Abstract
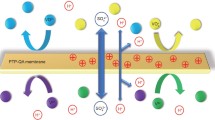
Vanadium redox flow battery (VRFB) is a promising technology for large-scale renewable energy storage. Design of ion-exchange membrane (IEM) with desired properties like low-cost, mechanically chemically stable, low vanadium ion permeability and high proton conductivity is one of the major challenges. Here, we report the design and synthesis of novel poly(tetrafluoroethylene)-zirconium phosphate (PTFE-ZrP) asymmetric IEM using a simple brush coating method. XRD results confirmed the presence of α-ZrP crystalline phase onto the top layer of the membrane. Excellent mechanical strength was observed with burst pressure of 3.22 × 105 N m−2. Oxidative stability of membrane in Fenton’s reagent was much better than Nafion-115. Vanadium ion (V4+) permeability of the membrane was more than three times lower than that of Nafion-115. Single-cell VRFB with PTFE-ZrP membrane showed ∼80% energy efficiency below 30 mA cm−2. Very high columbic efficiency ∼100% of VRFB with PTFE-ZrP membrane confirmed little contamination of electrolyte due to cross-mixing.










Similar content being viewed by others
References
Armaroli N, Balzani V (2011) Towards an electricity-powered world. Energy Environ Sci 4:3193–3222
Wang W, Luo Q, Li B, Wei X, Li L, Yang Z (2013) Recent progress in redox flow battery research and development. Adv Funct Mater 23:970–986
** J, Li Z, Yu L, Yin B, Wang L, Liu L et al (2015) Effect of degree of sulfonation and casting solvent on sulfonated poly (ether ether ketone) membrane for vanadium redox flow battery. J Power Sources 285:195–204
Sum E, Rychcik M, Skyllas-Kazacos M (1985) Investigation of the V (V)/V (IV) system for use in the positive half-cell of a redox battery. J Power Sources 16:85–95
Sum E, Skyllas-Kazacos M (1985) A study of the V (II)/V (III) redox couple for redox flow cell applications. J Power Sources 15:179–190
Parasuraman A, Lim TM, Menictas C, Skyllas-Kazacos M (2013) Review of material research and development for vanadium redox flow battery applications. Electrochim Acta 101:27–40
Yang Z, Zhang J, Kintner-Meyer MCW, Lu X, Choi D, Lemmon JP et al (2011) Electrochemical energy storage for green grid. Chem Rev 111:3577–3613
Ding C, Zhang H, Li X, Liu T, **ng F (2013) Vanadium flow battery for energy storage: prospects and challenges. J Phys Chem Lett 4:1281–1294
Zhang H, Zhang H, Li X, Mai Z, Wei W (2012) Silica modified nanofiltration membranes with improved selectivity for redox flow battery application. Energy Environ Sci 5:6299–6303
Zhang H, Zhang H, Zhang F, Li X, Li Y, Vankelecom I (2013) Advanced charged membranes with highly symmetric spongy structures for vanadium flow battery application. Energy Environ Sci 6:776–781
Mohammadi T, Kazacos MS (1997) Evaluation of the chemical stability of some membranes in vanadium solution. J Appl Electrochem 27:153–160
Yuan Z, Duan Y, Zhang H, Li X, Zhang H, Vankelecom I (2016) Advanced porous membranes with ultra-high selectivity and stability for vanadium flow batteries. Energy Environ Sci 9:441–447
Kim S, Yan J, Schwenzer B, Zhang J, Li L, Liu J et al (2010) Cycling performance and efficiency of sulfonated poly(sulfone) membranes in vanadium redox flow batteries. Electrochem Commun 12:1650–1653
Sun C, Chen J, Zhang H, Han X, Luo Q (2010) Investigations on transfer of water and vanadium ions across nafion membrane in an operating vanadium redox flow battery. J Power Sources 195:890–897
