Abstract
Geosmithia (Ascomycota: Hypocreales) and their associations with bark beetles have long been in the shadow of the more often studied beetle associates: Ophiostomatales and Microascales. The last 20 years of research have demonstrated that Geosmithia species are widespread and abundant in the subcortical habitat and that they show varied degrees of symbiosis with their insect vectors. This article reviews the taxonomic history of the genus, species diversity, host ranges, and biogeography. We also discuss the ecological roles of various Geosmithia species in relation to insect vectors and host trees, phytopathogenicity, and their potential for biotechnology. An extensive review of primary data shows that bark beetle species vector predominantly either Geosmithia or ophiostomatoid fungi, but not both, and that this dichotomy is due to the tree substrate features, especially water content and decay stage, which a given bark beetle vector exploits. In both Geosmithia and the other beetle-associated fungi, coevolution with insect vectors led from phloem inhabiting ancestors to the formation of ambrosia lineages and reciprocal adaptations in the beetles. Lastly, we define knowledge gaps and suggest further research directions.
Graphical Abstract






Similar content being viewed by others
Availability of data and material
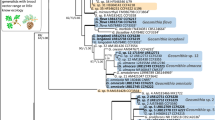
All important data are provided directly in the paper. The primary data used to create Fig. 1 are available from the corresponding author on request.
Code availability
Not applicable.
References
Ameen F, Moslem MA, Hadi S, Al-Sabri A (2014) Biodegradation of cellulosic materials by marine fungi isolated from South Corniche of Jeddah, Saudi Arabia. J Pure Appl Microbiol 8:3617–3626
Barras SJ (1972) Improved White’s solution for surface sterilization of Dendroctonus frontalis. J Econ Entomol 65:1504. https://doi.org/10.1093/jee/65.5.1504
Bastian F, Alabouvette C, Saiz-Jimenez C (2009) The impact of arthropods on fungal community structure in Lascaux Cave. J Appl Microbiol 106:1456–1462. https://doi.org/10.1111/j.1365-2672.2008.04121.x
Beaver RA (1989) Insect-fungus relationships in the bark and ambrosia beetles. In: Wilding N, Collins NM, Hammond PM, Webber JF (eds) Insect-Fungus Interactions. Academic Press, London, pp 121–143
Belhoucine L, Bouhraoua RT, Meijer M, Houbraken J, Harrak MJ, Samson RA, Equihua-Martinez A, Pujade-Villar J (2011) Mycobiota associated with Platypus cylindrus (Coleoptera: Curculionidae, Platypodidae) in cork oak stands of North West Algeria, Africa. African J Microbiol Res 5:4411–4423. https://doi.org/10.5897/Ajmr11.614
Benvenuti C, Strangi A, Iovinella I, Barzanti GP, Simoni S, Vitale S, Luongo L, Francardi V, Roversi P (2021) Xylosandrus compactus and Liparthrum colchicum (Coleoptera Scolytinae) in Tuscany: a preliminary screening of associated Fungi. J Zool 104:139–146. https://doi.org/10.19263/REDIA-104.21.14
Bettini PP, Frascella A, Comparini C, Carresi L, Pepori AL, Pazzagli L, Cappugi G, Scala F, Scala A (2012) Identification and characterization of GEO1, a new class II hydrophobin from Geosmithia spp. Canad J Microbiol 58:965–972. https://doi.org/10.1139/w2012-069
Bettini PP, Frascella A, Kolařík M, Comparini C, Pepori AL, Santini A, Scala F, Scala A (2014) Widespread horizontal transfer of the cerato-ulmin gene between Ophiostoma novo-ulmi and Geosmithia species. Fungal Biol 118:663–674. https://doi.org/10.1016/j.funbio.2014.04.007
Blood B, Klingeman W, Paschen M, Hadžiabdić Đ, Couture J, Ginzel M (2018) Behavioral responses of Pityophthorus juglandis (Coleoptera: Curculionidae: Scolytinae) to volatiles of black walnut and Geosmithia morbida (Ascomycota: Hypocreales: Bionectriaceae), the causal agent of thousand cankers disease. Environ Entomol 47:412–421. https://doi.org/10.1093/ee/nvx194
Bright DE, Stark RW (1973) The bark and ambrosia beetles of California (Coleoptera: Scolytidae and Platypodidae). Bulletin of the California Insect Survey 16:1–167
Buckingham JB (1996) Dictionary of Organic Compound (6th ed), vol 1. Chapman and Hall/CRC Press, London, pp 5517
Cala A, Masi M, Cimmino A, Molinillo JM, Macias FA, Evidente A (2018) (+)-epi-epoformin, a phytotoxic fungal cyclohexenepoxide: structure activity relationships. Molecules 23:1529. https://doi.org/10.3390/molecules23071529
Chahal K, Gazis R, Klingeman W, Hadziabdic D, Lambdin P, Grant J, Windham M (2019) Assessment of alternative candidate subcortical insect vectors from walnut crowns in habitats quarantined for thousand cankers disease. Environ Entomol 48:882–893. https://doi.org/10.1093/ee/nvz064
Čížková D, Šrůtka P, Kolařík M, Kubátová A, Pažoutová S (2005) Assessing the pathogenic effect of Fusarium, Geosmithia and Ophiostoma fungi from broad-leaved trees. Folia Microbiol 50:59–62. https://doi.org/10.1007/BF02931294
Crous P, Luangsa-Ard J, Wingfield M, Carnegie A, Hernández-Restrepo M, Lombard L, Roux J, Barreto R, Baseia I, Cano-Lira J (2018) Fungal planet description sheets: 785–867. Persoonia 41:238. https://doi.org/10.3767/persoonia.2018.41.12
Crous PW, Boers J, Holdom D, Steinrucken T, Tan Y, Vitelli J, Shivas R, Barrett M, Boxshall A-G, Broadbridge J (2022) Fungal planet description sheets: 1383–1435. Persoonia 48:261–371. https://doi.org/10.3767/persoonia.2022.48.08
