Abstract
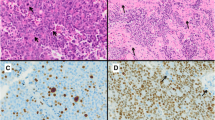
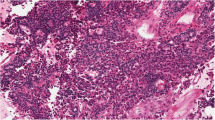
WHO classifications should be used for comparing the results from different groups of pathologist and clinicians by standardized histopathological methods. Our present report describes the important parameters of pituitary adenoma pathology as demand of the WHO classification for correlation to endocrine data and prognosis. The combination of HE stain based structures with immunostainings for pituitary hormones allows subclassification of adenomas as the best method not only for correlations to clinical hyperfunctions but also for statements to the sensitivity of drug therapies (somatostatin analogs, dopamine agonists). GH-, PRL- and ACTH-secreting pituitary adenomas are further classified based on the size and number of their secretory granules by electron microscopy, or as is mostly the case nowadays by cytokeratin staining pattern, into densely and sparsely granulated. Granulation pattern may be considered for the prediction of treatment response in patients with GH-secreting adenomas, since the sparsely granulated subtype was shown to be less responsive to somatostatin analog treatment. For prognosis, it is important to identify aggressive adenomas by measurements of the Ki-67 index, of the number of mitoses, and of nuclear expression of p53. Among the criteria for atypical adenomas, high Ki-67 labeling index and invasive character are the most important adverse prognostic factors. Promising molecular markers have been identified that might supplement the currently used proliferation parameters. For defining atypical adenomas in a future histopathological classification system, we propose to provide the proliferative potential and the invasive character separately.
Similar content being viewed by others
References
Lloyd RV, Kovacs K, Young WF, Jr., Farrell WE, Asa SL, Trouillas J, et al. Tumours of the pituitary. In: DeLellis RA, Lloyd RV, Heitz PU, editors. Pathology and Genetics. Tumours of Endocrine Tumours. 1 ed. Lyon: International Agency for Research and Cancer (IARC); 2004. p. 9–48.
Mete O, Asa SL. Therapeutic implications of accurate classification of pituitary adenomas. Semin Diagn Pathol 2013;30(3):158–64.
Mete O, Asa SL. Clinicopathological Correlations in Pituitary Adenomas. Brain Pathology 2012;22(4):443–53.
Scheithauer BW, Gaffey TA, Lloyd RV, Kovacs KT, Horvath E, Yapicier O, et al. Pathobiology of pituitary adenomas and carcinomas. Neurosurgery 2006;59(2):341–53.
Saitoh Y, Mori H, Matsumoto K, Ushio Y, Hayakawa T, Mori S, et al. Accumulation of amyloid in pituitary adenomas. Acta Neuropath (Berlin) 1985;68(2):87–92.
Röcken C, Uhlig H, Saeger W, Linke RP, Fehr S. Amyloid deposits in pituitaries and pituitary adenomas: Immunohistochemistry and in situ hybridization. Endocr Pathol 1995;6:135–43.
Lipper S, Isenberg HD, Kahn LB. Calcospherites in pituitary prolactinomas. A hypothesis for their formation. Arch Pathol Lab Med 1984 Jan;108(1):31–4.
Scheithauer BW, Horvath E, Lloyd RV, Kovacs K. Pathology of pituitary adenomas and pituitary hyperplasia. In: Thapar K, Kovacs K, Scheithauer BW, Lloyd RV, editors. Diagnosis and management pituitary tumors. 1 ed. Totowa,NJ: Humana Press; 2001. p. 91–154.
Kontogeorgos G. Predictive markers of pituitary adenoma behavior. Neuroendocrinology 2006;83(3–4):179–88.
Righi A, Agati P, Sisto A, Frank G, Faustini-Fustini M, Agati R, et al. A classification tree approach for pituitary adenomas. Hum Pathol 2012;43(10):1627–37.
Kovacs K, Stefaneanu L, Horvath E, Lloyd RV, Lancranjan I, Buchfelder M, et al. Effect of dopamine agonist medication on prolactin producing pituitary adenomas. A morphological study including immunocytochemistry, electron microscopy and in situ hybridization. Virchows Arch A Pathol Anat Histopath 1991;418(5):439–46.
Saeger W. Effect of drugs on pituitary ultrastructure. Microsc Res Techn 1992 Jan 15;20(2):162–76.
Sautner D, Saeger W, Tallen G, Lüdecke DK. Effects of octreotide on morphology of pituitary adenomas in acromegaly. Pathol Res Pract 1993;189:1044–51.
