Abstract
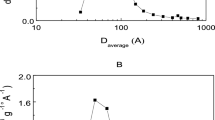
This work presents the immobilization in situ of commercial lipase from Candida antarctica B (CALB) by the sol–gel technique (xerogel) using silica from rice husk ash (RHA) as a source of silicon. It was used the Ionic Liquid (IL) 1-octyl-3-methylimidazolium bromide (C8MI.Br) as additive. The immobilized derivatives were characterized per SEM, XRD, and per method BET. The enzymatic activity of xerogels was evaluated with different tests, these being the reactional thermal analysis, immobilization yield, and operational and storage stability. The XDR showed that the obtained xerogels have halos in the region between 15 and 35° (2θ) what characterizes it as amorphous materials. The SEM analysis of xerogel shows irregular particles with dimensions less than 20 μm. The immobilized presented an esterification activity (EA) with 263.2 and 213.8 U/g, with and without IL, respectively, higher than the free enzyme (169.6 U/g). The immobilized, with and without IL, presented a significant improvement in the activity performance in relation to free enzyme for the three reactional temperatures (40, 60, and 80 °C) evaluated. The operational stability demonstrated that is possible to use xerogel without ionic liquid for 17 recycles and 21 recycles in IL presence. This methodology allows the preparation of new highly active and selective enzyme catalysts using the rice husk ash as a source of silicon, and the ionic liquid [C8MI]Br as additive. Furthermore, the new materials can provide greater viability in the processes, ensuring longer catalyst life.






Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Antunes, A., Ficanha, A. M. M., da Silva, L. M., Comin, T., Venquiaruto, L. D., Mignoni, M. L., Preczewski, J. D., Dallago, R. M., & Zeni, J. (2019). Imobilização da Lipase CALB em Xerogel Utilizando TMOS como Precursor sa Sílica e PVA como Aditivo. Revista Perspectiva, 43(163), 71–83. ISSN 0101-2908.
Azat, S., Korobeinyk, A. V., Moustakas, K., & Inglezakis, V. J. (2019). Sustainable production of pure silica from rice husk waste in Kazakhstan. Journal of Cleaner Production, 217, 352–359. https://doi.org/10.1016/j.jclepro.2019.01.142
Bakar, R. A., Yahya, R., & Gan, S. N. (2016). Production of high purity amorphous silica from rice husk. Procedia Chemistry, 19, 189–195. https://doi.org/10.1016/j.proche.2016.03.092
Battiston, C. S. Z., Ficanha, A. M. M., Oro, C. E. D., Dallago, R. M., & Mignoni, M. L. (2021). In situ calb enzyme immobilization in mesoporous material type MCM-48 synthesis using ionic solid [C14MI]Cl as structure-directing agent. Applied Biochemistry and Biotechnology, 194, 748. https://doi.org/10.1007/s12010-021-03648-z
Bordin, I., de Pedott, V. A., Oro, C. E. D., Junges, A., Dallago, R. M., & Mignoni, M. L. (2021). Nb-MCM-type mesoporous material synthesis using ionic solid as structure-directing agent for in situ lipase immobilization. Applied Biochemistry and Biotechnology, 193, 1072–1085. https://doi.org/10.1007/s12010-020-03484-7
Brunauer, S., Emmet, T. P. H., & Teller, E. (1938). Adsorption of gases in multimolecular layers. Journal of the American Chemical Society, 60, 309–319. https://doi.org/10.1021/ja01269a023
Chandrasekhar, S., Pramada, P. N., & Majeed, J. (2006). Effect of calcination temperature and heating rate on the optical properties and reactivity of rice husk ash. Journal of Materials Science, 41(23), 7926–7933. https://doi.org/10.1007/s10853-006-0859-0
