors and Affiliations
Contributions
YW and MY are joint first authors. All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by YW, MY, XW, HZ and XC. The first draft of the manuscript was written by YW and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical Approval
This study have been performed in accordance with the Declaration of Helsinki and was approved by the ethical review committee of the Medical Ethics Committee of the Second Affiliated Hospital of Army Medical University, PLA. Animal Ethical Registration number: AMUWEC20235027. Clinical Trial Ethical Registration number: ChiCTR2300075556.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Yang, M., Wang, X. et al. Role of Gpr124 in the Migration and Proliferation of Retinal Microvascular Endothelial Cells and Microangiopathies in Diabetic Retinopathy. Mol Biotechnol (2024). https://doi.org/10.1007/s12033-024-01210-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-024-01210-w