Abstract
The calcitonin gene-related peptide (CGRP) family of neuropeptides, consists of CGRP, adrenomedullin, amylin, and calcitonin. The receptors consist of either calcitonin receptor-like receptor (CLR) or calcitonin receptor (CTR) which for function needs an accessory protein, receptor activity-modifying proteins (RAMPs). CGRP has a pivotal role in primary headaches but the role of the other members of the CGRP family of peptides in headaches is not known. Here, we describe the expression of these molecules in the trigeminal ganglion (TG) to understand more on their possible role(s). Single or double immunohistochemistry were applied on frozen sections of rat TG using primary antibodies against CGRP, procalcitonin, calcitonin, adrenomedullin, amylin, RAMP1/2/3, CLR, and CTR. In addition, mRNA expression was measured by quantitative qPCR on TGs. CGRP and calcitonin showed rich expression in the cytoplasm of small to medium-sized neurons, and co-localized sometimes. Procalcitonin was observed in the glial cells. Immunoreactive fibers storing both CGRP and calcitonin were also observed. Adrenomedullin immunoreactivity was found in the satellite glial cells and in fibers, probably the myelinating Schwann cells. Amylin was found in the cytoplasm in many TG neurons. Levels of mRNA expression for adrenomedullin, amylin, CLR, RAMP1, RAMP2, RAMP3, and CTR were measured using qPCR. The experiments verified the expression of mRNA in the TG with the exception of CTR, which was above the limit of detection indicating little or no mRNA expression. In addition to the well-known CGRP receptor (CLR/RAMP1) and the receptor for calcitonin—CTR, we propose that other receptors exist in the rat TG: adrenomedullin receptor AM2 (CLR/RAMP3) in mainly the satellite glial cells, amylin receptors AMY1 (CTR/RAMP1) in mainly neurons, and AMY3 (CTR/RAMP3) in the satellite glial cells. It is important to compare peptides and receptors side-by-side in studies to help address questions of actions resulting from cross-reactivity between receptors. Several of the diverse biological actions of the CGRP family of peptides are clinically relevant. Our findings demonstrate the specific ligand and receptor sites in the rat trigeminal ganglion, highlighting recognition mechanisms to facilitate drug development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The trigeminal ganglion (TG) is primarily a sensory ganglion of the trigeminal nerve (the Vth cranial nerve) that occupies Meckel’s cave at the base of the brain and is surrounded by the dura mater. The neurons within the TG are firmly enveloped by the satellite glial cells (SGCs), demonstrating the close interaction between the neurons and glial cells coupled by gap junctions (Hanani 2005). TG is linked peripherally to the ophthalmic, maxillary and part of the mandibular nerves, and centrally to the trigeminal nucleus caudalis (TNC) and dorsal root C1-C3 (Edvinsson 2011).
Calcitonin gene-related peptide (CGRP) is richly located in numerous sites throughout the central and peripheral nervous systems (Russell et al. 2014; Warfvinge and Edvinsson 2019a). A sizeable population of CGRP neurons within the TG signifies a major role for CGRP in trigeminal transmission. About half of all neurons in the TG express CGRP which is visualized by immunohistochemical staining with CGRP antibodies and by in situ hybridization to localize mRNA for CGRP (Eftekhari et al. 2010; Miller et al. 2016). CGRP-positive neurons are predominantly of unmyelinated small-medium diameter which is indicative of cell bodies of C-type sensory pain fibers (Eftekhari et al. 2013). The human CALCA gene codes for both calcitonin and αCGRP via an alternative splicing mechanism in neural tissues (Amara et al. 1982). Both calcitonin and αCGRP are cleaved from larger pro-peptides known as procalcitonin (Pro-CT) and pro-CGRP (Buervenich et al. Full size image
Neuronal soma CT expression displayed a similar pattern as that seen for CGRP (and was co-localized in some cells); granular staining of mainly small to medium-sized neurons and pearl-like staining of C-type fibers. The number of CT-positive cells and pearl-like-positive fibers were, however, less than those seen in CGRP immunohistochemical stained TGs. Yet, the main difference between CGRP and CT immunoreactivity was the CT cytoplasmic granular staining of SGCs, and of Aδ-fibers, which did not occur at CGRP immunohistochemistry.
Procalcitonin (Pro-CT) expression in TG has previously published (Tajti et al. 2011). We here confirm these results. Pro-CT immunoreactivity was observed exclusively in the glial cells, the SGCs, and the myelinating Schwann cells (Fig. 2b). Pro-CT immunoreactivity was found in the soma of the Schwann cells. The myelinated layers of the Schwann cells, surrounding the thick Aδ-fibers, did not express Pro-CT.
