Abstract
Background
Cerebral hypoxia is a frequent cause of secondary brain damage in patients with acute brain injury. Although hypercapnia can increase intracranial pressure, it may have beneficial effects on tissue oxygenation. We aimed to assess the effects of hypercapnia on brain tissue oxygenation (PbtO2).
Methods
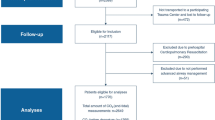
This single-center retrospective study (November 2014 to June 2022) included all patients admitted to the intensive care unit after acute brain injury who required multimodal monitoring, including PbtO2 monitoring, and who underwent induced moderate hypoventilation and hypercapnia according to the decision of the treating physician. Patients with imminent brain death were excluded. Responders to hypercapnia were defined as those with an increase of at least 20% in PbtO2 values when compared to their baseline levels.
Results
On a total of 163 eligible patients, we identified 23 (14%) patients who underwent moderate hypoventilation (arterial partial pressure of carbon dioxide [PaCO2] from 44 [42–45] to 50 [49–53] mm Hg; p < 0.001) during the study period at a median of 6 (4–10) days following intensive care unit admission; six patients had traumatic brain injury, and 17 had subarachnoid hemorrhage. A significant overall increase in median PbtO2 values from baseline (21 [19–26] to 24 [22–26] mm Hg; p = 0.02) was observed. Eight (35%) patients were considered as responders, with a median increase of 7 (from 4 to 11) mm Hg of PbtO2, whereas nonresponders showed no changes (from − 1 to 2 mm Hg of PbtO2). Because of the small sample size, no variable independently associated with PbtO2 response was identified. No correlation between changes in PaCO2 and in PbtO2 was observed.
Conclusions
In this study, a heterogeneous response of PbtO2 to induced hypercapnia was observed but without any deleterious elevations of intracranial pressure.



Similar content being viewed by others
Data Availability
Because of ethical restrictions, the data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request. All data generated after the analysis during this study are included in this published article and in its supplementary material.
References
Lantigua H, Ortega-Gutierrez S, Schmidt JM, Lee K, Badjatia N, Agarwal S, et al. Subarachnoid hemorrhage: who dies, and why? Crit Care. 2015;19:309. https://doi.org/10.1186/s13054-015-1036-0.
Popescu C, Anghelescu A, Daia C, Onose G. Actual data on epidemiological evolution and prevention endeavours regarding traumatic brain injury. J Med Life. 2015;8:272–7.
Hays LMC, Udy A, Adamides AA, Anstey JR, Bailey M, Bellapart J, et al. Effects of brain tissue oxygen (PbtO2) guided management on patient outcomes following severe traumatic brain injury: a systematic review and meta-analysis. J Clin Neurosci. 2022;99:349–58. https://doi.org/10.1016/j.jocn.2022.03.017.
Taccone FS, De Oliveira Manoel AL, Robba C, Vincent JL. Use a “GHOST-CAP” in acute brain injury. Crit Care. 2020;24:89. https://doi.org/10.1186/s13054-020-2825-7.
Wartenberg KE, Schmidt JM, Claassen J, Temes RE, Frontera JA, Ostapkovich N, et al. Impact of medical complications on outcome after subarachnoid hemorrhage. Crit Care Med. 2006;34:617–23. https://doi.org/10.1097/01.ccm.0000201903.46435.35.
Lazaridis C, Robertson CS. The role of multimodal invasive monitoring in acute traumatic brain injury. Neurosurg Clin N Am. 2016;27:509–17. https://doi.org/10.1016/j.nec.2016.05.010.
O’Leary RA, Nichol AD. Pathophysiology of severe traumatic brain injury. J Neurosurg Sci. 2018;62:542–8. https://doi.org/10.23736/S0390-5616.18.04501-0.
Lazaridis C, Rusin CG, Robertson CS. Secondary brain injury: predicting and preventing insults. Neuropharmacology. 2019;145:145–52. https://doi.org/10.1016/j.neuropharm.2018.06.005.
Vath A, Kunze E, Roosen K, Meixensberger J. Therapeutic aspects of brain tissue pO2 monitoring after subarachnoid hemorrhage. Acta Neurochir Suppl. 2002;81:307–9. https://doi.org/10.1007/978-3-7091-6738-0_78.
Chen HI, Stiefel MF, Oddo M, Milby AH, Maloney-Wilensky E, Frangos S, et al. Detection of cerebral compromise with multimodality monitoring in patients with subarachnoid hemorrhage. Neurosurgery. 2011;69:53–63. https://doi.org/10.1227/NEU.0b013e3182191451.
