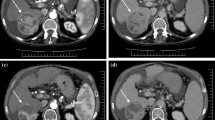
Abstract
Purpose
To investigate the utility of gadoxetate disodium-enhanced hepatic MRI (EOB-MRI) for stereotactic body radiotherapy (SBRT) to treat hepatocellular carcinoma (HCC).
Materials and methods
We divided 30 HCC patients who underwent SBRT into group A (no change in their Child-Pugh score 6 months post-SBRT) and group B (increased score 6 months post-SBRT). EOB-MRI was performed before and 6 months after SBRT. We calculated the liver-spleen contrast (LSC) ratio for each radiation dose area on hepatobiliary phase scans (LSCbefore using images obtained before SBRT and LSCafter using images acquired after SBRT) and the weighted LSC (W-LSC) as: [(mean LSCbefore (0–30 Gy) × liver volume (0–30 Gy) + mean LSCbefore (30 Gy-) × liver volume (30 Gy-))/total liver volume]. Then we compared the W-LSC, percentage of the liver volume exposed to >20 Gy (V20), and mean liver dose in the two groups.
Results
The LSCafter at 48, 40, and 30 Gy to the liver was statistically lower than the unirradiated area of the liver (p < 0.01). Univariate analysis showed that only W-LSC was significantly associated with group B (p = 0.02).
Conclusion
W-LSC was a useful parameter for predicting changes in hepatic function after SBRT.





Similar content being viewed by others
References
Thomas MB, Jaffe D, Choti MM, Belghiti J, Curley S, Fong Y, et al. Hepatocellular carcinoma: consensus recommendations of the National Cancer Institute clinical trials planning meeting. J Clin Oncol. 2010;28(25):3994–4005.
Ryder SD. Guidelines for the diagnosis and treatment of hepatocellular carcinoma (HCC) in adults. Gut. 2003;52(Suppl 3):1–8.
Ikai I, Kudo M, Arii S, Omata M, Kojiro M, Sakamoto M, et al. Report of the 18th follow-up survey of primary liver cancer in Japan. Hepatol Res. 2010;40:1043–59.
Takeda A, Takahashi M, Kunieda E, Takeda T, Sanuki N, Koike Y, et al. Hypofractionated stereotactic radiotherapy with and without transarterial chemoembolization for small hepatocellular carcinoma not eligible for other ablation therapies: preliminary results for efficacy and toxicity. Hepatol Res. 2008;38(1):60–9.
Takeda A, Sanuki N, Eriguchi T, Kobayashi T, Iwabutchi S, Matsunaga K, et al. Stereotactic ablative body radiotherapy for previously untreated solitary hepatocellular carcinoma. J Gastroenterol Hepatol. 2014;29(2):372–9.
Pan CC, Kavanagh BD, Dawson LA, Li XA, Das SK, Miften M, et al. Radiation-associated liver injury. Int J Radiat Oncol Biol Phys. 2010;76(3 Suppl):S94–100.
Takeda A, Oku Y, Sanuki N, Kunieda E, Koike N, Aoki Y, et al. Dose volume histogram analysis of focal liver reaction in follow-up multiphasic CT following stereotactic body radiotherapy for small hepatocellular carcinoma. Radiother Oncol. 2012;104(3):374–8.
Dawson LA, Ten Haken RK, Lawrence TS. Partial irradiation of the liver. Semin Radiat Oncol. 2001;11(3):240–6.
Liang SX, Zhu XD, Xu ZY, Zhu J, Zhao JD, Lu HJ, et al. Radiation-induced liver disease in three-dimensional conformal radiation therapy for primary liver carcinoma: the risk factors and hepatic radiation tolerance. Int J Radiat Oncol Biol Phys. 2006;65(2):426–34.
Huppertz A, Balzer T, Blakeborough A, Breuer J, Giovagnoni A, Heinz-Peer G, et al. Improved detection of focal liver lesions at MR imaging: multicenter comparison of gadoxetic acid-enhanced MR images with intraoperative findings. Radiology. 2004;230(1):266–75.
Ichikawa T, Saito K, Yoshioka N, Tanimoto A, Gokan T, Takehara Y, et al. Detection and characterization of focal liver lesions: a Japanese phase III, multicenter comparison between gadoxetic acid disodium-enhanced magnetic resonance imaging and contrast-enhanced computed tomography predominantly in patients with hepatocellular carcinoma and chronic liver disease. Invest Radiol. 2010;45(3):133–41.
Di Martino M, Marin D, Guerrisi A, Baski M, Galati F, Rossi M, et al. Intraindividual comparison of gadoxetate disodium-enhanced MR imaging and 64-section multidetector CT in the Detection of hepatocellular carcinoma in patients with cirrhosis. Radiology. 2010;256(3):806–16.