Dal Maso E, Linaldeddu BT, Fanchin G, Faccoli M, Montecchio L (2019) The potential for pesticide trunk injections for control of thousand cankers disease of walnut. Phytopathologia Mediterranea 58:73–79. https://doi.org/10.14601/Phytopathol_Mediterr-23598
Daniels DA, Nix KA, Wadl PA, Vito LM, Wiggins GJ, Windham MT, Ownley BH, Lambdin PL, Grant JF, Merten P (2016) Thousand cankers disease complex: a forest health issue that threatens Juglans species across the US. Forests 7:260. https://doi.org/10.3390/f7110260
Davydenko K, Vasaitis R, Meshokova V, Menkis A (2014) Fungi associated with the red-haired bark beetle, Hylurgus ligniperda (Coleoptera: Curculionidae) in the forest-steppe zone in eastern Ukraine. Eur J Entomol 111:561–565. https://doi.org/10.14411/eje.2014.070
Davydenko K, Vasaitis R, Menkis A (2017) Fungi associated with Ips acuminatus (Coleoptera: Curculionidae) in Ukraine with a special emphasis on pathogenicity of ophiostomatoid species. Eur J Entomol 114:77–85. https://doi.org/10.14411/eje.2017.011
Davydenko K, Vasaitis R, Elfstrand M, Baturkin D, Meshkova V, Menkis A (2021) Fungal communities vectored by Ips sexdentatus in declining Pinus sylvestris in Ukraine: focus on occurrence and pathogenicity of ophiostomatoid species. Insects 12:1119. https://doi.org/10.3390/insects12121119
Deka D, Jha DK (2018) Optimization of culture parameters for improved production of bioactive metabolite by endophytic Geosmithia pallida (KU693285) isolated from Brucea mollis Wall ex. Kurz, an endangered medicinal plant. J Pure Appl Microbiol 12:1205–1213. https://doi.org/10.22207/JPAM.12.3.21
Dighton J, Walsh E, Groben G, Zhang N (2021) Influence of southern pine beetle on fungal communities of wood and bark decomposition of coarse woody debris in the New Jersey pine barrens. Forestry Res 1:17. https://doi.org/10.48130/FR-2021-0017
Doehlemann G, Ökmen B, Zhu W, Sharon A (2017) Plant pathogenic fungi. Microbiology spectrum 5:5.1. 14. https://doi.org/10.1128/microbiolspec.FUNK-0023-2016
Dohet L, Grégoire J-C, Berasategui A, Kaltenpoth M, Biedermann PHW (2016) Bacterial and fungal symbionts of parasitic Dendroctonus bark beetles. FEMS Microbiol Ecol 92:fiw129. https://doi.org/10.1093/femsec/fiw129
Dori-Bachash M, Avrahami-Moyal L, Protasov A, Mendel Z, Freeman S (2015) The occurrence and pathogenicity of Geosmithia spp. and common blue-stain fungi associated with pine bark beetles in planted forests in Israel. Eur J Plant Pathol 143:627–639. https://doi.org/10.1007/s10658-015-0713-9
Flieger M, Stodulková E, Kolařík M, Šulc M, Kuzma M (2009) Submerzní kmeny Geosmithia sp. MK385B (CCM 8366) a Geosmithia sp. MK583B (CCM 8365), smes antrachinonových barviv jimi produkovaná a zpusob její produkce [Submerged strains of Geosmithia sp. MK385B (CCM 8366) and Geosmithia sp. MK583B (CCM 8365), a mixture of anthraquinone dyes produced and the method of its production]. Czech Republic Patent, CZ300810B6
Foit J (2010) Distribution of early-arriving saproxylic beetles on standing dead Scots pine trees. Agric for Entomol 12:133–141. https://doi.org/10.1111/j.1461-9563.2009.00461.x
Frascella A, Bettini PP, Kolařík M, Comparini C, Pazzagli L, Luti S, Scala F, Scala A (2014) Interspecific variability of class II hydrophobin GEO1 in the genus Geosmithia. Fungal Biol 118:862–871. https://doi.org/10.1016/j.funbio.2014.07.005
Ganapaty S, Thomas PS, Fotso S, Laatsch H (2004) Antitermitic quinones from Diospyros sylvatica. Phytochemistry 65:1265–1271. https://doi.org/10.1002/chin.200446205
Gao L, Li Y, Wang ZX, Zhao J, Hulcr J, Wang JG, Li YZ, Ju RT (2021) Biology and associated fungi of an emerging bark beetle pest, the sweetgum inscriber Acanthotomicus suncei (Coleoptera: Curculionidae). J Appl Entomol 145:508–517. https://doi.org/10.1111/jen.12861
Gazis R, Poplawski L, Klingeman W, Boggess SL, Trigiano RN, Graves AD, Seybold SJ, Hadziabdic D (2018) Mycobiota associated with insect galleries in walnut with thousand cankers disease reveals a potential natural enemy against Geosmithia morbida. Fungal Biol 122:241–253. https://doi.org/10.1016/j.funbio.2018.01.005
Giordano A, Di Landro F, De Carolis E, Criscuolo M, Dragonetti G, Fianchi L, Pagano L (2021) Disseminated Geosmithia argillacea infection in a patient with Ph-positive acute lymphoblastic leukemia. Case report and literature review. J Fungi 7:778. https://doi.org/10.3390/jof7090778
Giraud S, Favennec L, Bougnoux M-E, Bouchara J-P (2013) Rasamsonia argillacea species complex: taxonomy, pathogenesis and clinical relevance. Future Microbiol 8:967–978. https://doi.org/10.2217/fmb.13.63
Gonzalez-Escobedo R, Briones-Roblero CI, López MF, Rivera-Orduña FN, Zúñiga G (2019) Changes in the microbial community of Pinus arizonica saplings after being colonized by the bark beetle Dendroctonus rhizophagus (Curculionidae: Scolytinae). Microb Ecol 78:102–112. https://doi.org/10.1007/s00248-018-1274-1
Gregory TR (2001) Coincidence, coevolution, or causation? DNA content, cell size, and the C-value enigma. Biol Rev 76:65e101. https://doi.org/10.1017/s1464793100005595
