Abstract
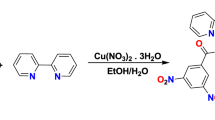
An energetic Copper (II) complex based on 3,5 dinitrobenzoic acid (DNBA) was synthesized from a copper nitrate, 3,5 dinitrobenzoic acid and 1,10 phenanthroline. This complex was characterized by elemental analysis, FTIR and UV–Vis spectrometry. Moreover, the structure of complex was studied by the single crystal X-ray diffraction method. The spectroscopic and XRD results confirmed that the complex consisted of mononuclear units comprising of two ligands coordinated with copper metal via nitrogen and oxygen 1,10 phenanthroline and DNBA with 1:1 stoichiometry. The apparent activation energies were determined from its derivative thermogravimetry (DTG) curves using Ozawa and Kissinger methods and were found to be 126.4 and 119.7 kJ mol−1 for the first stage and 178.1 and 170.4 kJ mol−1 for second stage, respectively. The results of the activation energies obtained from Flynn–Wall–Ozawa (FWO) and Kissinger–Akahira–Sunose (KAS) methods are similar and consistent to those obtained from the ASTM E698 method. The reaction models were calculated through model fitting by using Coats-Redfern and Madhusudanan methods and the results indicate that it is probably best described by Avrami-Erofeev (A4) model. Furthermore, critical ignition temperature, self-accelerating degradation temperature and thermodynamic parameters were also investigated. The sensitivity study reveals that energetic complex is less sensitive than the conventional RDX and HMX.
















Similar content being viewed by others
References
Agrawal JP (2015) High energy materials, propellants, explosives and pyrotechnics. Wiley-VCH, Weinheim
Akahira T, Sunose T (1971) Method of determining activation deterioration constant of electrical insulating materials. Res Report CHIBA Inst Technol 6:22–31
ASTM E698 (1879) Test methods for Arrhenius kinetic constants for thermally unstable materials 1979, ASTM, Philadelphia, PA
Baran E J (2005) Baran Structural Data and Vibrational Spectra of the Copper(II) Complex of L-Selenomethionine. Zeitschrift für Naturforschung B. 60b, 663–666. doi.org/https://doi.org/10.1515/znb-2005-0609
Boonchom B, Puttawong S (2010) Thermodynamics and kinetics of the dehydration reaction of FePO4.2H2O. Physica B 405:2350–2355. https://doi.org/10.1016/j.physb.2010.02.046
Boonchom B, Thongkam M (2010) Kinetics and thermodynamics of the formation of MnFeP4O12. J Chem Eng Data 55:211–216. https://doi.org/10.1021/je900310m
Coats AW, Redfern JP (1964) Kinetic Parameters from Thermogravimetric Data Nature (london) 201:68–69. https://doi.org/10.1038/201068a0
Fischer N, Klapötke TM, Stierstorfer J (2009) New nitriminotetrazoles - synthesis, structures and characterization. Z Anorg Allg Chem 635:271–281. https://doi.org/10.1002/zaac.200800430
Fischer D, Klapötke TM, Stierstorfer J (2014) Potassium 1,1’-Dinitramino-5,5’-bistetrazolate: A primary explosive with fast detonation and high initiation power. Angew Chem Int Ed 53:8172–8175. https://doi.org/10.1002/anie.201404790
Flynn JH, Wall LA (1966) General treatment of the thermogravimetry of polymers. J Res Natl Bur Std A Phys Chem 70:487–523. https://doi.org/10.6028/jres.070A.043
Friedman Y, Goldberg I (2018) Tetrazole-bridged manganese coordination polymer as high energy material. Polyhedron 139:327–330. https://doi.org/10.1016/j.poly.2017.11.010
