Abstract
Purpose of Review
Atrial fibrillation (AF), the most common sustained arrhythmia, is a major cause of stroke and systemic embolism, and is increasing in prevalence. Device closure of the left atrial appendage (LAA) represents a non-pharmacologic approach to stroke prevention in AF patients. This review presents the rationale for LAA closure (LAAC), describes current transcatheter approaches to LAAC, and summarizes the current evidence for LAAC for stroke prevention, highlighting the main randomized trials and the most recent data available.
Recent Findings
Meta-analysis of randomized clinical trials demonstrates similar rates of all-cause stroke with transcatheter LAAC compared with vitamin K antagonist therapy and significantly less bleeding with LAAC after cessation of mandated post-procedure pharmacology. Recent prospective observational studies, including those evaluating outcomes after commercial approval in the USA, show significantly improved procedure safety compared with earlier experiences.
Summary
LAAC appears to be an attractive alternative strategy for stroke prevention in AF patients, particularly in those who can take short-term oral anticoagulation (OAC), but are not optimal candidates for long-term OAC. Recent data suggests the procedure can be safely performed in patients with contraindications to OAC. Further, robust studies are needed to evaluate safety and efficacy in OAC-contraindicated patients, to compare outcomes with non-vitamin K antagonist OACs, and to explore the relative safety and efficacy of different LAAC devices.





Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • of Importance •• of Major Importance
Chugh SS, Havmoeller R, Narayanan K, Singh D, Rienstra M, Benjamin EJ, et al. Worldwide epidemiology of atrial fibrillation: a global burden of disease 2010 study. Circulation. 2014;129(8):837–47.
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, et al. Heart disease and stroke statistics—2013 update: a report from the American Heart Association. Vol. 127, Circulation 2013. 6–245 p.
De Backer O, Arnous S, Ihlemann N, Vejlstrup N, Jørgensen E, Pehrson S, et al. Percutaneous left atrial appendage occlusion for stroke prevention in atrial fibrillation: an update. Open Hear. 2014;1(1):e000020.
Lloyd-jones DM, Benjamin EJ, Jarett D, Borden WB, Bravata DM, Dai S, et al. Heart disease and stroke statistics—2012 update. A report from the American Heart Association. Circulation. 2015;125(1):2–22.
Blackshear JLOJA. Appendage obliteration to reduce stroke in cardiac surgical patients with atrial fibrillation. Ann Thorac Surg. 1996;61(2):755–9.
Johnson WD, Ganjoo AK, Stone CD, Srivyas RC, Howard M. The left atrial appendage: our most lethal human attachment! Surgical implications. Eur J Cardio-thoracic Surg. 2000;17(6):718–22.
January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: a report of the American college of cardiology/American heart association task force on practice guidelines and the heart rhythm society. J Am Coll Cardiol. 2014;64(21):2245–80.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;8;365(10):883–91.
Granger CB, Alexander JH, McMurray JJV, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;15;365(11):981–92.
Hsu JC, Maddox TM, Kennedy KF, Katz DF, Marzec LN, Lubitz SA, et al. Oral anticoagulant therapy prescription in patients with atrial fibrillation across the Spectrum of stroke risk. J Am Med Assoc Cardiol. 2016;1(1):55.
Albers GW, Diener HC, Drind M, Nevinson M, al w. **melagatran vs warfarin for stroke prevention in patients with atrial fibrillation: a randomized trial. J Am Med Assoc. 2005;293(6):690–8.
Olsson SB. Executive Steering Committee of the SPORTIF III Investigators. Stroke prevention with the oral direct thrombin inhibitor ximelagatran compared with warfarin in patients with non-valvular atrial fibrillation (SPORTIF III): randomised controlled trial. Lancet. 2003;22;362(9397):1691–8.
Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJGM, Lip GYHA. Novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation. Chest. 2010;1;138(5):1093–100.
Schulman S, Beyth RJ, Kearon C, Levine MN. Hemorrhagic complications of anticoagulant and thrombolytic treatment. Chest. 2008;133(6):257S–98S.
Sørensen R, Hansen ML, Abildstrom SZ, Hvelplund A, Andersson C, Jørgensen C, et al. Risk of bleeding in patients with acute myocardial infarction treated with different combinations of aspirin, clopidogrel, and vitamin K antagonists in Denmark: a retrospective analysis of nationwide registry data. Lancet. 2009;12;374(9706):1967–74.
Adam SS, McDuffie JR, Ortel TL, Williams JW. Comparative effectiveness of warfarin and new oral anticoagulants for the management of atrial fibrillation and venous thromboembolism: a systematic review. Ann Intern Med. 2012;4;157(11):796–807.
Kirley K, GouthamRao BV, Masi C. The role of NOACs in atrial fibrillation management: a qualitative study. J Atr Fibrillation. 2016;9(1):1416.
