Abstract
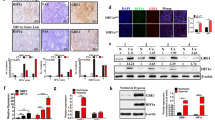
Dysregulated glycolysis promotes growth and metastasis, which is one of the metabolic characteristics of ovarian cancer. Based on bioinformatics analysis, liprin-alpha-4 (PPFIA4) is a gene associated with hypoxia, and we aimed to investigate the potential mechanism of PPFIA4 during the reprogramming of glucose metabolism in ovarian cancer cells. Currently, the cell viability of ovarian cancer cells under the hypoxia treatment was evaluated by CCK-8 assay, and cell migration and invasion were measured by transwell assay and western blot. The effects of hypoxia treatment on glucose uptake, lactate production, extracellular acidification rate (ECAR), adenosine triphosphate (ATP), reactive oxygen species (ROS), Nicotinamide adenine dinucleotide phosphate (NADPH) and its oxidized form NADP + , and oxygen consumption rate (OCR) in ovarian cancer cells were examined. Then PPFIA4 was identified through bioinformatic analysis, and the regulatory effects of PPFIA4 on glucose metabolic reprogramming. Our data suggested that hypoxia enhanced the migration and invasion ability of ovarian cancer cells in vitro, and promoted the glucose metabolic reprogramming of ovarian cancer cells. Ovarian cancer cell viability, migration, and invasion were inhibited after PPFIA4 knockdown. Inhibition of PPFIA4 inhibited hypoxic-induced glucose metabolic reprogramming in ovarian cancer cells. In addition, PPFIA4 was found to bind to hypoxia-inducible factor 1alpha (HIF1A), and HIF1A prominently induced PPFIA4 expression. Collectively, HIF1A mediated upregulation of PPFIA4 and promoted reprogramming of glucose metabolism in ovarian cancer cells. Therefore, PPFIA4 may be a therapeutic target for ovarian cancer intervention.






Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Mennerich D, Kubaichuk K, Kietzmann T. DUBs, hypoxia, and cancer. Trends Cancer. 2019;5:632.
Rankin EB, Giaccia AJ. Hypoxic control of metastasis. Science. 2016;352:175.
**g X, Yang F, Shao C, Wei K, **e M, Shen H, Shu Y. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer. 2019;18:157.
Bao MH, Wong CC. Hypoxia, metabolic reprogramming, and drug resistance in liver cancer. Cells. 2021;10:1715.
Hu G, Wei Y, Kang Y. The multifaceted role of MTDH/AEG-1 in cancer progression. Clin Cancer Res. 2009;15:5615.
Masoud GN, Li W. HIF-1alpha pathway: role, regulation and intervention for cancer therapy. Acta Pharm Sin B. 2015;5:378.
Tang BL. Glucose, glycolysis, and neurodegenerative diseases. J Cell Physiol. 2020;235:7653.
Ganapathy-Kanniappan S, Geschwind JF. Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer. 2013;12:152.
Zheng X, Boyer L, ** M, Mertens J, Kim Y, Ma L, Ma L, Hamm M, Gage FH, Hunter T. Metabolic reprogramming during neuronal differentiation from aerobic glycolysis to neuronal oxidative phosphorylation. Elife. 2016;5:e13374.
Li CH, Liao CC. The metabolism reprogramming of microRNA Let-7-mediated glycolysis contributes to autophagy and tumor progression. Int J Mol Sci. 2021;23:113.
**ang S, Gu H, ** L, Thorne RF, Zhang XD, Wu M. LncRNA IDH1-AS1 links the functions of c-Myc and HIF1alpha via IDH1 to regulate the Warburg effect. Proc Natl Acad Sci USA. 2018;115:E1465.
Hayashi Y, Yokota A, Harada H, Huang G. Hypoxia/pseudohypoxia-mediated activation of hypoxia-inducible factor-1alpha in cancer. Cancer Sci. 2019;110:1510.
Yang J, Ren B, Yang G, Wang H, Chen G, You L, Zhang T, Zhao Y. The enhancement of glycolysis regulates pancreatic cancer metastasis. Cell Mol Life Sci. 2020;77:305.
Liang Y, Wang H, Chen B, Mao Q, **a W, Zhang T, Song X, Zhang Z, Xu L, Dong G, Jiang F. circDCUN1D4 suppresses tumor metastasis and glycolysis in lung adenocarcinoma by stabilizing TXNIP expression. Mol Ther-Nucl Acids. 2021;23:355.
Zhao H, Yan G, Zheng L, Zhou Y, Sheng H, Wu L, Zhang Q, Lei J, Zhang J, **n R, Jiang L, Zhang X, Chen Y, Wang J, Xu Y, Li D, Li Y. STIM1 is a metabolic checkpoint regulating the invasion and metastasis of hepatocellular carcinoma. Theranostics. 2020;10:6483.