Costa, J. A. S., & Paranhos, C. M. (2018). Systematic evaluation of amorphous silica production from rice husk ashes. Journal of Cleaner Production, 192, 688–697. https://doi.org/10.1016/j.jclepro.2018.05.028
Costantini, A., & Califano, V. (2021). Lipase immobilization in mesoporous silica nanoparticles for biofuel production. Catalysts, 11(5), 629. https://doi.org/10.3390/catal11050629
Costa-Silva, T. A., Carvalho, A. K. F., Souza, C. R. F., de Castro, H. F., Bachmann, L., Said, S., & Oliveira, W. P. (2021). Immobilized enzyme-driven value enhancement of lignocellulosic-based agricultural byproducts: application in aroma synthesis. Journal of Cleaner Production, 284, 124728. https://doi.org/10.1016/j.jclepro.2020.124728
Costa-Silva, T. A., Souza, C. R. F., Said, S., & Oliveira, W. P. (2015). Drying of enzyme immobilized on eco-friendly supports. African Journal Of Biotechnology, 14(44), 3019–3026. https://doi.org/10.5897/ajb2015.14830
Ficanha, A. M. M., Antunes, A., Oro, C. E. D., Dallago, R. M., & Mignoni, M. L. (2020). Immobilization of candida antarctica B (Calb) in silica aerogel: Morphological characteristics and stability. Biointerface Research in Applied Chemistry., 10(6), 6744–6756. https://doi.org/10.33263/BRIAC106.67446756
Ficanha, A. M. M., Antunes, A., Oro, C. E. D., Valduga, A. T., Matuella Moreira, C., Dallago, R. M., & Mignoni, M. (2019). Study of drying conditions of the aerogel obtained by the sol-gel technique for immobilization in situ of lipase Candida antarctica B. Industrial Biotechnology, 15(6), 350–356. https://doi.org/10.1089/ind.2019.0003
Ficanha, A. M. M., Oro, C. E. D., Franceschi, E., Dallago, R. M., & Mignoni, M. L. (2021). Evaluation of different ionic liquids as additives in the immobilization of lipase CAL B by sol-gel technique. Applied Biochemistry and Biotechnology, 193, 2162–2181. https://doi.org/10.1007/s12010-021-03533-9
Furlani, I. L., Amaral, B. S., Oliveira, R. V., & Cassa, Q. B. (2020). Imobilização Enzimática: Conceito e Efeitos na Proteólise. Quimica Nova, 43(4), 463–473. https://doi.org/10.21577/0100-4042.20170525
Girelli, A. M., Astolfi, M. L., & Scuto, F. R. (2020). Agro-industrial wastes as potential carriers for enzyme immobilization: A review. Chemosphere, 244, 125368. https://doi.org/10.1016/j.chemosphere.2019.125368
Holbrey, J. D., Seddon, K. R., & J. (1999). The phase behaviour of 1-alkyl-3-methylimidazolium tetrafluoroborates; Ionic liquids and ionic liquid crystals. Journal of the Chemical Society, Dalton Transactions, 13, 2133–2140. https://doi.org/10.1039/A902818H
Hossain, S. K. S., Mathur, L., & Roy, P. K. (2018). Rice husk/rice husk ash as an alternative source of silica in ceramics: A review. Journal of Asian Ceramic Societies, 6(4), 299–313. https://doi.org/10.1080/21870764.2018.1539210
Imam, H. T., Marr, P. C., & Marr, A. C. (2021). Enzyme entrapment, biocatalyst immobilization without covalent attachment. Green Chemistry, 23(14), 4980–5005. https://doi.org/10.1039/d1gc01852c
Imoisili, P. E., Ukoba, K. O., & Tien-Chien, Jen. (2020). Synthesis and characterization of amorphous mesoporous silica from palm kernel shell ash. Boletin de La Sociedad Espanola de Ceramica y Vidrio, 59(4), 159–164. https://doi.org/10.1016/j.bsecv.2019.09.006
Kaya, G. G., Yilmaz, E., & Deveci, H. (2020). Synthesis of sustainable silica xerogels/aerogels using inexpensive steel slag and bean pod ash: A comparison study. Advanced Powder Technology, 31(3), 926–936. https://doi.org/10.1016/j.apt.2019.12.013