Bluemke DA, Sahani D, Amendola M, Balzer T, Breuer J, Brown JJ, et al. Efficacy and safety of MR imaging with liver-specific contrast agent: US multicenter phase III study. Radiology. 2005;237(1):89–98.
Motosugi U, Ichikawa T, Sou H, Sano K, Tominaga L, Kitamura T, et al. Liver parenchymal enhancement of hepatocyte-phase images in Gd-EOB-DTPA-enhanced MR imaging: which biological markers of the liver function affect the enhancement? J Magn Reson Imaging. 2009;30(5):1042–6.
Morita K, Namimoto T, Awai K, Komi M, Hashida M, Tsuji T, et al. Enhancement effects of hepatic dynamic MR imaging at 3.0 T and 1.5 T using gadoxetic acid in a phantom study: comparison with gadopentetate dimeglumine. Magn Reson Med. 2011;66(1):213–8.
Rueckert D, Sonoda LI, Hayes C, Hill DL, Leach MO, Hawkes DJ. Nonrigid registration using free-form deformations: application to breast MR images. IEEE Trans Med Imaging. 1999;18(8):712–21.
Shinagawa Y, Sakamoto K, Fujimitsu R, Shimakura M, Kora S, Takano K, et al. Pseudolesion of the liver on gadoxetate disodium-enhanced MR images obtained after transarterial chemoembolization for hepatocellular carcinoma: clinicoradiologic correlation. Am J Roentgenol. 2012;199(5):1010–7.
Sanuki N, Takeda A, Oku Y, Eriguchi T, Nishimura S, Aoki Y, et al. Threshold doses for focal liver reaction after stereotactic ablative body radiation therapy for small hepatocellular carcinoma depend on liver function: evaluation on magnetic resonance imaging with Gd-EOB-DTPA. Int J Radiat Oncol Biol Phys. 2014;88(2):306–11.
Kimura T, Takahashi S, Kenjo M, Nishibuchi I, Takahashi I, Takeuchi Y, et al. Dynamic computed tomography appearance of tumor response after stereotactic body radiation therapy for hepatocellular carcinoma: how should we evaluate treatment effects? Hepatol Res. 2013;43(7):717–27.
Sanuki N, Takeda A, Mizuno T, Oku Y, Eriguchi T, Iwabuchi S, et al. Tumor response on CT following hypofractionated stereotactic ablative body radiotherapy for small hypervascular hepatocellular carcinoma with cirrhosis. Am J Roentgenol. 2013;201(6):W812–20.
Takeda A, Oku Y, Sanuki N, Kunieda E, Koike N, Aoki Y, et al. Dose volume histogram analysis of focal liver reaction in follow-up multiphasic CT following stereotactic body radiotherapy for small hepatocellular carcinoma. Radiother Oncol. 2012;104(3):374–8.
Sanuki-Fujimoto N, Takeda A, Ohashi T, Kunieda E, Iwabuchi S, Takatsuka K, et al. CT evaluations of focal liver reactions following stereotactic body radiotherapy for small hepatocellular carcinoma with cirrhosis: relationship between imaging appearance and baseline liver function. Br J Radiol. 2010;83(996):1063–71.
Okamoto D, Nishie A, Asayama Y, Tajima T, Ishigami K, Kakihara D, et al. Gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid-enhanced MR finding of radiation-induced hepatic injury: relationship to absorbed dose and time course after irradiation. Magn Reson Imaging. 2014;32(6):660–4.
Kim SH, Lee J, Kim MJ, Jeon YH, Park Y, Choi D, et al. Gadoxetic acid-enhanced MRI versus triple-phase MDCT for the preoperative detection of hepatocellular carcinoma. Am J Roentgenol. 2009;192(6):1675–81.
Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53(3):1020–2.
Matsumata T, Kanematsu T, Yoshida Y, Furuta T, Yanaga K, Sugimachi K. The indocyanine green test enables prediction of postoperative complications after hepatic resection. World J Surg. 1987;11(5):678–81.
Lau H, Man K, Fan ST, Yu WC, Lo CM, Wong J. Evaluation of preoperative hepatic function in patients with hepatocellular carcinoma undergoing hepatectomy. Br J Surg. 1997;84(9):1255–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Kazuo Awai received a research grant from Toshiba Medical Systems Co., Ltd. The other authors declare that they have no conflict of interest.
Ethical statement
None.
About this article
Cite this article
Nakamura, Y., Kimura, T., Higaki, T. et al. Clinical utility of gadoxetate disodium-enhanced hepatic MRI for stereotactic body radiotherapy of hepatocellular carcinoma. Jpn J Radiol 33, 627–635 (2015). https://doi.org/10.1007/s11604-015-0465-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-015-0465-7