Hadj Taieb K, Gharsallah H, Ksentini I, Schuster C, Fernández-Bravo M, Garrido-Jurado I, Quesada-Moraga E, Leclerque A, Triki M, Ksantini M (2019) Phytopathogenic and antagonistic potentialities of fungi associated with pistachio bark beetle, Chaetoptelius vestitus (Coleoptera, Curculionidae), infesting pistachio (Pistacia vera) in Tunisia. J Appl Microbiol 126:1821–1834. https://doi.org/10.1111/jam.14272
Hadziabdic D, Vito LM, Windham MT, Pscheidt JW, Trigiano RN, Kolarik M (2014a) Genetic differentiation and spatial structure of Geosmithia morbida, the causal agent of thousand cankers disease in black walnut (Juglans nigra). Curr Genet 60:75–87. https://doi.org/10.1007/s00294-013-0414-x
Hadziabdic D, Windham M, Baird RE, Vito L, Cheng Q, Grant J, Lambdin P, Wiggins G, Windham A, Merten P (2014b) First report of Geosmithia morbida in North Carolina: the pathogen involved in thousand cankers disease of black walnut. Plant Dis 98:992. https://doi.org/10.1094/PDIS-06-13-0630-PDN
Hanzi M, Cochard B, Chablais R, Crovadore J, Lefort F (2016) First report of Geosmithia langdonii and Geosmithia spp. isolated from a decaying elm (Ulmus minor) in Geneva, Switzerland. Folia Forestalia Polonica Series A Forestry 58:96–102. https://doi.org/10.1515/ffp-2016-0011
Hefty AR, Aukema BH, Venette RC, Coggeshall MV, McKenna JR, Seybold SJ (2018) Reproduction and potential range expansion of walnut twig beetle across the Juglandaceae. Biol Invasions 20:2141–2155. https://doi.org/10.1007/s10530-018-1692-5
Hilker M, Köpf A (1994) Evaluation of the palatability of chrysomelid larvae containing anthraquinones to birds. Oecologia 100:421–429. https://doi.org/10.1007/BF00317864
Hofstetter RW, Dinkins-Bookwalter J, Davis TS, Klepzig KD (2015) Symbiotic associations of bark beetles. Bark Beetles. Elsevier, pp 209–245
Houbraken J, Spierenburg H, Frisvad JC (2012) Rasamsonia, a new genus comprising thermotolerant and thermophilic Talaromyces and Geosmithia species. Antonie Van Leeuwenhoek 101:403–421. https://doi.org/10.1007/s10482-011-9647-1
Hu X, Li M, Chen H (2015) Community structure of gut fungi during different developmental stages of the Chinese white pine beetle (Dendroctonus armandi). Sci Rep 5:8411. https://doi.org/10.1038/srep08411
Huang Y-T, Kolarik M, Kasson M, Hulcr J (2017) Two new Geosmithia species in G. pallida species complex from bark beetles in eastern USA. Mycologia 109:790–803. https://doi.org/10.1080/00275514.2017.1410422
Huang YT, Skelton J, Johnson AJ, Kolařík M, Hulcr J (2019) Geosmithia species in southeastern USA and their affinity to beetle vectors and tree hosts. Fungal Ecol 39:168–183. https://doi.org/10.1016/j.funeco.2019.02.005
Iwamoto S, Tokumasu S, Suyama Y, Kakishima M (2002) Molecular phylogeny of four selected species of the strictly anamorphic genus Thysanophora using nuclear ribosomal DNA sequences. Mycoscience 43:169–180. https://doi.org/10.1007/S102670200024
Jagadeeswaran G, Veale L, Mort AJ (2021) Do lytic polysaccharide monooxygenases aid in plant pathogenesis and herbivory? Trends Plant Sci 26:142–155. https://doi.org/10.1016/j.tplants.2020.09.013
Jankowiak R (2006a) Fungi associated with Tomicus piniperda in Poland and assessment of their virulence using Scots pine seedlings. Ann for Sci 63:801–808. https://doi.org/10.1051/forest:2006063
Jankowiak R (2006b) Mycobiota associated with Hylurgops palliatus (Gyll.) on Pinus sylvestris L. in Poland. Acta Soc Bot Pol 75:333–338. https://doi.org/10.5586/asbp.2006.040
Jankowiak R (2008) Fungi associated with Tomicus minor on Pinus sylvestris in Poland and their succession into the sapwood of beetle-infested windblown trees. Can J for Res 38:2579–2588. https://doi.org/10.1139/X08-101
Jankowiak R, Bilański P (2007) Fungal flora associated with Tomicus piniperda L. in an area close to a timber yard in southern Poland. J Appl Entomol 131:579–584. https://doi.org/10.1111/j.1439-0418.2007.01194.x
Jankowiak R, Bilanski P (2018) Geosmithia species associated with fir-infesting beetles in Poland. Acta Mycol 53:1115. https://doi.org/10.5586/am.1115
Jankowiak R, Hilszczański J (2005) Ophiostomatoid fungi associated with Ips typographus (L.) on Picea abies [(L.) H. Karst.] and Pinus sylvestris L. in north-eastern Poland. Acta Soc Bot Pol 74:345–350. https://doi.org/10.5586/asbp.2005.043
Jankowiak R, Kolarik M (2010) Fungi associated with the fir bark beetle Cryphalus piceae in Poland. For Pathol 40:133–144. https://doi.org/10.1111/j.1439-0329.2009.00620.x
Jankowiak R, Rossa R (2007) Filamentous fungi associated with Monochamus galloprovincialis and Acanthocinus aedilis (Coleoptera: Cerambycidae) in Scots pine. Polish Bot J 52:140–143
Jankowiak R, Rossa R (2008) Associations between Pityogenes bidentatus and fungi in young managed Scots pine stands in Poland. For Pathol 38:169–177. https://doi.org/10.1111/j.1439-0329.2007.00535.x
Jankowiak R, Kolarik M, Bilanski P (2014) Association of Geosmithia fungi (Ascomycota: Hypocreales) with pine- and spruce-infesting bark beetles in Poland. Fungal Ecol 11:71–79. https://doi.org/10.1016/j.funeco.2014.04.002
Juan Alfredo H-G, Gerardo C-R, Nallely Guadalupe A-O, Lourdes V-T, César H-R, Francisco A-T (2020) Phylogenetic position of Geosmithia spp. (Hypocreales) living in Juniperus spp. forests (Cupressaceae) with bark beetles of Phloeosinus spp. (Scolytinae) from the Northeast of Mexico. Forests 11:1142. https://doi.org/10.3390/f11111142