Levandoski, K. L. D., Ficanha, A. M. M., Antunes, A., Dallago, R. M., & Mignoni, M. L. (2015) Imobilização Da Lipase Cal B Em Xerogel Obtido Pela Técnica Sol-Gel. Congresso Brasileiro de Engenharia Química em Iniciação Científica, 1(3), 51–60. Acesso: http://pdf.blucher.com.br.s3-sa-east1.amazonaws.com/chemicalengineeringproceedings/cobeqic2015/356-33965-260902.pdf
Lima, S. P. B. D., de Paiva Vasconcelos, R. P., Paiva, O. A., Cordeiro, G. C., de Morais Chaves, M. R., Filho, R. D. T., & de Fairbairn, E. M. R. (2011). Production of silica gel from rice husk ash ali w anderimam obadiah. Química Nova, 34, 71–75. https://doi.org/10.1590/S0100-40422011000100014
Lisboa, M. C., Rodrigues, C. A., Barbosa, A. S., Mattedi, S., Freitas, L. S., Mendes, A. A., Dariva, C., Franceschi, E., Lima, Á. S., & Soares, C. M. F. (2018). New perspectives on the modification of silica aerogel particles with ionic liquid used in lipase immobilization with platform in ethyl esters production. Process Biochemistry, 75, 157–165. https://doi.org/10.1016/j.procbio.2018.09.015
Machado, N. B., Miguez, J. P., Bolina, I. C. A., Salviano, A. B., Gomes, R. A. B., Tavano, O. L., Luiz, J. H. H., Tardioli, P. W., Cren, É. C., & Mendes, A. A. (2019). Preparation, functionalization and characterization of rice husk silica for lipase immobilization via adsorption. Enzyme and Microbial Technology, 128, 9–21. https://doi.org/10.1016/j.enzmictec.2019.05.001
Monteiro, R. R. C., Lima, P. J. M., Pinheiro, B. B., Freire, T. M., Dutra, L. M. U., Fechine, P. B. A., Gonçalves, L. R. B., De Souza, M. C. M., Dos Santos, J. C. S., & Fernandez-Lafuente, R. (2019). Immobilization of lipase a from Candida antarctica onto Chitosan-coated magnetic nanoparticles. International Journal of Molecular Sciences, 20, 16. https://doi.org/10.3390/ijms20164018
Nyari, N. L. D., Fernandes, I. A., Bustamante-Vargas, C. E., Steffens, C., de Oliveira, D., Zeni, J., Rigo, E., & Dallago, R. M. (2016). In situ immobilization of Candida antarctica B lipase in polyurethane foam support. Journal of Molecular Catalysis B: Enzymatic, 124, 52–61. https://doi.org/10.1016/j.molcatb.2015.12.003
Nyari, N. L. D., Zabot, G. L., Zamadei, R., Paluzzi, A. R., Tres, M. V., Zeni, J., Venquiaruto, L. D., & Dallago, R. M. (2018). Activation of Candida antarctica lipase B in pressurized fluids for the synthesis of esters. Journal of Chemical Technology and Biotechnology, 93, 897–908. https://doi.org/10.1002/jctb.5447
Onoja, E., Chandren, S., Razak, F. I. A., & Wahab, R. A. (2018). Extraction of nanosilica from oil palm leaves and its application as support for lipase immobilization. Journal of Biotechnology, 283, 81–96. https://doi.org/10.1016/j.jbiotec.2018.07.036
Pode, R. (2016). Potential applications of rice husk ash waste from rice husk biomass power plant. Renewable and Sustainable Energy Reviews, 53, 1468–1485. https://doi.org/10.1016/j.rser.2015.09.051
Quayson, E., Amoah, J., Hama, S., Kondo, A., & Ogino, C. (2020). Immobilized lipases for biodiesel production: Current and future greening opportunities. Renewable and Sustainable Energy Reviews, 134, 110355. https://doi.org/10.1016/j.rser.2020.110355
Reaño, R. L. (2020). Assessment of environmental impact and energy performance of rice husk utilization in various biohydrogen production pathways. Bioresource Technology, 299, 122590. https://doi.org/10.1016/j.biortech.2019.122590
Rizki, K., Pranowo, D., & Joko Raharjo, T. (2020). Immobilization of lipase in silica gel from rice husk ash and its activity assay to hydrolyze palm oil. BIO Web of Conferences, 28, 03005. https://doi.org/10.1051/bioconf/20202803005