Yamasaki A, Nakayama K, Imaizumi A, Kawamoto M, Fujimura A, Oyama Y, Nagai S, Yanai K, Onishi H. Liprin-alpha4 as a possible new therapeutic target for pancreatic cancer. Anticancer Res. 2017;37:6649.
Onishi H, Yamasaki A, Nakamura K, Ichimiya S, Yanai K, Umebayashi M, Nagai S, Morisaki T. Liprin-alpha4 as a new therapeutic target for SCLC as an upstream mediator of HIF1alpha. Anticancer Res. 2019;39:1179.
Mattauch S, Sachs M, Behrens J. Liprin-alpha4 is a new hypoxia-inducible target gene required for maintenance of cell-cell contacts. Exp Cell Res. 2010;316:2883.
Gottmann P, Ouni M, Zellner L, Jahnert M, Rittig K, Walther D, Schurmann A. Polymorphisms in miRNA binding sites involved in metabolic diseases in mice and humans. Sci Rep. 2020;10:7202.
Huang J, Yang M, Liu Z, Li X, Wang J, Fu N, Cao T, Yang X. PPFIA4 promotes colon cancer cell proliferation and migration by enhancing tumor glycolysis. Front Oncol. 2021;11:653200.
Blum A, Wang P, Zenklusen JC. SnapShot: TCGA-analyzed tumors. Cell. 2018;173:530.
Srivastava C, Irshad K, Gupta Y, Sarkar C, Suri A, Chattopadhyay P, Sinha S, Chosdol K. NFsmall ka, CyrillicB is a critical transcriptional regulator of atypical cadherin FAT1 in glioma. BMC Cancer. 2020;20:62.
Fornes O, Castro-Mondragon JA, Khan A, van der Lee R, Zhang X, Richmond PA, Modi BP, Correard S, Gheorghe M, Baranasic D, Santana-Garcia W, Tan G, Cheneby J, Ballester B, Parcy F, Sandelin A, Lenhard B, Wasserman WW, Mathelier A. JASPAR 2020: update of the open-access database of transcription factor binding profiles. Nucleic Acids Res. 2020;48:D87.
Zhao S, Guan B, Mi Y, Shi D, Wei P, Gu Y, Cai S, Xu Y, Li X, Yan D, Huang M, Li D. LncRNA MIR17HG promotes colorectal cancer liver metastasis by mediating a glycolysis-associated positive feedback circuit. Oncogene. 2021;40:4709.
Bi J, Bi F, Pan X, Yang Q. Establishment of a novel glycolysis-related prognostic gene signature for ovarian cancer and its relationships with immune infiltration of the tumor microenvironment. J Transl Med. 2021;19:382.
Shi J, Ye J, Fei H, Jiang SH, Wu ZY, Chen YP, Zhang LW, Yang XM. YWHAZ promotes ovarian cancer metastasis by modulating glycolysis. Oncol Rep. 2019;41:1101.
Dorayappan K, Wanner R, Wallbillich JJ, Saini U, Zingarelli R, Suarez AA, Cohn DE, Selvendiran K. Hypoxia-induced exosomes contribute to a more aggressive and chemoresistant ovarian cancer phenotype: a novel mechanism linking STAT3/Rab proteins. Oncogene. 2018;37:3806.
Packer M. Mechanisms leading to differential hypoxia-inducible factor signaling in the diabetic kidney: modulation by SGLT2 inhibitors and hypoxia mimetics. Am J Kidney Dis. 2021;77:280.
Wigerup C, Påhlman S, Bexell D. Therapeutic targeting of hypoxia and hypoxia-inducible factors in cancer. Pharmacol Therapeut. 2016;164:152.
Li T, Mao C, Wang X, Shi Y, Tao Y. Epigenetic crosstalk between hypoxia and tumor driven by HIF regulation. J Exp Clin Cancer Res. 2020;39:224.
Basavaraju AM, Shivanna N, Yadavalli C, Garlapati PK, Raghavan AK. Ameliorative effect of Ananas comosus on cobalt chloride-induced hypoxia in Caco2 cells via HIF-1alpha, GLUT 1, VEGF, ANG and FGF. Biol Trace Elem Res. 2021;199:1345.
Acker T, Plate KH. Hypoxia and hypoxia inducible factors (HIF) as important regulators of tumor physiology. Cancer Treat Res. 2004;117:219.
Airley RE, Loncaster J, Raleigh JA, Harris AL, Davidson SE, Hunter RD, West CML, Stratford IJ. GLUT-1 and CAIX as intrinsic markers of hypoxia in carcinoma of the cervix: relationship to pimonidazole binding. Int J Cancer. 2003;104:85.