Juzwik J, Yang A, Heller S, Moore M, Chen Z, White M, Wantuch H, Ginzel M, Mack R (2021) Vacuum steam treatment effectiveness for eradication of the Thousand Cankers Disease vector and pathogen in logs from diseased walnut trees. J Econ Entomol 114:100–111. https://doi.org/10.1093/jee/toaa267
Kim YJ, Duraisamy K, Jeong M-H, Park S-Y, Kim S, Lee Y, Nguyen VT, Yu NH, Park AR, Kim J-C (2021) Nematicidal activity of grammicin biosynthesis pathway intermediates in Xylaria grammica KCTC 13121BP against Meloidogyne incognita. Molecules 26:4675. https://doi.org/10.3390/molecules26154675
Kirschner R (1998) Diversität mit Borkenkäfern assoziierter filamentöser Mikropilze. Eberhard-Karls Universität, Tübingen
Kirschner R, Sampaio JP, Gadanho M, Weiss M, Oberwinkler F (2001) Cuniculitrema polymorpha (Tremellales, gen. nov. and sp. nov.), a heterobasidiomycete vectored by bark beetles, which is the teleomorph of Sterigmatosporidium polymorphum. Antonie Van Leeuwenhoek 80:149–161. https://doi.org/10.1023/A:1012275204498
Kirschner R (2001) Diversity of filamentous fungi in bark beetle galleries in Central Europe. In: Misra JK, Horn BW (eds) In Trichomycetes and other fungal groups: Professor Robert W. Lichtwardt commemoration volume Science Publishers, Enfield (NH), USA, pp 175–196
Kolařík M, Jankowiak R (2013) Vector affinity and diversity of Geosmithia fungi living on subcortical insects inhabiting Pinaceae species in central and Northeastern Europe. Microb Ecol 66:682–700. https://doi.org/10.1007/s00248-013-0228-x
Kolařík M, Kirkendall LR (2010) Evidence for a new lineage of primary ambrosia fungi in Geosmithia Pitt (Ascomycota: Hypocreales). Fungal Biol 114:676–689. https://doi.org/10.1016/j.funbio.2010.06.005
Kolařík M, Vohník M (2017) When the ribosomal DNA does not tell the truth: the case of the taxonomic position of Kurtia argillacea, an ericoid mycorrhizal fungus residing among Hymenochaetales. Fungal Biol 122:1–18. https://doi.org/10.1016/j.funbio.2017.09.006
Kolařík M, Kubátová A, Pažoutová S, Šrůtka P (2004) Morphological and molecular characterisation of Geosmithia putterillii, G. pallida comb. nov. and G. flava sp. nov., associated with subcorticolous insects. Mycol Res 108:1053–1069. https://doi.org/10.1017/S0953756204000796
Kolařík M, Kubátová A, Čepička I, Pažoutová S, Šrůtka P (2005) A complex of three new white-spored, sympatric, and host range limited Geosmithia species. Mycol Res 109:1323–1336. https://doi.org/10.1017/S0953756205003965
Kolařík M, Sláviková E, Pažoutová S (2006) The taxonomic and ecological characterization of the clinically important heterobasiodiomycete Fugomyces cyanescens and its association with bark beetles. Czech Mycol 58:81–98. https://doi.org/10.33585/cmy.58106
Kolařík M, Kostovčík M, Pažoutová S (2007) Host range and diversity of the genus Geosmithia (Ascomycota: Hypocreales) living in association with bark beetles in the Mediterranean area. Mycol Res 111:1298–1310. https://doi.org/10.1016/j.mycres.2007.06.010
Kolařík M, Kubátová A, Hulcr J, Pažoutová S (2008) Geosmithia fungi are highly diverse and consistent bark beetle associates: evidence from their community structure in temperate Europe. Microb Ecol 56:198–199. https://doi.org/10.1007/s00248-008-9371-1
Kolařík M, Freeland E, Utley C, Tisserat N (2011) Geosmithia morbida sp. nov., a new phytopathogenic species living in symbiosis with the walnut twig beetle (Pityophthorus juglandis) on Juglans in USA. Mycologia 103:325–332. https://doi.org/10.3852/10-124
Kolařík M, Hulcr J, Kirkendall LR (2015) New species of Geosmithia and Graphium associated with ambrosia beetles in Costa Rica. Czech Mycol 67:29–35. https://doi.org/10.33585/cmy.67103
Kolařík M, Hulcr J, Tisserat N, De Beer W, Kostovčík M, Kolaříková Z, Seybold SJ, Rizzo DM (2017) Geosmithia associated with bark beetles and woodborers in the western USA: taxonomic diversity and vector specificity. Mycologia 109:185–199. https://doi.org/10.1080/00275514.2017.1303861
Kolařík M, Wei I, Hsieh S-Y, Piepenbring M, Kirschner R (2021) Nucleotide composition bias of rDNA sequences as a source of phylogenetic artifacts in Basidiomycota — a case of a new lineage of a uredinicolous Ramularia-like anamorph with affinities to Ustilaginomycotina. Mycol Prog 20:1553–1571. https://doi.org/10.1007/s11557-021-01749-x
Kubátová A, Kolařík M, Prášil K, Novotný D (2004) Bark beetles and their galleries: well-known niche for little known fungi, case of Geosmithia. Czech Mycol 55:1–18. https://doi.org/10.33585/cmy.56101
Kubátová A, Novotný D, Prášil K Bělokaz dubový jako přenašeč mikroskopických hub [Oak bark beetle as a vector of microscopic fungi]. In: Jankovský L, Krejčíř R, Antonín V (eds) Houby a les [Fungi and forest], Brno, 5.-6. 6. 1999 1999. pp 235–236
Kula E, Zabecki W (2000) Vliv některých růstových a porostních faktorů na obsazení smrku kambioxylofágy (Effect of selected growth and vegetations factors on distribution of cambioxylophages on spruce). Lesnická Práce 6:257–260
Kumar CG, Mongolla P, Pombala S, Bandi S, Babu K, Ramakrishna K (2017) Biological evaluation of 3-hydroxybenzyl alcohol, an extrolite produced by Aspergillus nidulans strain KZR-132. J Appl Microbiol 122:1518–1528. https://doi.org/10.1111/jam.13450