• Marzec LN, Wang J, Shah ND, Chan PS, Ting HH, Gosch KL, et al. Influence of direct oral anticoagulants on rates of oral anticoagulation for atrial fibrillation. J Am Coll Cardiol. 2017;69(20):2475–84. This paper describes the impact of newer oral anticoagulants on treatment rates, and further highlights the underutilization of oral anticoagulation in atrial fibrillation patients at risk for thromboembolism.
Xu H, Ruff CT, Giugliano RP, Murphy SA, Nordio F, Patel I, et al. Concomitant use of single antiplatelet therapy with edoxaban or warfarin in patients with atrial fibrillation: analysis from the engage af-timi48 trial. J Am Heart Assoc. 2016;5(2):1–9.
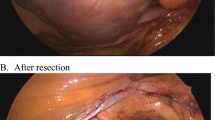
Madden JL. Resection of the left auricular appendix. J Am Med Assoc. 1949;2;140(9):769.
Holmes DR, Reddy VY, Turi ZG, Doshi SK, Sievert H, Buchbinder M, et al. Percutaneous closure of the left atrial appendage versus warfarin therapy for prevention of stroke in patients with atrial fibrillation: a randomised non-inferiority trial. Lancet. 2009;15;374(9689):534–42.
• Holmes DR, Kar S, Price MJ, Whisenant B, Sievert H, Doshi SK, et al. Prospective randomized evaluation of the watchman left atrial appendage closure device in patients with atrial fibrillation versus long-term warfarin therapy: the PREVAIL trial. J Am Coll Cardiol. 2014;8;64(1):1–12. This report describes the outcomes of the second randomized trial of the WATCHMAN LAA closure compared with warfarin, and confirms improved procedural safety compared with earlier trial experience.
•• Reddy VY, Sievert H, Halperin J, Doshi SK, Buchbinder M, Neuzil P, et al. Percutaneous left atrial appendage closure vs warfarin for atrial fibrillation a randomized clinical trial. J Am Med Assoc. 2014;19;312(19):1988–98. This first, randomized study demonstrates the non-inferiority of superiority of LAA closure compared with warfarin in patients who are candidates for long-term oral anticoagulation.
•• Reddy VY, Doshi SK, Kar S, Gibson DN, Price MJ, Huber K, et al. 5-year outcomes after left atrial appendage closure. J Am Coll Cardiol. 2017;pii:S0735–1097. This paper describes the final individual and pooled outcomes of the two randomized clinical trials of WATCHMAN LAA closure compared with warfarin.
Alli O, Doshi S, Kar S, Reddy V, Sievert H, Mullin C, et al. Quality of life assessment in the randomized PROTECT AF (percutaneous closure of the left atrial appendage versus warfarin therapy for prevention of stroke in patients with atrial fibrillation) trial of patients at risk for stroke with nonvalvular atrial. J Am Coll Cardiol. 2013;61(17):1790–8.
•• Price MJ, Reddy VY, Valderrábano M, Halperin JL, Gibson DN, Gordon N, et al. Bleeding outcomes after left atrial appendage closure compared with long-term warfarin a pooled, patient-level analysis of the WATCHMAN randomized trial experience. J Am Coll Cardiol. Cardiovasc Interv. 2015;8(15):1925–32. This analysis of randomized trial data demonstrates the reduction in post-procedural major bleeding in patients managed with a strategy of LAA closure compared with long-term oral anticoagulation with warfarin.
• Reddy VY, Möbius-Winkler S, Miller MA, Neuzil P, Schuler G, Wiebe J, et al. Left atrial appendage closure with the watchman device in patients with a contraindication for oral anticoagulation: The ASAP study (ASA plavix feasibility study with watchman left atrial appendage closure technology). J Am Coll Cardiol. 2013;25;61(25):2551–6. This observational study demonstrates the feasibility of LAA closure to reduce stroke in patients who have significant contraindications to oral anticoagulation.
Sharma D, Reddy VY, Sandri M, Schulz P, Majunke N, Hala P, et al. Left atrial appendage closure in patients with contraindications to oral anticoagulation. J Am Coll Cardiol. 2016;67(18):2190–2.
•• Boersma LV, Ince H, Kische S, Pokushalov E, Schmitz T, Schmidt B, et al. Efficacy and safety of left atrial appendage closure with WATCHMAN in patients with or without contraindication to oral anticoagulation: 1-year follow-up outcome data of the EWOLUTION trial. Hear Rhythm. 2017;14(9):1302–8. This large, observational study describes the commercial experience of WATCHMAN LAA closure outside of the USA, and shows that procedural complications have decreased since the early clinical trial experience.
Holmes DR, Reddy VY, Buchbinder M, Stein K, Elletson M, Bergmann MW, et al. The assessment of the Watchman device in patients unsuitable for oral anticoagulation (ASAP-TOO) trial. Am Heart J. 2017;189:68–74.