Haiges R, Christe KO (2015) 5-(Fluorodinitromethyl)-2H-tetrazole and its tetrazolates - Preparation and characterization of new high energy compounds. Dalton Trans 44:10166–10176. https://doi.org/10.1039/C5DT00291E
Kaur M, Yathirajan HS, Byrappa K, Hosten E (2014) Crystal structure of desvenlafaxinium 3,5-dinitrobenzoate 3,5-dinitrobenzoic acid monohydrate, C30H35N5O15. Z Kristallogr NCS 229:488–490. https://doi.org/10.1515/ncrs-2014-0131
Keshavarz MH, Klapötke TM (2019) A novel method for prediction of the critical diameter of solid pure and composite high explosives to assess their explosion safety in an industrial setting. J Energ Mater 37:331–339. https://doi.org/10.1080/07370652.2019.1613697
Kissinger H (1956) Variation of peak temperature with heating rate in differential thermal analysis. J Res Natl Bur Stand 57:217–221
Klapötke TM, Sabate CM (2009) Safe 5-nitrotetrazolate anion transfer reagents. Dalton Trans 10:1835–1841. https://doi.org/10.1039/B818900P
Klapötke TM, Stierstorfer J (2007) Nitration products of 5-amino-1H-tetrazole and methyl-5-amino-1H-tetrazoles - structures and properties of promising energetic materials. Helv Chim Acta 90:2132–2150. https://doi.org/10.1002/hlca.200790220
Klapötke TM, Stierstorfer J (2008) The new energetic compounds 1,5-diaminotetrazolium and 5-amino-1-methyltetrazolium dinitramide - synthesis, characterization and testing. Eur J Inorg Chem 26:4055–4062. https://doi.org/10.1002/ejic.200890071
Klapötke TM, Stierstorfer J (2009) Azidoformamidinium and 5-aminotetrazolium dinitramide-two highly energetic isomers with a balanced oxygen content. Dalton Trans 4:643–653. https://doi.org/10.1039/B811767E
Klapötke TM, Mayer P, Sabate CM, Welch JM, Wiegand N (2008a) Simple, nitrogen-rich, energetic salts of 5-nitrotetrazole. Inorg Chem 47:6014–6027. https://doi.org/10.1021/ic800353y
Klapötke TM, Stierstorfer J, Wallek AU (2008c) Nitrogen-rich salts of 1-methyl-5-nitriminotetrazolate: An auspicious class of thermally stable energetic materials. Chem Mater 20:4519–4530. https://doi.org/10.1021/cm8004166
Klapötke TM, Sabate CM, Rasp M (2009a) Synthesis and properties of 5-nitrotetrazole derivatives as new energetic materials. J Mater Chem 19:2240–2252. https://doi.org/10.1039/B818925K
Klapötke TM, Sabate CM, Welch JM (2009b) Alkaline earth metal salts of 5-Nitro-2H-tetrazole: prospective candidates for environmentally friendly energetic applications. Eur J Inorg Chem 6:769–776. https://doi.org/10.1002/ejic.200801080
Klapötke TM, Stierstorfer J, Weber B (2009c) New energetic materials: synthesis and characterization of copper 5-nitriminotetrazolates. Inorg Chim Acta 362:2311–2320. https://doi.org/10.1016/j.ica.2008.10.014
Klapötke TM, Sabate CM, Welch JM (2008b) Alkali metal 5-nitrotetrazolate salts: prospective replacements for service Lead(II) azide in explosive initiators. Dalton Trans 45:6372–6380
Li Z, Yuan Y, Zhang Y, Liu L, Zhang S (2017) Synthesis, Crystal Structure, and Properties of Energetic Copper (II) Complex based on 3,5-Dinitrobenzoic Acid and 1,5-Diaminotetrazole. Z Anorg Allg Chem 643:647–652. https://doi.org/10.1002/zaac.201700086
Liu X, Su Z, Ji W (2014a) Structure, physicochemical property and DFT calculation of high-energy density materials constructed with intermolecular interaction: nitro group charge determines sensitivity. J Phys Chem C 118:23487–23498. https://doi.org/10.1021/jp5062418