Labuda R, Tancinová D (2006) Fungi recovered from Slovakian poultry feed mixtures and their toxinogenity. Ann Agric Environ Med 13:193–200
Lee S, Kim JJ, Breuil C (2006) Diversity of fungi associated with the mountain pine beetle, Dendroctonus ponderosae and infested lodgepole pines in British Columbia. Fungal Diversity 22:91–105. https://doi.org/10.1007/0-387-26336-5_666
Li Y, Bateman C, Skelton J, Wang B, Black A, Huang Y-T, Gonzalez A, Jusino MA, Nolen ZJ, Freeman S, Mendel Z, Kolařík M, Knížek M, Park J-H, Sittichaya W, Pham T-H, Ito S-i, Torii M, Gao L, Johnson AJ, Lu M, Sun J, Zhang Z, Adams DC, Hulcr J (2022) Preinvasion assessment of exotic bark beetle-vectored fungi to detect tree-killing pathogens. Phytopathology 112:261–270. https://doi.org/10.1094/phyto-01-21-0041-r
Liang W-L, Le X, Li H-J, Yang X-L, Chen J-X, Xu J, Liu H-L, Wang L-Y, Wang K-T, Hu K-C (2014) Exploring the chemodiversity and biological activities of the secondary metabolites from the marine fungus Neosartorya pseudofischeri. Mar Drugs 12:5657–5676. https://doi.org/10.3390/md12115657
Lim YW, Kim JJ, Lu M, Breuil C (2005) Determining fungal diversity on Dendroctonus ponderosae and Ips pini affecting lodgepole pine using cultural and molecular methods. Fungal Diversity 19:79–94
Lin YT, Shih HH, Huang YT, Lin CS, Chen CY (2016) Two species of beetle-associated Geosmithia in Taiwan. Fungal Sci 31:29–36
Linnakoski R, de Beer ZW, Rousi M, Niemelä P, Pappinen A, Wingfield MJ (2008) Fungi, including Ophiostoma karelicum sp. nov., associated with Scolytus ratzeburgi infesting birch in Finland and Russia. Mycol Res 112:1475–1488. https://doi.org/10.1016/j.mycres.2008.06.007
Lynch SC, Wang DH, Mayorquin JS, Rugman-Jones P, Stouthamer R, Eskalen A (2014) First report of Geosmithia pallida causing foamy bark canker, a new disease on coast live oak (Quercus agrifolia) in association with Pseudopityophthorus pubipennis (western oak bark beetle, Coleoptera: Curculionidae: Scolytinae) in California. Plant Dis 98:1276–1276. https://doi.org/10.1094/pdis-03-14-0273-pdn
Machingambi NM, Roux J, Dreyer LL, Roets F (2014) Bark beetles (Curculionidae: Scolytinae), their phoretic mites (Acari) and associated Geosmithia species (Ascomycota: Hypocreales) from Virgilia trees in South Africa. Fungal Biol 118:472–483. https://doi.org/10.1016/j.funbio.2014.03.006
Malak L, Bishay D, Abdel-Baky A, Moharram A, Cutler SJ, Ross SA (2013a) New secondary metabolites from Geosmithia lavendula. Planta Med 79:P51. https://doi.org/10.1055/s-0033-1336493
Malak LG, Bishay DW, Abdel-baky AM, Moharram AM, Cutler SJ, Ross SA (2013) Bioactive secondary metabolites from Geosmithia langdonii. Planta Medica 79:PL7. https://doi.org/10.1055/s-0033-1348648
Malak LG, Bishay DW, Abdel-Baky AM, Moharram AM, Cutler SJ, Ross SA (2013) New anthraquinone derivatives from Geosmithia lavendula. Nat Prod Commun 8:1934578X1300800215. https://doi.org/10.1177/1934578X1300800215
Malak LG, Ibrahim MA, Bishay DW, Abdel-Baky AM, Moharram AM, Tekwani B, Cutler SJ, Ross SA (2014) Antileishmanial metabolites from Geosmithia langdonii. J Nat Prod 77:1987–1991. https://doi.org/10.1021/np5000473
Malak LG, Ibrahim MA, Moharram AM, Pandey P, Tekwani B, Doerksen RJ, Ferreira D, Ross SA (2018) Antileishmanial carbasugars from Geosmithia langdonii. J Nat Prod 81:2222–2227. https://doi.org/10.1021/acs.jnatprod.8b00473
Marchioro M, Faccoli M (2022) Dispersal and colonization risk of the Walnut Twig Beetle, Pityophthorus juglandis, in southern Europe. J Pest Sci 95:303–313. https://doi.org/10.1007/s10340-021-01372-5
McPherson BA, Erbilgin N, Bonello P, Wood DL (2013) Fungal species assemblages associated with Phytophthora ramorum-infected coast live oaks following bark and ambrosia beetle colonization in northern California. For Ecol Manage 291:30–42. https://doi.org/10.1016/j.foreco.2012.11.010
Meshram V, Sharma G, Maymon M, Protasov A, Mendel Z, Freeman S (2022) Symbiosis and pathogenicity of Geosmithia and Talaromyces spp. associated with the cypress bark beetles Phloeosinus spp. and their parasitoids. Environ Microbiol 24:3369–3389. https://doi.org/10.1111/1462-2920.16016
Montecchio L, Faccoli M (2014) First record of thousand cankers disease Geosmithia morbida and walnut twig beetle Pityophthorus juglandis on Juglans nigra in Europe. Plant Dis 98:696–696. https://doi.org/10.1094/PDIS-10-13-1027-PDN
Montecchio L, Fanchin G, Berton V, Scattolin L (2015) Vegetative incompatibility and potential involvement of a mycovirus in the Italian population of Geosmithia morbida. Phytopathologia Mediterranea 54:465–476. https://doi.org/10.14601/Phytopathol_Mediterr-16376
Morales-Rodríguez C, Sferrazza I, Aleandri MP, Dalla Valle M, Speranza S, Contarini M, Vannini A (2021) The fungal community associated with the ambrosia beetle Xylosandrus compactus invading the Mediterranean maquis in central Italy reveals high biodiversity and suggests environmental acquisitions. Fungal Biol 125:12–24. https://doi.org/10.1016/j.funbio.2020.09.008
Moricca S, Bracalini M, Benigno A, Ghelardini L, Furtado EL, Marino CL, Panzavolta T (2020) Observations on the non-native Thousand Cankers Disease of walnut in Europe’s southernmost outbreak. Global Ecology and Conservation e01159. https://doi.org/10.1016/j.gecco.2020.e01159