• Reddy VY, Gibson DN, Kar S, O’Neill W, Doshi SK, Horton RP, et al. Post-approval U.S. experience with left atrial appendage closure for stroke prevention in atrial fibrillation. J Am Coll Cardiol. 2017;24;69(3):253–61. This study represents the first description of the procedural outcomes of commercial implants of the WATCHMAN device in the United States.
• Saw J, Price MJ. Assessing the safety of early U.S. commercial application of left atrial appendage closure. J Am Coll Cardiol. 2016;69(3):262–4. Important critique of the strengths and limitations of procedural outcomes in the current commercial watchman experience.
Boersma LV, Schmidt B, Betts TR, Sievert H, Tamburino C, Teiger E, et al. Implant success and safety of left atrial appendage closure with the WATCHMAN device: peri-procedural outcomes from the EWOLUTION registry. Eur Heart J. 2016;37:2465–74.
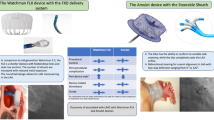
• Landmesser U, Schmidt B, Nielsen-Kudsk JE, Lam SCC, Park J-W, Tarantini G, et al. Left atrial appendage occlusion with the AMPLATZER Amulet device: periprocedural and early clinical/echocardiographic data from a global prospective observational study. EuroIntervention. 2017;1–10. This is the first large prospective study to evaluate the safety and efficacy of LAA closure with the Amulet device.
Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013;28;369(22):2093–104.
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;17;361(12):1139–51.
Koifman E, Lipinski MJ, Escarcega RO, Didier R, Kiramijyan S, Torguson R, et al. Comparison of Watchman device with new oral anti-coagulants in patients with atrial fibrillation: a network meta-analysis. Int J Cardiol. 2017;21;205:17–22.
Lempereur M, Aminian A, Freixa X, Gafoor S, Kefer J, Tzikas A, et al. Device-associated thrombus formation after left atrial appendage occlusion: a systematic review of events reported with the Watchman, the Amplatzer Cardiac Plug and the Amulet. Catheter Cardiovasc Interv. 2017;1:90(5):E111–21.
Viles-Gonzalez JF, Kar S, Douglas P, Dukkipati S, Feldman T, Horton R, et al. The clinical impact of incomplete left atrial appendage closure with the watchman device in patients with atrial fibrillation. J Am Coll Cardiol. 2012;59(10):923–9.
Saw J, Tzikas A, Shakir S, Gafoor S, Omran H, Nielsen-Kudsk JE, et al. Incidence and clinical impact of device-associated thrombus and peri-device leak following left atrial appendage closure with the Amplatzer Cardiac Plug. JACC Cardiovasc Interv. 2017 Feb;10(4):391–9.
Price MJ, Valderrábano M. Left atrial appendage closure to prevent stroke in patients with atrial fibrillation. Circulation. 2014;8;130(2):202–12.
• Price MJ, Gibson DN, Yakubov SJ, Schultz JC, Di Biase L, Natale A, et al. Early safety and efficacy of percutaneous left atrial appendage suture ligation: results from the U.S. transcatheter LAA ligation consortium. J Am Coll Cardiol. 2014;12;64(6):565–72. This paper is the first multicenter study to examine the outcomes of transcatheter LAA ligation, and defines the relatively high procedural risk associated with the procedure.
• Gianni C, Di Biase L, Trivedi C, Mohanty S, Göko Y, Güne MF, et al. Clinical implications of leaks following left atrial appendage ligation with the LARIAT device. J Am Coll Cardiol. 2016;9(10):1051–7. This study links residual or late leaks after transcatheter LAA ligation with thromboembolic events.
Mosley WJ, Smith MR, Price MJ. Percutaneous management of late leak after lariat transcatheter ligation of the left atrial appendage in patients with atrial fibrillation at high risk for stroke. Catheter Cardiovasc Interv. 2014;83(4):664–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Matthew J. Price has received consulting honoraria from Abbott Vascular, Boston Scientific, W.L. Gore, Medtronic, and AstraZeneca; has received speaker honoraria from Abbott Vascular, Medtronic, Terumo, and AstraZeneca; has received research grants from Daiichi Sankyo; has served as a site investigator for trials involving the Watchman device; has received honoraria as a proctor for Abbott Vascular and Boston Scientific; and is a member of the Steering Committee for the NCDR LAAO registry.
Ricardo Kosturakis has nothing to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Structural Heart Disease
Rights and permissions
About this article
Cite this article
Kosturakis, R., Price, M.J. Current State of Left Atrial Appendage Closure. Curr Cardiol Rep 20, 42 (2018). https://doi.org/10.1007/s11886-018-0981-z
Published:
DOI: https://doi.org/10.1007/s11886-018-0981-z