AMY was exclusively expressed in the cytoplasm of small- to medium-sized neurons. In some of the cells, the expression was granular, but in others, a general cytoplasmic immunoreactivity was observed.
AM was expressed in the thin cytoplasm of the glial cells, both the SGCs and cells envelo** the neuronal processes, probably Schwann cells. In addition, immunoreactivity was found in blood vessel walls, indicating vascular endothelial staining (not shown).
Receptor Elements Expression (Fig. 3)
CLR and RAMP1 expression has earlier been examined in detail (Eftekhari et al. 2010; Lennerz et al. 2008; Miller et al. 2016). The present paper confirms these results. The receptor components were expressed in the cytoplasm of the neurons (mainly the larger ones) and the SGCs, and the thick fibers, typical for Aδ-fibers.
Receptor immunohistochemistry CLR and RAMP1 expression has earlier been examined in some detail (Eftekhari et al. 2010; Lennerz et al. 2008). The present paper confirm these results. The receptor components were expressed in neurons (mainly the larger ones, short arrows), in the SGCs (arrow heads) and the thick fibers, typical for Aδ-fibers (long arrows). RAMP2 and RAMP3 were expressed in the nuclei, both in those of neurons (arrows) and glia cell nuclei (arrow heads). In addition, RAMP3 was expressed in the glial cells (long arrow), however weaker than the nuclei staining. CTR was expressed in varying intensity in most neurons (short arrows) and SGCs (arrow heads). In addition, the nuclei of the glial cells surrounding the thick fibers showed immunoreactivity. The thick fibers also expressed CTR (long arrows), but weaker compared to CLR staining
RAMP2 and RAMP3 were expressed in the nuclei, both in those of neurons and of glia cells. In addition, RAMP3 was expressed in the cytoplasm of the glial cells (possibly both SGCs and Schwann cells envelo** the Aδ-fibers); however, the expression was considerably weaker than the nuclei staining.
CTR was expressed with varying intensity in the cytoplasm of the most neurons and SGCs. In addition, the nuclei of the glial cells (Schwann cells) surrounding the Aδ-fibers showed CTR immunoreactivity. In addition, the Aδ-fibers also expressed CTR, but in a weak manner.
Ligand/Ligand Expression (Fig. 4)
To provide morphological clues for functionality of the CGRP family of peptides in the TG and cross-reactivity, i.e., selectivity of the antibodies used, we performed a series of double immunohistochemistry experiments. In these experiments, we suggest that areas with no co-localization represent areas with no cross-reactivity.
Ligand/ligand immunohistochemistry AMY and CGRP double staining showed co-expression in some of the small- to medium-sized neurons (short arrows) and thin C-fibers (long arrows); however, some were seen to only express CGRP or AMY (arrow heads). The number of CGRP-positive cells appeared to be more abundant than those storing AMY. Double staining with CT and CGRP antibodies showed co-expression in the small- to medium-sized neurons (short arrows) and in fibers (long arrows). In addition, CT was expressed in the SGCs (arrow heads)
Double staining with CGRP and CT antibodies showed co-expression in the cytoplasm of small-sized neurons. We accordingly suggest that this could indicate cross-reactivity between CGRP and CT antibodies. In addition, CT alone was expressed in the SGCs and thus CT expression in SGCs represent no cross-reactivity.
CGRP and AMY double staining showed co-expression in the cytoplasm of some of the small- to medium-sized neurons and in thin C-fibers; however, some were seen to only express CGRP and not AMY. The number of CGRP-positive cells appeared to be more abundant than those storing AMY.
AM was double stained with CGRP or AMY, respectively (not shown). No co-localization was found in neither with AM plus CGRP, nor with AM plus AMY double immunohistochemistry. AM and CT double staining was not performed since both antibodies were made in mouse.
Receptor/Receptor Expression (Fig. 5)
RAMP1 and CLR are co-expressed mainly in the cytoplasm of larger neurons and in Aδ neuronal fibers (not shown).
Receptor/receptor immunohistochemistry RAMP1 is expressed in neurons, in the SGCs and the thick fibers. CTR is expressed with varying intensity in most neurons and SGCs. With RAMP1 and CTR double immunohistochemistry, it was shown that co-expression exists in neurons (short arrows), SGCs (arrow heads), and fibers (long arrows), indicative of AMY1 receptor expression. RAMP3 is expressed in the nuclei, both neuronal and glia cell nuclei, and in addition in some SGCs. CLR is expressed in neurons, in the SGCs, and the thick fibers. Double immunohistochemistry shows co-expression in the SGCs (arrow heads), which suggests a presence of AM2 receptor
RAMP1 was expressed in neurons, in the SGCs and the Aδ fibers. CTR alone was expressed with varying intensity in most neurons and SGCs. RAMP1 and CTR double immunohistochemistry showed co-expression in some neurons, SGCs and fibers, which indicates AMY1 receptor expression.