Kett-White R, Hutchinson PJ, Al-Rawi PG, Gupta AK, Pickard JD, Kirkpatrick PJ. Adverse cerebral events detected after subarachnoid hemorrhage using brain oxygen and microdialysis probes. Neurosurgery. 2002;50:1213–21. https://doi.org/10.1097/00006123-200206000-00008.
Maloney-Wilensky E, Gracias V, Itkin A, Hoffman K, Bloom S, Yang W, et al. Brain tissue oxygen and outcome after severe traumatic brain injury: a systematic review. Crit Care Med. 2009;37:2057–63. https://doi.org/10.1097/CCM.0b013e3181a009f8.
Oddo M, Levine JM, Mackenzie L, Frangos S, Feihl F, Kasner SE, et al. Brain hypoxia is associated with short-term outcome after severe traumatic brain injury independently of intracranial hypertension and low cerebral perfusion pressure. Neurosurgery. 2011;69:1037–45. https://doi.org/10.1227/NEU.0b013e3182287ca7.
van den Brink WA, van Santbrink H, Steyerberg EW, Avezaat CJ, Suazo JA, Hogesteeger C, et al. Brain oxygen tension in severe head injury. Neurosurgery. 2000;46:868–76. https://doi.org/10.1097/00006123-200004000-00018.
Haitsma IK, Maas AI. Advanced monitoring in the intensive care unit: brain tissue oxygen tension. Curr Opin Crit Care. 2002;8:115–20. https://doi.org/10.1097/00075198-200204000-00005.
Bohman LE, Heuer GG, Macyszyn L, Maloney-Wilensky E, Frangos S, Le Roux PD, et al. Medical management of compromised brain oxygen in patients with severe traumatic brain injury. Neurocrit Care. 2011;14:361–9. https://doi.org/10.1007/s12028-011-9526-7.
Gouvea Bogossian E, Diaferia D, Ndieugnou Djangang N, Menozzi M, Vincent JL, Talamonti M, et al. Brain tissue oxygenation guided therapy and outcome in non-traumatic subarachnoid hemorrhage. Sci Rep. 2021;11:16235. https://doi.org/10.1038/s41598-021-95602-6.
Kovacs M, Peluso L, Njimi H, De Witte O, Gouvêa Bogossian E, Quispe-Cornejo A, et al. Optimal cerebral perfusion pressure guided by brain oxygen pressure measurement. Front Neurol. 2021;12:732830. https://doi.org/10.3389/fneur.2021.732830.
Gouvea Bogossian E, Diosdado A, Barrit S, Al Barajraji M, Annoni F, Schuind S, et al. The impact of invasive brain oxygen pressure guided therapy on the outcome of patients with traumatic brain injury: a systematic review and meta-analysis. Neurocrit Care. 2022;37:779–89. https://doi.org/10.1007/s12028-022-01613-0.
Schmieder K, Jarus-Dziedzic K, Wronski J, Harders A. CO2 reactivity in patients after subarachnoid haemorrhage. Acta Neurochir (Wien). 1997;139:1038–41. https://doi.org/10.1007/BF01411557.
Brian JE Jr. Carbon dioxide and the cerebral circulation. Anesthesiology. 1998;88:1365–86. https://doi.org/10.1097/00000542-199805000-00029.
Okonkwo DO, Shutter LA, Moore C, Temkin NR, Puccio AM, Madden CJ, et al. Brain oxygen optimization in severe traumatic brain injury phase-II: a phase II randomized trial. Crit Care Med. 2017;45:1907–14. https://doi.org/10.1097/CCM.0000000000002619.
Chesnut R, Aguilera S, Buki A, Bulger E, Citerio G, Cooper DJ, et al. A management algorithm for adult patients with both brain oxygen and intracranial pressure monitoring: the Seattle International Severe Traumatic Brain Injury Consensus Conference (SIBICC). Intensive Care Med. 2020;46:919–29. https://doi.org/10.1007/s00134-019-05900-x.
Godoy DA, Rovegno M, Lazaridis C, Badenes R. The effects of arterial CO2 on the injured brain: two faces of the same coin. J Crit Care. 2021;61:207–15. https://doi.org/10.1016/j.jcrc.2020.10.028.