Muñoz-Adalia EJ, Sanz-Ros AV, Flores-Pacheco JA, Hantula J, Diez JJ, Vainio EJ, Fernández M (2017) Sydowia polyspora dominates fungal communities carried by two Tomicus species in pine plantations threatened by Fusarium circinatum. Forests 8:127. https://doi.org/10.3390/f8040127
O’Gorman CM, Fuller HT, Dyer PS (2008) Discovery of a sexual cycle in the opportunistic fungal pathogen Aspergillus fumigatus. Nature 457:471–474. https://doi.org/10.1038/nature07528
Ogawa H, Sugiyama J (2000) Evolutionary relationships of the cleistothecial genera with Penicillium, Geosmithia, Merimbla and Sarophorum anamorphs as inferred from 18S rDNA sequence divergence. In: Samson RA, Pitt JI (eds) Integration of modern taxonomic methods for Penicillium and Aspergillus classification. Harwood, Amsterdam, pp 149–161
Ogawa H, Yoshimura A, Sugiyama J (1997) Polyphyletic origins of species of the anamorphic genus Geosmithia and the relationships of the cleistothecial genera: evidence from 18S, 5S and 28S rDNA sequence analyses. Mycologia 89:756–771. https://doi.org/10.2307/3761132
Pepori AL, Kolařík M, Bettini PP, Vettraino AM, Santini A (2015) Morphological and molecular characterisation of Geosmithia species on European elms. Fungal Biol 119:1063–1074. https://doi.org/10.1016/j.funbio.2015.08.003
Pepori AL, Bettini PP, Comparini C, Sarrocco S, Bonini A, Frascella A, Ghelardini L, Scala A, Vannacci G, Santini A (2018) Geosmithia-Ophiostoma: a new fungus-fungus association. Microb Ecol 75:632–646. https://doi.org/10.1007/s00248-017-1062-3
Persson Y, Vasaitis R, Långström B, Öhrn P, Ihrmark K, Stenlid J (2009) Fungi vectored by the bark beetle Ips typographus following hibernation under the bark of standing trees and in the forest litter. Microb Ecol 58:651–659. https://doi.org/10.1007/s00248-009-9520-1
Peterson SW (2000) Phylogenetic analysis of Penicillium species based on ITS and LSU-rDNA nucleotide sequences. In: Samson RA, Pitt JI (eds) Integration of modern taxonomic methods for Penicillium and Aspergillus classification. Harwood Academic Publishers, Reading, pp 163–178
Pietsch GM, Gazis R, Klingeman WE, Huff ML, Staton ME, Kolařík M, Hadziabdic D (2022) Characterization and microsatellite marker development for a common bark and ambrosia beetle associate, Geosmithia obscura. MicrobiologyOpen 11:e1286. https://doi.org/10.1002/mbo3.1286
Pitt JI (1979) The genus Penicillium and its teleomorphic states Eupenicillium and Talaromyces. Academic Press, London
Pitt JI, Hocking AD (1985) Interfaces among genera related to Aspergillus and Penicillium. Mycologia 77:810–824. https://doi.org/10.2307/3793288
Pitt JI, Hocking AD (2009) Fungi and food spoilage, 3rd edn. Springer, Dordrecht, The Netherlands
Pitt JI, Samson RA (1993) Species names in current use in the Trichocomaceae (Fungi: Eurotiales). Regnum Vegetabile 128:13–57
Pitt JI, Samson RA, Frisvad JC (2000) List of accepted species and their synonyms in the family Trichocomaceae. In: Samson RA, Pitt JI (eds) Integration of modern taxonomic methods for Penicillium and Aspergillus classification. Harwood Academic Publishers, pp 9–49
Plishka MJR, Tsuneda A, Currah RS (2009) Morphology and development of Nigrosabulum globosum, a cleistothecial coprophile in the Bionectriaceae (Hypocreales). Mycol Res 113:815–821. https://doi.org/10.1016/j.mycres.2009.02.005
Poche RM (1998) Development of a new bird repellent, Flight Control. Proceedings of the vertebrate pest conference vol 18. pp 337–334
Postner M (1974) Scolytidae (= Ipidae), Borkenkäfer. In: Schwenke W (ed) Die Forstschädlinge Europas, II Käfer, vol 2. Verlag Paul Parey. Hamburg, Berlin, pp 334–482
Proctor RH, McCormick SP, Kim H-S, Cardoza RE, Stanley AM, Lindo L, Kelly A, Brown DW, Lee T, Vaughan MM, Alexander NJ, Busman M, Gutiérrez S (2018) Evolution of structural diversity of trichothecenes, a family of toxins produced by plant pathogenic and entomopathogenic fungi. PLoS Pathog 14:e1006946. https://doi.org/10.1371/journal.ppat.1006946
Ramirez C (1982) Manual and atlas of the Penicillia. Elsevier Biomedical Press, Amsterdam etc
Raper KB, Thom VC (1949) A manual of the Penicillia. The Williams & Wilkins Comp, Baltimore
Ratnaweera P, de Silva ED, Wijesundera RL, Andersen RJ (2016) Antimicrobial constituents of Hypocrea virens, an endophyte of the mangrove-associate plant Premna serratifolia L. J Natl Sci Found Sri Lanka 44:43–51. https://doi.org/10.4038/jnsfsr.v44i1.7980
Rossman AY, Samuels GJ, Rogerson CT, Lowen R (1999) Genera of Bionectriaceae, Hypocreaceae and Nectriaceae (Hypocreales, Ascomycetes). Stud Mycol 42:1–248. https://doi.org/10.5598/imafungus.2013.04.01.05
Sakalidis ML, Hardy GES, Burgess TI (2011) Endophytes as potential pathogens of the baobab species Adansonia gregorii: a focus on the Botryosphaeriaceae. Fungal Ecol 4:1–14. https://doi.org/10.1016/j.funeco.2010.06.001
Salvatore MM, Alves A, Andolfi A (2020) Secondary metabolites of Lasiodiplodia theobromae: distribution, chemical diversity, bioactivity, and implications of their occurrence. Toxins 12:457. https://doi.org/10.3390/toxins12070457