Zeng J, Jiang C, Wang Y, Chen J, Zhu S, Zhao B et al (2008) Studies on polypyrrole modified nafion membrane for vanadium redox flow battery. Electrochem Commun 10:372–375
Teng X, Zhao Y, ** J, Wu Z, Qiu X, Chen L (2009) Nafion/organic silica modified TiO2 composite membrane for vanadium redox flow battery via in situ sol–gel reactions. J Membr Sci 341:149–154
Alberti G, Casciola M (2003) Composite membranes for medium-temperature PEM fuel cells. Annu Rev MaterRes 33:129–154
Chieng SC, Kazacos M, Skyllas-Kazacos M (1992) Modification of Daramic, microporous separator, for redox flow battery applications. J Membr Sci 75:81–91
Mohammadi T, Skyllas-Kazacos M (1995) Preparation of sulfonated composite membrane for vanadium redox flow battery applications. J Membr Sci 107:35–45
Mohammadi T, Skyllas-Kazacos M (1995) Characterisation of novel composite membrane for redox flow battery applications. J Membr Sci 98:77–87
Wei X, Nie Z, Luo Q, Li B, Sprenkle V, Wang W (2013) Polyvinyl chloride/silica Nanoporous composite separator for all-vanadium redox flow battery applications. J Electrochem Soc 160:A1215–A12A8
Dai W, Shen Y, Li Z, Yu L, ** J, Qiu X (2014) SPEEK/graphene oxide nanocomposite membranes with superior cyclability for highly efficient vanadium redox flow battery. J Mater Chem A 2:12423–12432
Wang N, Yu J, Zhou Z, Fang D, Liu S, Liu Y (2013) SPPEK/TPA composite membrane as a separator of vanadium redox flow battery. J Membr Sci 437:114–121
Mai Z, Zhang H, Li X, **ao S, Zhang H (2011) Nafion/polyvinylidene fluoride blend membranes with improved ion selectivity for vanadium redox flow battery application. J Power Sources 196:5737–5741
Seepana MM, Pandey J, Shukla A (2012) Synthesis and characterization of PWA based inorganic ion-exchange membrane. Sep Purif Technol 98:193–198
Miyatake K, Zhou H, Matsuo T, Uchida H, Watanabe M (2004) Proton conductive polyimide electrolytes containing trifluoromethyl groups: synthesis, properties, and DMFC performance. Macromolecules 37:4961–4966
Luo X, Lu Z, ** J, Wu Z, Zhu W, Chen L (2005) Influences of permeation of vanadium ions through PVDF-g-PSSA membranes on performances of vanadium redox flow batteries. J Phys Chem B 109:20310–20314
Chen D, Hickner MA (2013) V5+ degradation of sulfonated Radel membranes for vanadium redox flow batteries. Phys Chem Chem Phys 15:11299–11305
Moosavi K, Setayeshi S, Maragheh MG, Javadahmadi S, Kardan MR, Nosrati S (2009) Synthesis and ion-exchange properties of inorganic ion exchanger zirconium phosphate. J Appl Sci 9:2180–2184
Zhang R, Hu Y, Li B, Chen Z, Fan W (2007) Studies on the preparation and structure of polyacrylamide/α-zirconium phosphate nanocomposites. J Mater Sci 42:5641–5646
Tanaka Y. Chapter 2 Membrane Property Measurements. In: Yoshinobu T, editor. Membr Sci Technol: Elsevier; 2007. p. 17–36.
Jiang B, Wu L, Yu L, Qiu X, ** J (2016) A comparative study of nafion series membranes for vanadium redox flow batteries. J Membr Sci 510:18–26
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seepana, M.M., Pandey, J. & Shukla, A. Design and synthesis of highly stable poly(tetrafluoroethylene)-zirconium phosphate (PTFE-ZrP) ion-exchange membrane for vanadium redox flow battery (VRFB). Ionics 23, 1471–1480 (2017). https://doi.org/10.1007/s11581-016-1967-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-016-1967-8