RAMP3 and CTR double immunohistochemistry revealed a possible AMY3 receptor in the cytoplasm of the SGCs.
RAMP3 is expressed in the nuclei, both neuronal and SGCs nuclei. CLR is expressed in the cytoplasm of neurons, in the SGCs and the Aδ fibers. Double immunohistochemistry showed co-expression in the SGCs, which suggests a presence of AM2 receptor.
Double immunohistochemistry using combinations between CLR, RAMP2, and CTR antibodies could not be performed since all three antibodies were made in rabbit.
Ligand/Receptor Expression (Fig. 6)
The ligands are expressed in different manners: CGRP and CT are expressed in neurons and in pearl-like fibers. In addition, SGCs are CT immunoreactive. AMY was exclusively expressed in the neurons. AM was expressed in the glial cells.
Ligand/receptor immunohistochemistry CT and RAMP1 double staining revealed co-localization in some neurons (long arrow). However, some neurons were only CT immunoreactive (thin arrow) and some only RAMP1 immunoreactive (arrow head). The same pattern of immunoreactivity was found using AMY and RAMP1 antibodies; some neurons showed co-localization (arrow), some AMY (arrow head), and some RAMP1 immunoreactivity (thin arrow). AM is expressed in the glial cells (arrow) and RAMP2 in the nuclei (arrow head); consequently, no co-localization was found using double immunohistochemistry
The receptor components show the same diversity in their pattern: CLR and RAMP1 immunoreactivity in the neurons, in the SGCs, and in the thick fibers; RAMP2/3 in the neuron; and SGC nuclei and CTR in most neurons and SGCs.
We have previously shown that no co-localization exists between CGRP and CLR/RAMP1 (Eftekhari et al. 2010). We confirm the results in the present paper (data not shown).
Double CT and CTR immunohistochemistry showed that all CT immunoreactive cells also were CTR positive (data not shown). No co-localization was found in the fibers (CT positive) or in the glial cell nuclei (CTR positive).
CT and RAMP1 double staining revealed co-localization in some neurons. However, some neurons were only CT immunoreactive and some only RAMP1 immunoreactive. The same pattern of immunoreactivity was found using AMY and RAMP1 antibodies; some neurons showed co-localization, some AMY, and some RAMP1 immunoreactivity.
AM is expressed in the glial cells and RAMP2 in the nuclei; consequently, no co-localization was found using double immunohistochemistry.
Distribution of the CGRP Family of Peptides and their Receptors (Fig. 7)
The figure describes, in a schematic drawing, the simplified distribution of the CGRP family of peptides and their receptors. The CGRP receptor (CLR/RAMP1) is mainly expressed in the large neurons and SGCs, the adrenomedullin2 receptor (CLR/RAMP3) in some SGCs, the amylin1 receptor (CTR/RAMP1) in large neurons and SGCs, and the amylin3 receptor (CTR/RAMP3) in some SGCs.
RAMP1 Immunohistochemistry of TG Neuron and Cerebellar Purkinje Cell (Fig. 8)
To give an example on how the cellular distribution of the different receptor components may vary, we compared the expression of RAMP1 in a TG neuron with that of a cerebellar Purkinje cell. The TG neuron expresses RAMP1 in the cytoplasm and the Purkinje cell on the cell surface (results previously published by (Edvinsson et al. 2011; Eftekhari et al. 2010)).
RAMP1 immunohistochemistry of TG neuron and cerebellar Purkinje cell. The left image shows a large neuron of rat TG and the right image a rat cerebellar Purkinje cell, using RAMP1 immunohistochemistry. The TG neuron displays RAMP1 staining in the cytoplasm and the Purkinje cell the cell surface. Scale bars 25 μm
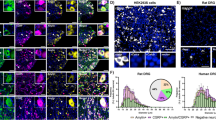
Quantitative PCR (Fig. 9)
Levels of mRNA expression for adrenomedullin, amylin, CLR, RAMP1, RAMP2, RAMP3, and CTR were measured using qPCR. The experiments confirmed the expression of all the genes except CTR (Ct values > 35, see dotted line) indicating little or no gene expression of CTR in TG.
qPCR experiments confirmed the expression of mRNA for adrenomedullin (Adm), amylin (IAPP), CLR (Calcrl), RAMP1 (Ramp1), RAMP2 (Ramp2), and RAMP3 (Ramp3) in rat TG. The experiment confirmed the expression of all the genes except CTR due to the expression above the limit of detection (Ct values > 35, see dotted line). Values are expressed as mean ± SEM and n = 6