Saurat C, Mouttet R, Jeandel C, Prost J, Tellez D, Ioos R (2023) First report of thousand cankers disease caused by the fungus Geosmithia morbida and its vector Pityophthorus juglandis on Juglans regia in France. New Disease Reports 47:e12151. https://doi.org/10.1002/ndr2.12151
Scala A, Comparini C, Tegli S, Scala F (2007) A non-Ophiostoma fungus expresses the gene encoding the hydrophobin cerato-ulmin. J Plant Pathol 89:233–240. https://doi.org/10.1007/s00248-017-1062-3
Schroers HJ, Geldenhuis MM, Wingfield MJ, Schoeman MH, Yen YF, Shen WC, Wingfield BD (2005) Classification of the guava wilt fungus Myxosporium psidii, the palm pathogen Gliocladium vermoesenii and the persimmon wilt fungus Acremonium diospyri in Nalanthamala. Mycologia 97:375–395. https://doi.org/10.3852/mycologia.97.2.375
Schuelke TA, Wu G, Westbrook A, Woeste K, Plachetzki DC, Broders K, MacManes MD (2017) Comparative genomics of pathogenic and nonpathogenic beetle-vectored fungi in the genus Geosmithia. Genome Biol Evol 9:3312–3327. https://doi.org/10.1093/gbe/evx242
Seabright KW, Myers SW, Fraedrich SW, Mayfield AE III, Warden ML, Taylor A (2019) Methyl bromide fumigation to eliminate thousand cankers disease causal agents from black walnut. For Sci 65:452–459. https://doi.org/10.1093/forsci/fxz001
Silva X, Terhonen E, Sun H, Kasanen R, Heliövaara K, Jalkanen R, Asiegbu FO (2015) Comparative analyses of fungal biota carried by the pine shoot beetle (Tomicus piniperda L.) in northern and southern Finland. Scand J for Res 30:497–506. https://doi.org/10.1080/02827581.2015.1031824
Sitz RA, Luna EK, Caballero JI, Tisserat NA, Cranshaw WS, Stewart JE (2017) Virulence of genetically distinct Geosmithia morbida isolates to black walnut and their response to coinoculation with Fusarium solani. Plant Dis 101:116–120. https://doi.org/10.1094/PDIS-04-16-0535-RE
Sitz RA, Luna EK, Ibarra Caballero J, Tisserat NA, Cranshaw WS, McKenna JR, Stolz J, Stewart JE (2021) Eastern black walnut (Juglans nigra L.) originating from native range varies in their response to inoculation with Geosmithia morbida. Front For Glob Change 4:12. https://doi.org/10.3389/ffgc.2021.627911
Six DL (2013) The bark beetle holobiont: why microbes matter. J Chem Ecol 39:989–1002. https://doi.org/10.1007/s10886-013-0318-8
Six DL (2020) Niche construction theory can link bark beetle-fungus symbiosis type and colonization behavior to large scale causal chain-effects. Curr Opin Insect Sci 39:27–34. https://doi.org/10.1016/j.cois.2019.12.005
Six DL, Wingfield MJ (2011) The role of phytopathogenicity in bark beetle-fungus symbioses: a challenge to the classic paradigm. Annu Rev Entomol 56:255–272. https://doi.org/10.1146/annurev-ento-120709-144839
Six DL, Doug SW, de Beer ZW, Woolfolk SW (2009) Ambrosiella beaveri, sp. nov., associated with an exotic ambrosia beetle, Xylosandrus mutilatus (Coleoptera: Curculionidae, Scolytinae), in Mississippi, USA. Antonie Van Leeuwenhoek 96:17–29. https://doi.org/10.1007/s10482-009-9331-x
Skelton J, Jusino MA, Li Y, Bateman C, Thai PH, Wu C, Lindner DL, Hulcr J (2018) Detecting symbioses in complex communities: the fungal symbionts of bark and ambrosia beetles within Asian pines. Microb Ecol 76:839–850. https://doi.org/10.1007/s00248-018-1154-8
Skelton J, Loyd A, Smith JA, Blanchette RA, Held BW, Hulcr J (2020) Fungal symbionts of bark and ambrosia beetles can suppress decomposition of pine sapwood by competing with wood-decay fungi. Fungal Ecol 45:100926. https://doi.org/10.1016/j.funeco.2020.100926
Smith SM, Cognato AI (2014) A taxonomic monograph of Nearctic Scolytus Geoffroy (Coleoptera, Curculionidae, Scolytinae). ZooKeys 450:1–182. https://doi.org/10.3897/zookeys.450.7452
Solheim H (1991) Oxygen deficiency and spruce resin inhibition of growth of blue stain fungi associated with Ips typographus. Mycol Res 95:1387–1392. https://doi.org/10.1016/S0953-7562(09)80390-0
Stackhouse T, Boggess SL, Hadziabdic D, Trigiano RN, Ginzel MD, Klingeman WE (2021) Conventional gel electrophoresis and TaqMan probes enable rapid confirmation of Thousand Cankers Disease from diagnostic samples. Plant Dis 105:3171–3180. https://doi.org/10.1094/PDIS-10-20-2258-RE
Stodůlková E, Kolařík M, Křesinová Z, Kuzma M, Šulc M, Man P, Novák P, Maršík P, Landa P, Olšovská J, Chudíčková M, Pažoutová S, Černý J, Bella J, Flieger M (2009) Hydroxylated anthraquinones produced by Geosmithia species. Folia Microbiol 54:179–187. https://doi.org/10.1007/s12223-009-0028-3
Stodůlková E, Man P, Kolařík M, Flieger M (2010) High-performance liquid chromatography - off line mass spectrometry analysis of anthraquinones produced by Geosmithia lavendula. J Chromatogr 1217:6296–6302. https://doi.org/10.1016/j.chroma.2010.08.009
Stolk AC, Samson RA (1986) A new taxonomic scheme for Penicillium anamorphs. Advances in Penicillium and Aspergillus systematics. Springer, pp 163–192
Strzałka B, Kolařík M, Jankowiak R (2021) Geosmithia associated with hardwood-infesting bark and ambrosia beetles, with the description of three new species from Poland. Antonie Van Leeuwenhoek 114:169–194. https://doi.org/10.1007/s10482-020-01510-6
Sun Y, Takada K, Takemoto Y, Yoshida M, Nogi Y, Okada S, Matsunaga S (2012) Gliotoxin analogues from a marine-derived fungus, Penicillium sp., and their cytotoxic and histone methyltransferase inhibitory activities. J Nat Prod 75:111–114. https://doi.org/10.1021/np200740e