Liu X, Su Z, Ji W, Chen S, Wei Q, Yang X, Gao S (2014b) Structure, physicochemical properties, and density functional theory calculation of high-energy-density materials constructed with intermolecular interaction: nitro group charge determines sensitivity. J Phys Chem C 118:23487–23498. https://doi.org/10.1021/jp5062418
Liu X, Yang Q, Su Z, Chen S, **e G, Wei Q (2014c) Gao S (2014c) 3D high-energy-density and low sensitivity materials: synthesis, structure and physicochemical properties of an azide–Cu(ii) complex with 3,5-dinitrobenzoic acid. RSC Adv 4:16087–16093. https://doi.org/10.1039/C4RA00635F
Madhusudanan PM, Krishnan K, Ninan KN (1986) New approximation for the p(x) function in the evaluation of non-isothermal kinetic data. Thermochim Acta 97:189–201. https://doi.org/10.1016/0040-6031(86)87019-8
Madhusudanan PM, Krishnan K, Ninan KN (1993) New equations for kinetic analysis of non-isothermal reactions. Thermochim Acta 221:13–21. https://doi.org/10.1016/0040-6031(93)80519-G
Nie X, Chen MS, Yu JX (2013) Synthesis, Crystal Structure, Thermal Stability and Luminescent Properties of Cd(II) Complex Constructed from 3,5-Dinitrobenzoic Acid. Chin J Inorg Chem 29:2704–2708. https://doi.org/10.3969/j.issn.1001-4861.2013.00.395
Ozawa T (1965) A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn 38:1881–1886. https://doi.org/10.1246/bcsj.38.1881
Singh A, Soni PK, Singh M, Srivastava A (2012) Thermal degradation, kinetic and correlation models of poly(vinylidene-fluoride–chlorotrifluoroethylene) copolymers. Thermochim Acta 548:88–92. https://doi.org/10.1016/j.tca.2012.08.031
Singh A, Soni PK, Shekharam T, Srivastava A (2013b) A study on thermal behaviour of a poly (VDF-CTFE) copolymers binder for high energy materials. J Appl Polym Sci 127:1751–1757. https://doi.org/10.1002/app.37780
Singh A, Sharma TC, Narang JK, Kishore P (2017) Thermal decomposition and kinetics of plastic bonded explosives based on mixture of HMX and TATB with polymer matrices. Def Technol 13:22–32. https://doi.org/10.1016/j.dt.2016.11.005
Singh A, Singh S, Sharma TC, Kishore P (2018b) Physicochemical properties and kinetic analysis for some fluoropolymers by differential scanning calorimetry. Polym Bull 75:2315–2338. https://doi.org/10.1007/s00289-017-2153-5
Singh A, Kumar R, Soni PK, Singh V (2019a) Compatibility and thermokinetics studies of octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine with polyether-based polyurethane containing different curatives. J Energ Mater 37:141–153. https://doi.org/10.1080/07370652.2018.1552337
Singh A, Soni PK, Sarkar C, Mukherjee N (2019c) Thermal reactivity of aluminized polymer-bonded explosives based on non-isothermal thermogravimetry and calorimetry measurements. J Therm Anal Calorim 136:1021–1035. https://doi.org/10.1007/s10973-018-7730-9
Singh A, Kumar M, Soni PK, Singh M, Srivastava A (2013a) Mechanical and explosive properties of plastic bonded explosives based on mixture of HMX and TATB. Def Sci J 63:622–629. https://doi.org/10.14429/dsj.63.5764
Singh A, Kaur G, Sarkar C, Mukherjee N (2018a) Investigations on chemical, thermal decomposition behavior, kinetics, reaction mechanism and thermodynamic properties of aged TATB. Cent Eur J Energ Mater 15:258–282
Singh A, Sharma TC, Singh V, Mukherjee N (2019b) Thermal degradation behaviour and kinetics of aged TNT-based melt cast composition B. Cent Eur J Energ Mater 16:360–379. https://doi.org/10.22211/cejem/112234