Asa SL. Pituitary adenomas. In: Silverberg SG, Gardner WA, Sobin LH, editors. Tumors of the pituitary gland. 1 ed. Washington: Armed Forces Institute of Pathology; 2011. p. 55–172.
Al Brahim NYY, Asa SL. My approach to pathology of the pituitary gland. Journal of Clinical Pathology 2006;59(12):1245–53.
Asa SL. Practical pituitary pathology - What does the pathologist need to know? Arch Pathol Lab Med 2008;132(8):1231–40.
Ezzat S, Asa SL, Couldwell WT, Barr CE, Dodge WE, Vance ML, et al. The prevalence of pituitary adenomas - A systematic review. Cancer 2004;101(3):613–9.
Jahangiri A. A comprehensive long-term retrospective analysis of silent corticotrophic adenomas vs hormone-negative adenomas. Neurosurgery 73, 8–17 Discussion 17–18. 2013.
Asa SL, Ezzat S. The pathogenesis of pituitary tumors. Annu Rev Pathol 4, 97–126. 2015.
Sivapragasam M, Rotondo F, Lloyd RV, Scheithauer BW, Cusimano M, Syro LV, et al. MicroRNAs in the Human Pituitary. Endocr Pathol 2011;22(3):134–43.
Wierinckx A. Integrated genomic profiling identifies loss of chromosome 11p impacting transcriptomic activity in aggressive pituitary PRL tumors. Brain Pathol 21, 533–543. 2015.
Cornelius A, Cortet-Rudelli C, Assaker R, Kerdraon O, Gevaert MH, Prevot V, et al. Endothelial Expression of Endocan Is Strongly Associated with Tumor Progression in Pituitary Adenoma. Brain Pathology 2012;22(6):757–64.
Al Shraim M, Asa SL. The 2004 World Health Organization classification of pituitary tumors: What is new? Acta Neuropathologica 2006;111(1):1–7.
Kovacs K, Horvath E. Tumors of the pituitary gland. Washington,D.C.: Armed Forces Institute of Pathology; 1986.
Horvath E, Kovacs K, Smyth HS, Cusimano M, Singer W. Silent adenoma subtype 3 of the pituitary - Immunohistochemical and ultrastructural classification: A review of 29 cases. Ultrastruct Pathol 2005;29(6):511–24.
Horvath E, Kovacs K. Ultrastructural classification of pituitary adenomas. J Canad Sci Neurol 1976;3:9–21.
Sano T, Ohshima T, Yamada S. Expression of glycoprotein hormones and intracytoplasmic distribution of cytokeratin in growth hormone-producing pituitary adenomas. Pathol Res Pract 1991 Jun;187(5):530–3.
Obari A, Sano T, Ohyama K, Kudo E, Qian ZR, Yoneda A, et al. Clinicopathological features of growth hormone-producing pituitary adenomas: Difference among various types defined by cytokeratin distribution pattern including a transitional form. Endocr Pathol 2008;19(2):82–91.
Nishioka H, Haraoka J, Akada K. Fibrous bodies are associated with lower GH production and decreased expression of E-cadherin in GH-producing pituitary adenomas. Clin Endorin 2003;59(6):768–72.
Bakhtiar Y, Hirano H, Arita K, Yunoue S, Fujio S, Tominaga A, et al. Relationship between cytokeratin staining patterns and clinico-pathological features in somatotropinomae. Eur J Endocrin 2010;163(4):531–9.
Spada A, Vallar L, Faglia G. G-Protein oncogenes in pituitary tumors. Trends Endocrinol Metab 1992;3:355–60.
Fougner SL, Casar-Borota O, Heck A, Berg JP, Bollerslev J. Adenoma granulation pattern correlates with clinical variables and effect of somatostatin analogue treatment in a large series of patients with acromegaly. Clin Endorin 2012;76(1):96–102.
Larkin S, Reddy R, Karavitaki N, Cudlip S, Wass J, Ansorge O. Granulation pattern but nit GSP or GHR mutation is associated with clinical characteristics in somatostatin-naive patients with somatotroph adenomas. Eur J Endocrinol 2013;168(4):491–9.
Yamada S, Aiba T, Sano T, Kovacs K, Shishiba Y, Sawano S, et al. Growth hormone-producing pituitary adenomas: correlations between clinical characteristics and morphology. Neurosurgery 1993 Jul;33(1):20–7.
