Abstract
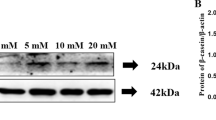
This study aimed to investigate the effect of boron on porcine mammary epithelial cells (PMECs) survival, cell cycle, and milk fat synthesis. PMECs from boron-treated groups were exposed to 0–80 mmol/L boric acid concentrations. Cell counting kit-8 and flow cytometry assays were performed to assess cell survival and the cell cycle, respectively. Triacylglycerol (TAG) levels in PMECs and culture medium were determined by a triacylglycerol kit while PMECs lipid droplet aggregation was investigated via oil red staining. Milk fat synthesis–associated mRNA levels were determined by quantitative real-time polymerase chain reaction (qPCR) while its protein expressions were determined by Western blot. Low (0.2, 0.3, 0.4 mmol/L) and high (> 10 mmol/L) boron concentrations significantly promoted and inhibited cell viabilities, respectively. Boron (0.3 mmol/L) markedly elevated the abundance of G2/M phase cells. Ten mmol/L boron significantly increased the abundances of G0/G1 and S phase cells, but markedly suppressed G2/M phase cell abundance. At 0.3 mmol/L, boron significantly enhanced ERK phosphorylation while at 0.4, 0.8, 1, and 10 mmol/L, it markedly decreased lipid droplet diameters. Boron (10 mmol/L) significantly suppressed ACACA and SREBP1 protein expressions. The FASN protein levels were markedly suppressed by 0.4, 0.8, 1, and 10 mmol/L boron. Both 1 and 10 mmol/L markedly decreased FASN and SREBP1 mRNA expressions. Ten mmol/L boron significantly decreased PPARα mRNA levels. Low concentrations of boron promoted cell viability, while high concentrations inhibited PMECS viabilities and reduced lipid droplet diameters, which shows the implications of boron in pregnancy and lactation.






Similar content being viewed by others
Data Availability
The raw data supporting this study will be available by authors to any qualified researcher upon request.
References
Schwertfeger KL, McManaman JL, Palmer CA, Neville MC, Anderson SM (2003) Expression of constitutively activated Akt in the mammary gland leads to excess lipid synthesis during pregnancy and lactation. J Lipid Res 44(6):1100–12. https://doi.org/10.1194/jlr.M300045-JLR200
Russell TD, Palmer CA, Orlicky DJ, Fischer A, Rudolph MC, Neville MC, McManaman JL (2007) Cytoplasmic lipid droplet accumulation in develo** mammary epithelial cells: roles of adipophilin and lipid metabolism. J Lipid Res 48(7):1463–75. https://doi.org/10.1194/jlr.M600474-JLR200
Lv Y, Guan W, Qiao H, Wang C, Chen F, Zhang Y, Liao Z (2015) Veterinary medicine and omics (veterinomics): metabolic transition of milk triacylglycerol synthesis in sows from late pregnancy to lactation. Omics : a J Integrative Biol 19(10):602–16. https://doi.org/10.1089/omi.2015.0102
Craig A, Henry W, Magowan E (2016) Effect of phase feeding and valine-to-lysine ratio during lactation on sow and piglet performance. J Anim Sci 94(9):3835–3843. https://doi.org/10.2527/jas.2016-0648
Anthony VC, Steven EE (2013) Comparative aspects of mammary gland development and homeostasis. Annu Rev Anim Biosci 1:179–202. https://doi.org/10.1146/annurev-animal-031412-103632
Macias H, Hinck L (2012) Mammary gland development. Wiley Interdiscip Rev Dev Biol 1(4):533–57. https://doi.org/10.1002/wdev.35
Anthony VC, Ratan KC (2020) Symposium review: determinants of milk production: understanding population dynamics in the bovine mammary epithelium. J Dairy Sci 103(3):2928–2940. https://doi.org/10.3168/jds.2019-17241
Che L, Xu M, Gao K, Zhu C, Wang L, Yang X, Wen X, **ao H, Jiang Z, Wu D (2019) Valine increases milk fat synthesis in mammary gland of gilts through stimulating AKT/MTOR/SREBP1 pathwaydagger. Biol Reprod 101(1):126–137. https://doi.org/10.1093/biolre/ioz065
Bionaz M, Loor J (2008) Gene networks driving bovine milk fat synthesis during the lactation cycle. Bmc Genomics 9(1):1–21. https://doi.org/10.1186/1471-2164-9-366
Rudolph MC, Monks J, Burns V, Phistry M, Marians R, Foote MR, Bauman DE, Anderson SM, Neville MC (2010) Sterol regulatory element binding protein and dietary lipid regulation of fatty acid synthesis in the mammary epithelium. Am J Physiol-Endocrino Metabol 299(6):E918-27. https://doi.org/10.1152/ajpendo.00376.2010
Devirian TA, Volpe SL (2003) The physiological effects of dietary boron. Crit Rev Food Sci 43(2):219–31. https://doi.org/10.1080/10408690390826491
Forrest HN (2009) Boron deprivation decreases liver S -adenosylmethionine and spermidine and increases plasma homocysteine and cysteine in rats. J Trace Elem Med Bio 23(3):204–13. https://doi.org/10.1016/j.jtemb.2009.03.001