Srinivasan K, Stalin T (2014) Study of inclusion complex between 2,6-dinitrobenzoic acid and b-cyclodextrin by 1H NMR, 2D 1H NMR (ROESY), FT-IR, XRD, SEM and photophysical methods. Spectrochim Acta Part A Mol Biomol Spectrosc 130:105–115. https://doi.org/10.1016/j.saa.2014.03.106
Turmanova SC, Genieva SD, Dimitrova AS, Vlaev LT (2008) Non-isothernal degradation kinetics of filled with rice husk ash polypropene composites. Polym Letters 2:133–146. https://doi.org/10.3144/expresspolymlett.2008.18
Vlaev L, Nedelchev N, Gyurova K, Zagorcheva M (2008) A comparative study of non-isothermal kinetics of decomposition of calcium oxalatemonohydrate. J Anal Appl Pyrolysis 81:253–262. https://doi.org/10.1016/j.jaap.2007.12.003
Vyazovkin S, Burnham AK, Criado JM, Maqueda LAP, Popescu C, Sbirrazzuoli N (2011) ICTAC kinetics committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta 520:1–19. https://doi.org/10.1016/j.tca.2011.03.034
Wang Y, Zhang J, Su H, Li S, Zhang S, Pang S (2014) A simple method for the prediction of the detonation performances of metal-containing explosives. J Phys Chem A 118:4575–4581. https://doi.org/10.1021/jp502857d
Wang K, Zeng DH, Zhang JG, Cui Y, Zhang TL, Li ZM, ** X (2015) Controllable explosion: fine-tuning the sensitivity of high-energy complexes. Dalton Trans 44:12497–12501. https://doi.org/10.1039/C5DT01462J
Xu C, Zhang J, Yin X, ** X, Li T, Zhang TL, Zhou ZN (2015) Cd(II) complexes with different nuclearity and dimensionality based on 3-hydrazino-4-amino-1,2,4-triazole. J Solid State Chem 226:59–65. https://doi.org/10.1016/j.jssc.2015.02.004
Xue H, Gao H, Twamley B, Shreeve JM (2007) Energetic salts of 3-nitro-1,2,4-triazole-5-one, 5-nitroaminotetrazole, and other nitro-substituted azoles. Chem Mater 19:1731–1739. https://doi.org/10.1021/cm062964g
Yang Q, Chen SP, **e G, Gao SL (2011) Synthesis and characterization of an energetic compound Cu(Mtta)2(NO3)2 and effect on thermal decomposition of ammonium perchlorate. J Hazard Mater 197:199–203. https://doi.org/10.1016/j.jhazmat.2011.09.074
Yin P, Parrish DA, Shreeve JM (2015) Energetic multi-functionalized nitraminopyrazoles and their ionic derivatives: ternary hydrogen-bond induced high energy density materials. J Am Chem Soc 137:4778–4786
Yu XK, Zheng YQ (2013) Syntheses, structures, dielectric and ferroelectric properties of a chiral coordination compound with m-nitro-benzoic acid. J Coord Chem 66:2208–2216. https://doi.org/10.1080/00958972.2013.803219
Zhang S, Yang Q, Liu X, Qu X, Wei Q, **e G, Chen S, Gao S (2016a) High-energy metal–organic frameworks (HE-MOFs): synthesis, structure and energetic performance. Coord Chem Rev 307:292–312. https://doi.org/10.1016/j.ccr.2015.08.006
Zhang JH, Yin P, Mitchell LA, Parrish DA, Shreeve JM (2016b) N-functionalised nitroxy/azido fused-ring azoles as high-performance energetic materials. J Mater Chem A 4:7430–7436. https://doi.org/10.1039/C6TA02384C
Acknowledgements
Authors would like to express their gratitude to Sh. N Mukherjee, Technology Director for his constant guidance and support. We acknowledge the contribution of Prof. Ganga Ram Chaudhary, Director, Panjab University, Chandigarh for samples analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singla, P., Singh, A., Sahoo, S.C. et al. Synthesis, characterization and thermal decomposition kinetics of energetic copper complex based on 3,5 dinitrobenzoic acid and 1,10 phenanthroline. Chem. Pap. 76, 2111–2124 (2022). https://doi.org/10.1007/s11696-021-01992-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01992-2