Mazal PR, Czech T, Sedivy R, Aichholzer M, Wanschitz J, Klupp N, et al. Prognostic relevance of intracytoplasmic cytokeratin pattern, hormone expression profile, and cell proliferation in pituitary adenomas of akromegalic patients. Clin Neuropathol 2001;20(4):163–71.
Ito M, Yoshida K, Kyo E, Ayhan A, Nakayama H, Yasui W, et al. Expression of several growth factors and their receptor genes in human colon carcinomas. Virchows Arch B Cell Pathol 1990;59(3):173–8.
Ezzat S, Kontogeorgos G, Redelmeier DA, Horvath E, Harris AG, Kovacs K. In vivo responsiveness of morphological variants of growth hormone-producing pituitary adenomas to octreotide. Eur J Endocrin 1995;133(6):686–90.
Bhayana S, Booth GL, Asa SL, Kovacs K, Ezzat S. The implication of somatotroph adenoma phenotype to somatostatin analog responsiveness in acromegaly. J Clin Endocrinol Metab 2005;90(11):6290–5.
Brzana J, Yedinak CG, Gultekin SH, Delashaw JB, Fleseriu M. Growth hormone granulation pattern and somatostatin receptor subtype 2A correlate with postoperative somatostatin receptor ligand response in acromegaly: a large single center experience. Pituitary 2013;16(4):490–8.
Mayr B, Buslei R, Theodoropoulou M, Stalla GK, Buchfelder M, Schofl C. Molecular and functional properties of densely and sparsely granulated GH-producing pituitary adenomas. Eur J Endocrin 2013;169(4):391–400.
Velkeniers B, Hooghe-Peters EL. From prolactin cell to prolactinoma: implications of ontogenic mechanisms in diagnosis and management. Endocr Relat Cancer 1998;5:27–36.
Saeger W, Lüdecke DK, Buchfelder M, Fahlbusch R, Quabbe HJ, Petersenn S. Pathohistological classification of pituitary tumors: 10 years of experience with the German Pituitary Tumor Registry. Eur J Endocrinol 2007;156:205–17.
Asa SL, Ezzat S. Medical management of pituitary adenomas: structural and ultrastructural changes. Pituitary 2002;5(2):133–9.
Asa SL, Ezzat S. The pathogenesis of pituitary tumors. Annu Rev Pathol 2009;4:97–126.
Reed CT, Adams K, Shenoy V. Xanthogranulomatous Adrenalitis: A Case Report of a Diabetic, 55-Year-Old Male. Endocr Pathol 2015;26(3):229–31.
Di Ieva A, Davidson JM, Syro LV, Rotondo F, Montoya JF, Horvath E, et al. Crooke’s Cell Tumors of the Pituitary. Neurosurgery 2015;76(5):616–22.
Syro LV, Rotondo F, Cusimano MD, Di Ieva A, Horvath E, Restrepo LM, et al. Current status on histological classification in Cushing’s disease. Pituitary 2015;18(2):217–24.
Horvath E, Kovacs K, Killinger DW, Smyth HS, Platts ME, Singer W. Silent corticotropic adenomas of the human pituitary gland. A histologic, immunocytologic, and ultrastructural study. Amer J Pathol 1980;98:617–38.
Reincke M, Allolio B, Saeger W, Kaulen D, Winkelmann W. A pituitary adenoma secreting high molecular weight adrenocorticotropin without evidence of Cushing’s disease. J Clin Endocrinol Metab 1987;65:1296–300.
Osamura RY, Kajiya H, Takei M, Egashira N, Tobita M, Takekoshi S, et al. Pathology of the human pituitary adenomas. Histochemistry and Cell Biology 2008;130(3):495–507.
Saeger W, Lüdecke DK. Pituitary adenomas with hyperfunction of TSH. Frequency, histological classification, immunocytochemistry and ultrastructure. Virchows Archiv 1982;394:255–67.
Horvath E, Kovacs K. Gonadotroph adenomas of the human pituitary: sex-related fine-structural dichotomy. A histologic, immunocytochemical, and electron-microscopic study of 30 tumors. Am J Pathol 1984 Dec;117(3):429–40.
Grossman AB. The 2004 World health organization classification of pituitary tumors: is it clinically helpful? Acta Neuropathologica 2006;111(1):76–7.
Landolt AM, Heitz PU. Alpha-subunit-producing pituitary adenomas. Immunocytochemical and ultrastructural studies. Virchows Arch A Pathol Anat Histopath 1986;409(4):417–31.