Stetter C, Weidner F, Lilla N, Weiland J, Kunze E, Ernestus RI, et al. Therapeutic hypercapnia for prevention of secondary ischemia after severe subarachnoid hemorrhage: physiological responses to continuous hypercapnia. Sci Rep. 2021;11:11715. https://doi.org/10.1038/s41598-021-91007-7.
Westermaier T, Stetter C, Kunze E, Willner N, Holzmeier J, Weiland J, et al. Controlled hypercapnia enhances cerebral blood flow and brain tissue oxygenation after aneurysmal subarachnoid hemorrhage: results of a phase 1 study. Neurocrit Care. 2016;25:205–14. https://doi.org/10.1007/s12028-016-0246-x.
Meyfroidt G, Bouzat P, Casaer MP, Chesnut R, Hamada SR, Helbok R, et al. Management of moderate to severe traumatic brain injury: an update for the intensivist. Intensive Care Med. 2022;48:649–66. https://doi.org/10.1007/s00134-022-06702-4. (Erratum in: Intensive Care Med. 2022;48(7):989–91).
Gouvea Bogossian E, Battaglini D, Fratino S, Minini A, Gianni G, Fiore M, et al. The role of brain tissue oxygenation monitoring in the management of subarachnoid hemorrhage: a sco** review. Neurocrit Care. 2023. https://doi.org/10.1007/s12028-023-01680-x.
Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13:818–29.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 2017;2:81–4.
Coppalini G, Duvigneaud E, Diosdado A, Migliorino E, Schuind S, Creteur J, et al. Effect of inotropic agents on oxygenation and cerebral perfusion in acute brain injury. Front Neurol. 2022;13:963562. https://doi.org/10.3389/fneur.2022.963562.
Gouvea-Bogossian E, Rass V, Lindner A, Iaquaniello C, Miroz JP, Cavalcante-Dos-Santos E, et al. Factors associated with brain tissue oxygenation changes after RBC transfusion in acute brain injury patients. Crit Care Med. 2022;50:e539–47. https://doi.org/10.1097/CCM.0000000000005460.
Firth D. Bias reduction of maximum likelihood estimates. Biometrika. 1993;80:27–38. https://doi.org/10.1093/biomet/80.1.27.
Nangunoori R, Maloney-Wilensky E, Stiefel M, Park S, Andrew Kofke W, Levine JM, et al. Brain tissue oxygen-based therapy and outcome after severe traumatic brain injury: a systematic literature review. Neurocrit Care. 2012;17:131–8. https://doi.org/10.1007/s12028-011-9621-9.
**e Q, Wu HB, Yan YF, Liu M, Wang ES. Mortality and outcome comparison between brain tissue oxygen combined with intracranial pressure/cerebral perfusion pressure-guided therapy and intracranial pressure/cerebral perfusion pressure-guided therapy in traumatic brain injury: a meta-analysis. World Neurosurg. 2017;100:118–27. https://doi.org/10.1016/j.wneu.2016.12.097.
Harper AM, Glass HI. Effect of alterations in the arterial carbon dioxide tension on the blood flow through the cerebral cortex at normal and low arterial blood pressures. J Neurol Neurosurg Psychiatry. 1965;28:449–52. https://doi.org/10.1136/jnnp.28.5.449.
Ito H, Kanno I, Ibaraki M, Suhara T, Miura S. Relationship between baseline cerebral blood flow and vascular responses to changes in PaCO2 measured by positron emission tomography in humans: implication of inter-individual variations of cerebral vascular tone. Acta Physiol (Oxf). 2008;193:325–30. https://doi.org/10.1111/j.1748-1716.2008.01847.x.
Ito H, Yokoyama I, Iida H, Kinoshita T, Hatazawa J, Shimosegawa E, et al. Regional differences in cerebral vascular response to PaCO2 changes in humans measured by positron emission tomography. J Cereb Blood Flow Metab. 2000;20:1264–70. https://doi.org/10.1097/00004647-200008000-00011.
Ito H, Ibaraki M, Kanno I, Fukuda H, Miura S. Changes in the arterial fraction of human cerebral blood volume during hypercapnia and hypocapnia measured by positron emission tomography. J Cereb Blood Flow Metab. 2005;25:852–7. https://doi.org/10.1038/sj.jcbfm.9600076.
Hawryluk GW, Phan N, Ferguson AR, Morabito D, Derugin N, Stewart CL, et al. Brain tissue oxygen tension and its response to physiological manipulations: influence of distance from injury site in a swine model of traumatic brain injury. J Neurosurg. 2016;125:1217–28. https://doi.org/10.3171/2015.7.JNS15809.