Sun Z-H, Gu J, Ye W, Wen L-X, Lin Q-B, Li S-N, Chen Y-C, Li H-H, Zhang W-M (2018) Geospallins A-C: new thiodiketopiperazines with inhibitory activity against angiotensin-converting enzyme from a deep-sea-derived fungus Geosmithia pallida FS140. Mar Drugs 16:464. https://doi.org/10.3390/md16120464
Thom CH (1930) The Penicillia. Williams & Wilkins, Baltimore
Tisserat N, Cranshaw W, Leatherman D, Utley C, Alexander K (2009) Black walnut mortality in Colorado caused by the walnut twig beetle and Thousand Cankers Disease. Plant Health Progr 10:1–10. https://doi.org/10.1094/php-2009-0811-01-rs
van Dyk M, Spies CFJ, Mostert L, van der Rijst M, du Plessis IL, Moyo P, van Jaarsveld WJ, Halleen F (2021) Pathogenicity testing of fungal isolates associated with olive trunk diseases in South Africa. Plant Dis 105:4060–4073. https://doi.org/10.1094/pdis-08-20-1837-re
Veselská T, Kolařík M (2015) Application of flow cytometry for exploring the evolution of Geosmithia fungi living in association with bark beetles: the role of conidial DNA content. Fungal Ecol 13:83–92. https://doi.org/10.1016/j.funeco.2014.08.007
Veselská T, Skelton J, Kostovčík M, Hulcr J, Baldrian P, Chudíčková M, Cajthaml T, Vojtová T, Garcia-Fraile P, Kolařík M (2019) Adaptive traits of bark and ambrosia beetle-associated fungi. Fungal Ecol 41:165–176. https://doi.org/10.1016/j.funeco.2019.06.005
Vitale S, Luongo L, Barzanti GP, Binazzi F, Petrucci M, Galli M, Pennacchio F, Francardi V (2021) First report of Geosmithia pallida and G langdonii asscociated with Liparthrum colchicum in Central Italy. J Zool 104:167–170. https://doi.org/10.19263/REDIA-104.21.18
Wang M, Kornsakulkarn J, Srichomthong K, Feng T, Liu J-K, Isaka M, Thongpanchang C (2019) Antimicrobial anthraquinones from cultures of the ant pathogenic fungus Cordyceps morakotii BCC 56811. J Antibiot 72:141–147. https://doi.org/10.1038/s41429-018-0135-y
Williams GM, Ginzel MD (2021) Competitive advantage of Geosmithia morbida in low-moisture wood may explain historical outbreaks of Thousand Cankers Disease and predict the future fate of Juglans nigra within its native range. Front For Glob Change 4:725066. https://doi.org/10.3389/ffgc.2021.725066
Wood SL (1982) The bark and ambrosia beetles of North and Central America (Coleoptera: Scolytidae), a taxonomic monograph. Great Basin Naturalist Memoirs 6:1–1359
Wright E (1938) Further investigations of brown-staining fungi associated with engraver beetles (Scolytus) in white fir. J Agric Res 57:759–773
Wu Y-M, Xu J-J, Wang H-F, Zhang T-Y (2013) Geosmithia tibetensis sp. nov. and new Gibellulopsis and Scopulariopsis records from Qinghai-Tibet. Mycotaxon 125:59–64. https://doi.org/10.5248/125.59
Yaguchi T, Miyadoh S, Udagawa S (1993) Chromocleista, a new cleistothecial genus with a Geosmithia anamorph. Trans Mycol Soc Japan 34:101–108. https://doi.org/10.2307/3761132
Yaguchi T, Someya A, Udagawa S (1994) Two new species of Talaromyces from Taiwan and Japan. Mycoscience 35:249–255. https://doi.org/10.1007/BF02268446
Yaguchi T, Udagawa S, Nishimura K (2005) Geosmithia argillacea is the anamorph of Talaromyces eburneus as a heat resistant fungus. Cryptogamie: Mycol 26:133–141. https://doi.org/10.3343/alm.2013.33.2.136
Zerillo MM, Caballero JI, Woeste K, Graves AD, Hartel C, Pscheidt JW, Tonos J, Broders K, Cranshaw W, Seybold SJ (2014) Population structure of Geosmithia morbida, the causal agent of thousand cankers disease of walnut trees in the United States. PLoS ONE 9:e112847. https://doi.org/10.1371/journal.pone.0112847
Zhang X, Li Y, Si H, Zhao G, Kolařík M, Hulcr J, Jiang X, Dai M, Chang R (2022) Geosmithia species associated with bark beetles from China, with the description of nine new species. Front Microbiol 13:820402. https://doi.org/10.3389/fmicb.2022.820402
Zhu H-y, Zhang D, Zhang Q, Zhao Y, He Z-m, Gao Y-g, Zhang L-x (2018) 4-hydroxybenzyl alcohol derivatives and their sedative–hypnotic activities. RSC Adv 8:19539–19550. https://doi.org/10.1039/c8ra01972j
Acknowledgements
We are greatly indebted to the reviewers and especially to the Associate Editor, Roland Kirschner, for suggestions and significant improvements to our manuscript.
Funding
M.K. benefitted from funding by the European Union’s Horizon 2020 research and innovation program (RISE) under the Marie Skłodowska-Curie grant agreement no. 101008129, project acronym “Mycobiomics”, and from Czech Science Foundation (GAČR), grant number 19-09072S.
Author information
Authors and Affiliations
Contributions
M.K. prepared the first draft of the manuscript, figures, and tables. J.H. contributed significantly to the editing of the manuscript and improved the form of presentation of the results (figures and tables).
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third-party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
Additional information
Section Editor: Roland Kirschner
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kolařík, M., Hulcr, J. Geosmithia—widespread and abundant but long ignored bark beetle symbionts. Mycol Progress 22, 32 (2023). https://doi.org/10.1007/s11557-023-01880-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11557-023-01880-x