Ruya K, Sahin Y, Gulsah B, Burcin AT, Pakize NT, Serap A, Feyza YO, Yuksel A, Aysen Y, Fikrettin S (2019) Boron-rich diet may regulate blood lipid profile and prevent obesity: a non-drug and self-controlled clinical trial. J Trace Elem Med Bio 54:191–198. https://doi.org/10.1016/j.jtemb.2019.04.021
Zheng Y, He X (2010) Characteristics and EGFP expression of porcine mammary gland epithelial cells. Res Vet Sci 89(3):383–390. https://doi.org/10.1016/j.rvsc.2010.03.023
Zhang X, **ao K, Qiu W, Wang J, Li P, Peng K (2020) The immune regulatory rffect of boron on ostrich chick splenic lymphocytes. Biol Trace Elem Res 199(7):2695–2706. https://doi.org/10.1007/s12011-020-02392-5
Lu L, Zhang Q, **E, Hu Q, Zhao C, Li S, Ren M (2020) Effects of boron on cytotoxicity, apoptosis, and cell cycle of cultured rat sertoli cells in Vitro. Biol Trace Elem Res 196(1):223–230. https://doi.org/10.1007/s12011-019-01911-3
Zhao C, Han Y, Wang C, Ren M, Hu Q, Gu Y, Ye P, Li S, ** E (2021) Transcriptome profiling of duodenum reveals the importance of boron supplementation in modulating immune activities in rats. Biol Trace Elem Res 200(8):3762–3773. https://doi.org/10.1007/s12011-021-02983-w
Hunt CD (1994) The biochemical effects of physiologic amounts of dietary boron in animal nutrition models. Environ Health Persp 102(suppl 7):35–43. https://doi.org/10.1289/ehp.94102s735
Osuka K, Watanabe Y, Usuda N, Atsuzawa K, Aoyama M, Niwa A, Nakura T, Takayasu M (2012) Activation of Ras/MEK/ERK signaling in chronic subdural hematoma outer membranes. Brain Res 13(1489):98–103. https://doi.org/10.1016/j.brainres.2012.10.013
Hadrup N, Frederiksen M, Sharma AK (2021) Toxicity of boric acid, borax and other boron containing compounds: a review. Regul Toxicol Pharm 121:104873. https://doi.org/10.1016/j.yrtph.2021.104873
Harvatine KJ, Boisclair YR, Bauman DE (2009) Recent advances in the regulation of milk fat synthesis. Animal 3(1):40–54. https://doi.org/10.1017/S1751731108003133
Li J, Huang Q, Long X, Zhang J, Huang X, Aa J, Yang H, Chen Z, **ng J (2015) CD147 Reprograms fatty acid metabolism in hepatocellular carcinoma cells through Akt/mTOR/SREBP1c and P38/PPARalpha pathways. J Hepatol 63(6):1378–89. https://doi.org/10.1016/j.jhep.2015.07.039
Kim YM, Shin HT, Seo YH, Byun HO, Yoon SH, Lee IK, Hyun DH, Chung HY, Yoon G (2010) Sterol regulatory element-binding protein (SREBP)-1-mediated lipogenesis is involved in cell senescence. J Biol Chem 285(38):29069–77. https://doi.org/10.1074/jbc.M110.120386
Han M, Zhang M (2021) The regulatory mechanism of amino acids on milk protein and fat synthesis in mammary epithelial cells: a mini review. Anim Biotechnol 1–11. https://doi.org/10.1080/10495398.2021.1950743.
Joel B, David EM (2002) The mechanisms of action of ppars. Annu Rev Med 53:409–35. https://doi.org/10.1146/annurev.med.53.082901.104018
Xu H, Luo J, Zhao W, Yang Y, Tian H, Shi H, Bionaz M (2016) Overexpression of SREBP1 (sterol regulatory element binding protein 1) promotes de novo fatty acid synthesis and triacylglycerol accumulation in goat mammary epithelial cells. J Dairy Sci 99(1):783–95. https://doi.org/10.3168/jds.2015-9736
Doğan A, Demirci S, Apdik H, Bayrak OF, Gulluoglu S, Tuysuz EC, Gusev O, Rizvanov AA, Nikerel E, Şahin F (2017) A new hope for obesity management: boron inhibits adipogenesis in progenitor cells through the Wnt/β-catenin pathway. Metabolism 69:130–142. https://doi.org/10.1016/j.metabol.2017.01.021
Abdik EA, Abdik H, Taşlı PN, Deniz AAH, Şahin F (2019) Suppressive role of boron on adipogenic differentiation and fat deposition in human mesenchymal stem cells. Biol Trace Elem Res 188(2):384–392. https://doi.org/10.1007/s12011-018-1428-5
Hunt CD, Stoecker BJ (1996) Deliberations and evaluations of the approaches, endpoints and paradigms for boron, chromium and fluoride dietary recommendations. J Nutri 126(9 Suppl):2441S-2451S. https://doi.org/10.1093/jn/126.suppl_9.2441S
Funding
This study was funded by the National Natural Science Foundation of China (project no.31501968), Major Special Projects of Science and Technology in Anhui Province (project no. 201903a06020002), Academic Support Project for University Top Talents in Anhui Province (gxbjZD2020080), Chuzhou Municipal Science and Technology Project (2022ZN002), and Anhui Provincial Scientific Research Planning Project (Excellent Youth Scientific Research Project) (2022AH030146).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Yanan Yang and Ya Yang. The first draft of the manuscript was written by Yanan Yang, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Y., Yang, Y., Li, X. et al. Effects of Boron on Fat Synthesis in Porcine Mammary Epithelial Cells. Biol Trace Elem Res 202, 190–198 (2024). https://doi.org/10.1007/s12011-023-03663-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03663-7