Zoerle T, Lombardo A, Colombo A, Longhi L, Zanier ER, Rampini P, et al. Intracranial pressure after subarachnoid hemorrhage. Crit Care Med. 2015;43:168–76. https://doi.org/10.1097/CCM.0000000000000670.
Godoy DA, Lubillo S, Rabinstein AA. Pathophysiology and management of intracranial hypertension and tissular brain hypoxia after severe traumatic brain injury: an integrative approach. Neurosurg Clin N Am. 2018;29:195–212. https://doi.org/10.1016/j.nec.2017.12.001.
Lumb AB, Slinger P. Hypoxic pulmonary vasoconstriction: physiology and anesthetic implications. Anesthesiology. 2015;122:932–46. https://doi.org/10.1097/ALN.0000000000000569.
Marhong J, Fan E. Carbon dioxide in the critically ill: too much or too little of a good thing? Respir Care. 2014;59:1597–605. https://doi.org/10.4187/respcare.03405.
Moriyama S, Ichinose M, Dobashi K, Matsutake R, Sakamoto M, Fujii N, et al. Hypercapnia elicits differential vascular and blood flow responses in the cerebral circulation and active skeletal muscles in exercising humans. Physiol Rep. 2022;10:e15274. https://doi.org/10.14814/phy2.15274.
Chapman CL, Schlader ZJ, Reed EL, Worley ML, Johnson BD. Renal and segmental artery hemodynamic response to acute, mild hypercapnia. Am J Physiol Regul Integr Comp Physiol. 2020;318:R822–7. https://doi.org/10.1152/ajpregu.00035.2020.
Zeiler FA, Ercole A, Czosnyka M, Smielewski P, Hawryluk G, Hutchinson PJA, et al. Continuous cerebrovascular reactivity monitoring in moderate/severe traumatic brain injury: a narrative review of advances in neurocritical care. Br J Anaesth. 2020;124:440–53. https://doi.org/10.1016/j.bja.2019.11.031.
Bøthun ML, Haaland ØA, Moen G, Logallo N, Svendsen F, Thomassen L, et al. Impaired cerebrovascular reactivity may predict delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. J Neurol Sci. 2019;407:116539. https://doi.org/10.1016/j.jns.2019.116539.
Phillips AA, Chan FH, Zheng MM, Krassioukov AV, Ainslie PN. Neurovascular coupling in humans: physiology, methodological advances and clinical implications. J Cereb Blood Flow Metab. 2016;36:647–64. https://doi.org/10.1177/0271678X15617954.
Lidington D, Kroetsch JT, Bolz SS. Cerebral artery myogenic reactivity: The next frontier in develo** effective interventions for subarachnoid hemorrhage. J Cereb Blood Flow Metab. 2018;38:17–37. https://doi.org/10.1177/0271678X17742548.
Lidington D, Wan H, Bolz SS. Cerebral autoregulation in subarachnoid hemorrhage. Front Neurol. 2021;12:688362. https://doi.org/10.3389/fneur.2021.688362.
Claassen J, Thijssen DHJ, Panerai RB, Faraci FM. Regulation of cerebral blood flow in humans: physiology and clinical implications of autoregulation. Physiol Rev. 2021;101:1487–559. https://doi.org/10.1152/physrev.00022.2020.
Hosmann A, Schnackenburg P, Rauscher S, Hopf A, Bohl I, Engel A, et al. Brain tissue oxygen response as indicator for cerebral lactate levels in aneurysmal subarachnoid hemorrhage patients. J Neurosurg Anesthesiol. 2022;34:193–200. https://doi.org/10.1097/ANA.0000000000000713.
Funding
None.
Author information
Authors and Affiliations
Contributions
MA, EGB, and FST contributed to conception and design of the study. MA, MS, LP, and FA collected data. EGB and MA performed data curation. EGB, MA, and FST performed the statistical analysis. MA, EGB, and FST wrote the first draft of the manuscript. KD and SS revised the manuscript for intellectual content and English editing. All authors contributed to manuscript revision and read and approved the submitted version.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests regarding this article.
Ethical Approval/Informed Consent
The study followed ethical guidelines, and the Erasme Hospital Ethics Committee approved this study (P2022/449). Written informed consent for study participation was waived by the ethics committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anderloni, M., Schuind, S., Salvagno, M. et al. Brain Oxygenation Response to Hypercapnia in Patients with Acute Brain Injury. Neurocrit Care 40, 750–758 (2024). https://doi.org/10.1007/s12028-023-01833-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-023-01833-y