Abstract
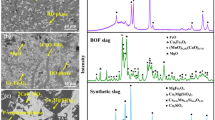
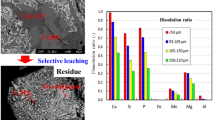
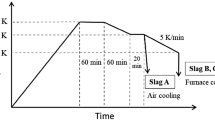
In the present paper, dephosphorization of steelmaking slag by leaching with acidic aqueous solution composed of citric acid, sodium hydroxide, hydrochloric acid and ion-exchanged water was investigated. The buffer solution of C6H8O7-NaOH-HCl system prevented changes in the pH values. Kinetic parameters including leaching temperature, slag particle size and pH values of the solution were optimized. The results showed that temperature has no obvious effect on the dissolution ratio of phosphorus. However, it has a significant effect on the dissolution ratio of iron. The dephosphorization rate increases with the decrease of slag particle size and the pH value of the solution. Over 90% of the phosphorus can be dissolved in the solution while the corresponding leaching ratio of iron was only 30% below the optimal condition. Leaching kinetics of dephosphorization follow the unreacted shrinking core model with a rate controlled step by the solid diffusion layer, the corresponding apparent activation energy being 1.233 kJ mol−1. A semiempirical kinetic equation was established. After leaching, most of the nC2S-C3P solid solution in the steelmaking slag was selectively dissolved in the aqueous solution and the iron content in the solid residue was correspondingly enriched.














Similar content being viewed by others
References
X. Gao, M. Okubo, N. Maruoka, H. Shibata, T. Ito, and S.Y. Kitamura, Trans. Inst. Min. Metall. C 124, 116 (2015).
D.M. Proctor, K.A. Fehling, E.C. Shay, J.L. Wittenborn, J.J. Greem, C. Avent, R.D. Bigham, M. Connolly, B. Lee, T.O. Shepker, and M.A. Zak, Environ. Sci. Technol. 34, 1576 (2000).
R. Dippenaar, Ironmak. Steelmak. 32, 35 (2005).
H. Shen and E. Forssberg, Waste Manag. 23, 933 (2003).
W. Fix, H. Heymann, and R. Heinke, J. Am. Ceram. Soc. 52, 346 (1969).
K. Ito, M. Yanagisawa, and N. Sano, Tetsu-to-Hagane (J. Iron Steel Inst. Jpn.) 68, 342 (1982).
F. Pahlevani, S.Y. Kitamura, H. Shibata, and N. Maruoka, ISIJ Int. 50, 822 (2010).
H. Ono, Tetsu-to-Hagane (J. Iron Steel Inst. Jpn.) 66, 1317 (1980).
T. Fujita and I. Iwasaki, Iron Steelmak. 16, 47 (1989).
H. Kubo, K. Matsubae-Yokoyama, and T. Nagasaka, ISIJ Int. 50, 59 (2010).
J. Diao, B. **e, Y.H. Wang, and X. Guo, ISIJ Int. 52, 955 (2012).
T. Miki, K. Shitogiden, Y. Samada, T. Nagasaka, and M. Hino, Tetsu-to-Hagane (J. Iron Steel Inst. Jpn.) 89, 388 (2003).
T. Futatsuka, K. Shitogiden, T. Miki, and M. Hino, Tetsu-to-Hagane (J. Iron Steel Inst. Jpn.) 89, 382 (2003).
T. Futatsuka, K. Shitogiden, T. Miki, and M. Hino, ISIJ Int. 44, 753 (2004).
T. Teratoko, N. Maruoka, H. Shibata, and S.Y. Kitamura, High Temp. Mater. Proc. 31, 329 (2012).
M. Numata, N. Maruoka, S.J. Kim, and S.Y. Kitamura, ISIJ Int. 54, 1983 (2014).
O. Levenspiel, Chemical Reaction Engineering, 2nd ed. (New York: Wiley, 1999), pp. 566–590.
E.A. Abdel-Aal, Hydrometallurgy 55, 247 (2000).
Z. Yang, H.Y. Li, X.C. Yin, Z.M. Yan, X.M. Yan, and B. **e, Int. J. Miner. Process. 133, 105 (2014).
M. Ashraf, Z.I. Zafar, and T.M. Ansari, Hydrometallurgy 80, 286 (2005).
A. Ekmekyapar, E. Aktaş, A. Künkül, and N. Demirkiran, Metall. Mater. Trans. B 43, 764 (2012).
Acknowledgement
This work was partially supported by the Fundamental Research Funds for the Central Universities (Project CDJZR 14130001) and National Basic Research Program of China (No. 2013CB632604). We express our gratitude to all those who helped us during the writing of this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qiao, Y., Diao, J., Liu, X. et al. Dephosphorization of Steelmaking Slag by Leaching with Acidic Aqueous Solution. JOM 68, 2511–2519 (2016). https://doi.org/10.1007/s11837-015-1760-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-015-1760-6