Abstract
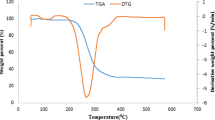
Characterization and thermal pyrolysis of oil sand was conducted. The experiment was performed on Circle Cliffs, Utah, U.S.A. and the results were compared with the data from Alberta, Canada. The reaction character identified by TGA was dual mode of vaporization of light hydrocarbon and thermal cracking of high molecular hydrocarbon. The pyrolysis experiment was performed in a 2 kg/h capacity fluidized bed externally heated by electricity. The process variables investigated were a temperature range of 723-923 K, fluidization gas velocity of 1.5-2 times of the minimum fluidization velocity, solid retention time of 15-30 minutes, and average particle size of 435 microns. The results of TGA and elemental analysis of bitumen provided necessary information regarding maximum liquid yield from the pyrolysis prior to pyrolysis experiment. The oil yield was maximum at 823 K. The yield of liquid was not exceeding the weight percent of maltenes in original bitumen. The optimum reaction condition should be fast vaporization of light hydrocarbon and minimizing thermal cracking of high molecular hydrocarbon. To maximize the liquid yield, fast heating and vaporization of oil sand bitumen and then the rapid removal of the vaporized product from the heating zone is recommended, i.e., operation in a fluidized bed reactor.
Similar content being viewed by others
References
Oil Shale & Tar Sands Programmatic EIS, http://ostseis.anl.gov/ guide/tarsands/index.htm (2017).
R. Engelhardt and M. Todirscu, An Introduction to Development in Alberta’s Oil Sand, University of Alberta School of Business (2005).
J. Masliyah, Z. Zhou, Z. Xu, J. Czarnecki and H. Hamza, Can. J. Chem. Eng., 82(4), 628 (2004).
Q. Dai and K. H. Chung, Fuel, 75(2), 220 (1996).
C. J. Coronella and J.D. Seader, Fuel, 71, 143 (1992).
J.-S. Shin, Y. K. Sun, Y. C. Park, D.-H. Bae, S.-H. Jo and D. Shun, Korean Chem. Eng. Res., 48(1), 68 (2010).
J.V. Fletcher, M.D. Deo and F.V. Hanson, Power Technol., 76, 141 (1993).
H. Alboudwarej, J. Beck, W.Y. Svrcek, H.W. Yarranton and K. Akbarzadeh, Energy Fuels, 16(2), 462 (2002).
S. H. Ng, A. Humprhies, C. Fairbridge, Y. Zhu, C. Kluibe, T. Tsai, F. Ding, J.-P. Charland and S. Yui, Fuel Process. Technol., 86, 1335 (2005).
E. Czarnecka and J. E. Gillott, Clays and Clay Minerals, 28(3), 197 (1980).
Y.C. Park, J.-Y. Pack, D. H. Bae and D. Shun, Korean J. Chem. Eng., 26(6), 1608 (2009).
J.V. Fletcher, M.D. Deo and F.V. Hanson, Fuel, 74(3), 311 (1995).
Q. Liu, Z. Cui and T. H. Etsell, Fuel, 85, 807 (2007).
O. C. Mullins, E.Y. Sheu, A. Hammami and A. G. Marshall (eds.) Asphaltenes, Heavy Oils and Petroleomics, Springer, New York (2007).
F.V. Hanson, S. M. Cha, M.D. Deo and A. G. Oblad, Fuel, 71(12), 1455 (1992).
J. H. Gary and G. E. Handwerk, Petroleum refining, Marcel Dekker, New York (1994).
C. Phillips, R. Luymes and T. M. Hlahei, Fuel, 61, 639 (1982).
D.D. Deo, J.V. Fletcher, D. Shun, F.V. Hanson and A. G. Oblad, Fuel, 70, 1271 (1991).
D.C. Longstaff, M.D. Deo and F.V. Hanson, Fuel, 73(9), 1523 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shun, D., Shin, JS., Bae, DH. et al. A comparison of fluidized bed pyrolysis of oil sand from Utah, USA, and Alberta, Canada. Korean J. Chem. Eng. 34, 3125–3131 (2017). https://doi.org/10.1007/s11814-017-0233-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-017-0233-8