Sun HL, Liu YN, Huang YT, Pan SL, Huang DY, Guh JH, Lee FY, Kuo SC, Teng CM. YC-1 inhibits HIF-1 expression in prostate cancer cells: contribution of Akt/NF-kappaB signaling to HIF-1alpha accumulation during hypoxia. Oncogene. 2007;26:3941.
Zeng L, Zhou HY, Tang NN, Zhang WF, He GJ, Hao B, Feng YD, Zhu H. Wortmannin influences hypoxia-inducible factor-1 alpha expression and glycolysis in esophageal carcinoma cells. World J Gastroenterol. 2016;22:4868.
Couto-Vieira J, Nicolau-Neto P, Costa EP, Figueira FF, Simão TDA, Okorokova-Façanha AL, Ribeiro Pinto LF, Façanha AR. Multi-cancer V-ATPase molecular signatures: a distinctive balance of subunit C isoforms in esophageal carcinoma. EBioMedicine. 2020;51:102581.
Cruciat CM, Ohkawara B, Acebron SP, Karaulanov E, Reinhard C, Ingelfinger D, Boutros M, Niehrs C. Requirement of prorenin receptor and vacuolar H+-ATPase-mediated acidification for Wnt signaling. Science. 2010;327:459.
Matassa DS, Amoroso MR, Maddalena F, Landriscina M, Esposito F. New insights into TRAP1 pathway. Am J Cancer Res. 2012;2:235.
Yoshida S, Tsutsumi S, Muhlebach G, Sourbier C, Lee MJ, Lee S, Vartholomaiou E, Tatokoro M, Beebe K, Miyajima N, Mohney RP, Chen Y, Hasumi H, Xu W, Fukushima H, Nakamura K, Koga F, Kihara K, Trepel J, Picard D, Neckers L. Molecular chaperone TRAP1 regulates a metabolic switch between mitochondrial respiration and aerobic glycolysis. Proc Natl Acad Sci USA. 2013;110:E1604.
Song HY, Dunbar JD, Zhang YX, Guo D, Donner DB. Identification of a protein with homology to hsp90 that binds the type 1 tumor necrosis factor receptor (∗). J Biol Chem. 1995;270:3574.
Jiao HL, Weng BS, Yan SS, Lin ZM, Wang SY, Chen XP, Liang GH, Li XQ, Zhao WY, Huang JY, Zhang D, Zhang LJ, Han FY, Li SN, Chen LJ, Zhu JH, He WF, Ding YQ, Ye YP. Upregulation of OSBPL3 by HIF1A promotes colorectal cancer progression through activation of RAS signaling pathway. Cell Death Dis. 2020;11:571.
Chen Y, Liu L, **a L, Wu N, Wang Y, Li H, Chen X, Zhang X, Liu Z, Zhu M, Liao Q, Wang J. TRPM7 silencing modulates glucose metabolic reprogramming to inhibit the growth of ovarian cancer by enhancing AMPK activation to promote HIF-1alpha degradation. J Exp Clin Cancer Res. 2022;41:44.
Li L, Yang L, Fan Z, Xue W, Shen Z, Yuan Y, Sun X, Wang D, Lian J, Wang L, Zhao J, Zhang Y. Hypoxia-induced GBE1 expression promotes tumor progression through metabolic reprogramming in lung adenocarcinoma. Signal Transduct Target Ther. 2020;5:54.
Zhao R, Feng T, Gao L, Sun F, Zhou Q, Wang X, Liu J, Zhang W, Wang M, **ong X, Jia W, Chen W, Wang L, Han B. PPFIA4 promotes castration-resistant prostate cancer by enhancing mitochondrial metabolism through MTHFD2. J EXP Clin Cancer Res. 2022;41:125.
Funding
This work was supported by the Haiyan Scientific Research Fund Youth Project of Harbin Medical University Cancer Hospital (Grant Number JJQN2019-03); Heilongjiang Natural Science Foundation (Grant Number YQ2019H025).
Author information
Authors and Affiliations
Contributions
All authors participated in the design, interpretation of the studies and analysis of the data and review of the manuscript. ST drafted the work and revised it critically for important intellectual content; HY and YX were responsible for the acquisition and analysis of data for the work; YZ was responsible for the interpretation of data for the work; GL made substantial contributions to the conception or design of the work.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tan, S., Yu, H., Xu, Y. et al. Hypoxia-induced PPFIA4 accelerates the progression of ovarian cancer through glucose metabolic reprogramming. Med Oncol 40, 272 (2023). https://doi.org/10.1007/s12032-023-02144-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02144-0