Abstract
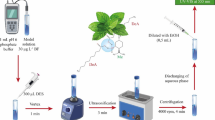
Hydrophobic deep eutectic solvent (DES) was synthesized from TBAB and decanoic acid and used to study the extraction of aromatic hydrocarbon from contaminated water samples. DES was screened by selecting different ratios between the TBAB and decanoic acid. A ratio of 1: 2 was observed to be the best among others and was used for the desired application. Within the temperature range of 25–70 °C, physicochemical properties such as density (944–915 kg/m3), viscosity (1,636–179 mPa·s), conductivity (141–1,007 µS/cm), and pH (3.14–2.73) of the synthesized DES were determined. For the extraction study, benzene, toluene, and xylene (BTX)-doped water were used as a simulated contaminated water. Response surface methodology was employed in modeling and optimizing the effects of temperature, time, and solvent mass fraction on the extraction efficiency of the DES. Extraction efficiency of 68.1%, 70.84% and 77.73% for BTX, respectively, was recorded at optimum values of 25 °C, 60 min and 0.6 (solvent mass fraction). Extraction efficiency as high as 86.61%, 88.94%, and 92.71% for BTX, respectively, can be obtained within the design space. Effective regeneration and reuse of the DES after each extraction was carried out for five consecutive cycles; their results showed no significant decrease in their respective extraction efficiencies and recovery of the DES. This, therefore, improves the overall performance of hydrophobic DES for the extraction process.
Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
E. Manoli and C. Samara, Trends Analyt. Chem., 18, 417 (1999).
N. U. Benson, J. P. Essien, F. E. Asuquo and A. L. Eritobor, Environ. Monit. Assess., 186, 5519 (2014).
T. Khezeli, A. Daneshfar and R. Sahraei, J. Chromatogr. A., 1425, 25 (2015).
A. M. Taiwo, O. O. Olujimi, O. Bamgbose and T. A. Arowolo, Surface water monitoring in nigeria: Situational analysis and future management strategy, water quality monit. assess., InTech publications, London (2012).
K. Z. Kponee, A. Chiger, I. I. Kakulu, D. Vorhees and W. Heiger-Bernays, J. Environ. Health Sci. Eng., 14, 86 (2015).
L. F. Lima, J. I. R. De-Andrade, M. G. Da-Silva and M. G. Vieira, Ind. Eng. Chem. Res., 56, 6326 (2017).
A. P. Abbott, G. Capper, D. L. Davies, R. K. Rasheed and V. Tambyrajah, Chem. Commun., 1, 70 (2003).
P. Makos, A. Przyjazny and G. Boczkaj, J. Chromatogr. A., 1570, 28 (2018).
M. K. I. Hadj-Kali and M. Hayyan, J. Chem. Technol. Biotechnol., 93, 945 (2018).
Gahiszka, Z. Migaszewski and J. Namiesnik, Trends Analyt. Chem., 50, 78 (2013).
D.J. van Osch, L. F. Zubeir, A. van den Bruinhorst, M.A. Rocha and M. C. Kroon, J. Green Chem., 17, 4518 (2015).
Florindo, L. Branco and I. Marrucho, Fluid Phase Equilib., 448, 135 (2017).
M. Faraji, Microchem. J., 150, 104130 (2019).
S. Garcia, M. Gil, J. Pis, F. Rubiera and C. Pevida, Int. J. Greenh. Gas Control, 12, 35 (2013).
A. K. Dwamena, J. Sep. Sci., 6, 9 (2019).
B. Socas-Rodriguez, A. Santana-Mayor, A. V. Herrera-Herrerra and M. A. Rodriguez-Delgado, Green Sustain. Process Chem. Environ. Eng. Sci., 5, 123 (2020).
Z.S. Gano, F. S. Mjalli, T. Al-Wahaibi, Y. Al-Wahaibi and I. M. AlNashef, Chem. Eng. Process, 93, 10 (2015).
C. Montgomery and G. C. Runger, Applied statistics and probability for engineers, John Wiley & Sons Publications, New Jersey (2010).
B. Jibril, F. Mjalli, J. Naser and Z. Gano, J. Mol. Liq., 199, 462 (2014).
S. M. Yousefi, F. Shemirani and S. A. Ghorbanian, J. Chromatographia, 81, 1201 (2018).
J. G. P. van Osch, C. H. J. T. Dietz, J. van Spronsen, M. C. Kroon, F. Gallucci, M. V. S. Annaland and R. Tuinier, ACS. Sustain. Chem. Eng., 7, 2933 (2019).
H. Ghaedi, M. Ayoub, S. Sufian, S. M. Hailegiorgis, G. Murshid, S. Farrukh and S. N. Khan, Thermochim. Acta., 657, 123 (2017).
A. Hayyan, F. S. Mjalli, I. M. AlNashef, T. Al-Wahaibi, Y. M. Al-Wahaibi and M. A. Hashim, Thermochim. Acta., 541, 70 (2012).
X. Li and K. H. Row, J. Sep. Sci., 39, 3505 (2016).
L. Zhao and H. K. Lee, J. Chromatogr. A., 919, 381 (2001).
D. Ge, Y. Zhang, Y. Dai and S. Yang, J. Sep. Sci., 41, 1635 (2018).
T. Li, Y. Song, J. Xu and J. Fan, Talanta, 195, 298 (2019).
K. S. Kim, J. Y. Lee, S. J. Lee, T. K. Ha and D. H. Kim, J. Am. Chem. Soc., 116, 7399 (1994).
J. Gao, L. W. Chou and A. Auerbach, Biophys. J., 65, 43 (1993).
A. Orabi and G. Lamoureux, J. Phys. Chem. B., 122, 2251 (2018).
D. A. Dougherty and D. A. Stauffer, J. Sci., 250, 1558 (1990).
L. Metcalf, Waste water engineering: Treatment and reuse, Metcalf and Eddy Inc. (2003).
W. H. Organization, A compendium of standards for waste water reuse in the Eastern Mediterranean Region (2006).
Acknowledgements
The authors appreciate the National Research Institute for Chemical Technology (NARICT), Zaria, Nigeria and the Department of Chemical Engineering, Ahmadu University, Zaria, Nigeria for financial and technical supports rendered in carrying out this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Rights and permissions
About this article
Cite this article
Yakubu, A., Gano, Z.S., Ahmed, O.U. et al. Application of hydrophobic deep eutectic solvent for the extraction of aromatic compounds from contaminated water. Korean J. Chem. Eng. 39, 1299–1306 (2022). https://doi.org/10.1007/s11814-021-0994-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-0994-y