Saeger W. Elektronenoptische Untersuchungen zur Problematik der onkocytären Hypophysenadenome. Verhandlungen der Deutschen Gesellschaft für Pathologie 58, 544. 1974.
Niveiro M, Aranda FI, Paya A, Boix E, Peiro G, Pico A. Oncocytic transformation in pituitary adenomas - Immunohistochemical analyses of 65 cases. Arch Pathol Lab Med 2004;128(7):776–80.
Asa SL. Tumors of the pituitary gland. Washington,D.C.: Armed Forces Institute of Pathology; 1998.
Crooke AC. A change in the basophil cells of the pituitary gland common to conditions which wxhibit the syndrome attributed to basophil adenoma. J Pathol Bacteriol 1935;41:339–49.
George DH, Scheithauer BW, Kovacs K, Horvath E, Young WF, Lloyd RV, et al. Crooke’s cell adenoma of the pituitary - An aggressive variant of corticotroph adenoma. Amer J Surg Pathol 2003;27(10):1330–6.
Lim JS, Ku CR, Lee MK, Kim TS, Kim SH, Lee EJ. A case of fugitive acromegaly, initially presented as invasive prolactinoma. Endocrine 2010;38(1):1–5.
Zada G, Woodmansee WW, Ramkissoon S, Amadio J, Nose V, Laws ER. Atypical pituitary adenomas: incidence, clinical characteristics, and implications Clinical article. J Neurosurg 2011;114(2):336–44.
Gejman R, Swearingen B, Hedley-Whyte ET. Role of Ki-67 proliferation index and p53 expression in predicting progression of pituitary adenomas. Hum Pathol 2008;39(5):758–66.
Mizoue T, Kawamoto H, Arita K, Kurisu K, Tominaga A, Uozumi T. MIB-1 immunopositivity is associated with rapid regrowth of pituitary adenomas. acta neurochirurgica (Wien,Oester.) 139, 426–432. 1997.
Widhalm G, Wolfsberger S, Preusser M, Fischer I, Woehrer A, Wunderer J, et al. Residual nonfunctioning pituitary adenomas: prognostic value of MIB-1 labeling index for tumor progression Clinical article. J Neurosurg 2009;111(3):563–71.
Chiloiro S, Bianchi A, Doglietto F, de Waure C, Giampietro A, Fusco A, et al. Radically resected pituitary adenomas: prognostic role of Ki 67 labeling index in a monocentric retrospective series and literature review. Pituitary 2014;17(3):267–76.
Lee EH, Kim KH, Kwon JH, Kim HD, Kim YZ. Results of Immunohistochemical Staining of Cell-Cycle Regulators: The Prediction of Recurrence of Functioning Pituitary Adenoma. World Neurosurgery 2014;81(3–4):563–75.
Trouillas J, Roy P, Sturm N, Dantony E, Cortet-Rudelli C, Viennet G, et al. A new prognostic clinicopathological classification of pituitary adenomas: a multicentric case–control study of 410 patients with 8 years post-operative follow-up. Acta Neuropathologica 2013;126(1):123–35.
Aguilar PH, Aires R, Laws ER, Isolan GR, Logullo A, Patil C, et al. Labeling index in pituitary adenomas evaluated by means of MIB-1: is there a prognostic role? A critical review. Neurological Research 32, 1060–1071. 2010.
Ekramullah SM, Saitoh Y, Arita N, Ohnishi T, Hayakawa T. The correlation of Ki-67 staining indices with tumor doubling times in regrowing non-functioning pituitary adenomas. acta neurochir (Wien) 1996;138:1449–55.
Honegger J, Prettin C, Feuerhake F, Petrick M, Schulte-Monting J, Reincke M. Expression of Ki-67 antigen in nonfunctioning pituitary adenomas: correlation with growth velocity and invasiveness. J Neurosurg 2003;99(4):674–9.
Tanaka Y, Hongo K, Tada T, Sakai K, Kakizawa Y, Kobayashi S. Growth pattern and rate in residual nonfunctioning pituitary adenomas: correlations among tumor volume doubling time, patient age, and MIB-1 index. J Neurosurg 2003;98(2):359–65.
Hsu CY, Guo TY, Chien CP, Ho DM. MIB-1 labeling index correlated with magnetic resonance imaging detected tumor volume doubling time in pituitary adenoma. Europ J Endocrinol 162, 1027–1033. 2010.