Thommes, M., Kaneko, K., Neimark, A. V., Olivier, J. P., Rodriguez-Reinoso, F., Rouquerol, J., & Sing, K. S. W. (2015). Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure and Applied Chemistry, 87, 1051–1069. https://doi.org/10.1515/pac-2014-1117
Ulker, C., Gokalp, N., & Guvenilir, Y. (2016). Immobilization of Candida antarctica lipase B (CALB) on surface-modified rice husk ashes (RHA) via physical adsorption and cross-linking methods. Biocatalysis And Biotransformation, 34(4), 172–180. https://doi.org/10.1080/10242422.2016.1247818
**e, H., Huang, J., Woo, M. W., Hu, J., **ong, H., & Zhao, Q. (2021). Effect of cold and hot enzyme deactivation on the structural and functional properties of rice dreg protein hydrolysates. Food Chemistry, 345, 128784. https://doi.org/10.1016/j.foodchem.2020.128784
**e, W., & Zang, X. (2018). Lipase immobilized on ionic liquid-functionalized magnetic silica composites as a magnetic biocatalyst for production of trans-free plastic fats. Food Chemistry, 257, 15–22. https://doi.org/10.1016/j.foodchem.2018.03.010
Zhang, W., Wang, N., Zhang, L., Wu, W., Hu, C., & Yu, X. (2014). Effects of additives on lipase immobilization in microemulsion-based organogels. Applied Biochemistry and Biotechnology, 172(6), 3128–3140. https://doi.org/10.1007/s12010-014-0746-0
Zhong, L., He, C., **ao, C., Yao, C., Pyatt, I. H., & Lu, Y. (2021). Covalent immobilization of Candida antarctica Lipase B on functionalized hollow mesoporous silica nanoparticles. ChemistrySelect, 6(14), 3453–3460. https://doi.org/10.1002/slct.202100713
Acknowledgements
The authors thank to URI Erechim, National Council for Scientific and Technological Development (CNPq), Coordination for the Improvement of Higher Education Personnel (CAPES) and Research Support Foundation of the State of Rio Grande do Sul (FAPERGS).
Funding
This work was financially supported by CAPES, FAPERGS, and CNPq.
Author information
Authors and Affiliations
Contributions
J.F. Vesoloski: Investigation & Writing.
A.S. Todero: Investigation & Writing
R.J. Macieski: Investigation & Writing
F.O. Pereira: Investigation & Writing
R.M. Dallago: Formal Analysis, Writing, Review & Editing
M.L. Mignoni: Supervision, Formal Analysis, Writing, Review & Editing
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
All authors gave their consent to participate in the article. (Marcelo L. Mignoni—on behalf of all co-authors.)
Consent for Publication
All authors gave their consent for publication of the article if it was accepted. (Marcelo L. Mignoni—on behalf of all co-authors.)
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
•Silicon from rice husk ash was successfully used for in situ immobilization of CALB in xerogel.
•CALB immobilized in sol-gel presented 21 recycles with IL C8MI.Br and 17 without IL C8MI.Br.
•Enzymatic activity remained stable for 96 days; however, the tests just with enzyme reached 126 days.
•The supports were able to catalyze the esterification reaction at 40, 60, and 80 °C.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vesoloski, J.F., Todero, A.S., Macieski, R.J. et al. Immobilization of Lipase from Candida antarctica B (CALB) by Sol–Gel Technique Using Rice Husk Ash as Silic Source and Ionic Liquid as Additive. Appl Biochem Biotechnol 194, 6270–6286 (2022). https://doi.org/10.1007/s12010-022-04096-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04096-z