Salehi F, Agur A, Scheithauer BW, Kovacs K, Lloyd RV, Cusimano M. Ki-67 in Pituitary Neoplasms: A Review-Part I. Neurosurgery 2009;65(3):429–37.
Mete O, Ezzat S, Asa SL. Biomarkers of aggressive pituitary adenomas. Journal of Molecular Endocrinology 2012;49(2):R69-R78.
Thapar K, Scheithauer BW, Kovacs K, Pernicone PJ, Laws ER, Jr. p53 expression in pituitary adenomas and carcinomas: Correlation with invasiveness and tumor growth fractions. Neurosurgery 1996;38(4):765–70.
Meij BP, Lopes MBS, Ellegala DB, Alden TD, Laws ER. The long-term significance of microscopic dural invasion in 354 patients with pituitary adenomas treated with transsphenoidal surgery. J Neurosurg 2002 Feb;96(2):195–208.
Mastronardi L, Guiducci A, Spera C, Puzzilli F, Liberati F, Ruggeri A, et al. Adrenocorticotropic hormone secreting pituitary adenomas: Analysis of growth fraction using the MIB-1 antibody. Tumori 2000 May;86(3):229–32.
Chacko G, Chacko AG, Kovacs K, Scheithauer BW, Mani S, Muliyil JP, et al. The clinical significance of MIB-1 labeling index in pituitary adenomas. Pituitary 2010;13(4):337–44.
Knosp E, Steiner E, Kitz K, Matula C. Pituitary Adenomas with Invasion of the Cavernous Sinus Space - A Magnetic Resonance Imaging Classification Compared with Surgical Findings. Neurosurgery 1993;33:610–8.
Thapar K, Kovacs K, Scheithauer BW, Stefaneanu L, Horvath E, Pernicone PJ, et al. Proliferative activity and invasiveness among pituitary adenomas and carcinomas: An analysis using the MIB-1 antibody. Neurosurgery 1996;38(1):99–106.
Sarkar S, Chacko AG, Chacko G. An analysis of granulation patterns, MIB-1 proliferation indices and p53 expression in 101 patients with acromegaly. Acta Neurochirurgica 2014;156(12):2221–30.
Mastronardi L, Guiducci A, Puzzilli F. Lack of correlation between Ki-67 labellin index and tumor size of anterior pituitary adenomas. Bmc Cancer 1, 12–16. 2001.
Madsen C, Schroder HD. Ki-67 immunoreactivity in meningiomas - determination of the proliferation potential of meningiomas using the monclonal antibody Ki-67. Clinical Neuropathology 16, 137–142. 1997.
Raverot G, Castinetti F, Jouanneau E, Morange I, Figarella-Branger D, Dufour H, et al. Pituitary carcinomas and aggressive pituitary tumours: merits and pitfalls of temozolomide treatment. Clin Endorin 2012;76(6):769–75.
Di Ieva A, Rotondo F, Syro LV, Cusimano MD, Kovacs K. Aggressive pituitary adenomas-diagnosis and emerging treatments. Nature Reviews Endocrinology 2014;10(7):423–35.
Miermeister CP, Petersenn S, Buchfelder M, Fahlbusch R, Lüdecke DK, Hölsken A, et al. Histological criteria for atypical adenomas: data from German Pituitary Tumor Registry suggest modifications. acta neuropath communications . 2015.
Acknowledgments
The funding for the German Pituitary Tumor Registry to WS from Novartis Pharma GmbH (Nuremberg, Germany), Novo Nordisk Pharma GmbH (Mainz, Germany), Pfizer Pharma GmbH (Karlsruhe, Germany), and Ipsen Pharma GmbH (Ettlingen, Germany) is gratefully acknowledged. We thank all colleagues for sending tumor material to the German Pituitary Tumor Registry.
Authors Contributions
W. Saeger: General pathology, electron microscopy, special adenoma subtypes.
J. Honegger: Atypical adenomas.
M. Theodoropoulou: Granulation.
R. Buslei: Immunocytochemistry.
U.J. Knappe: Invasion.
C. Schöfl: Correlation of clinic and morphology.
St. Petersen: Correlation of clinic and morphology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Due to the character and kind (review) of the theme of the manuscript, an approval on ethics appears to be not necessary.
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Saeger, W., Honegger, J., Theodoropoulou, M. et al. Clinical Impact of the Current WHO Classification of Pituitary Adenomas. Endocr Pathol 27, 104–114 (2016). https://doi.org/10.1007/s12022-016-9418-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12022-016-9418-7