Abstract
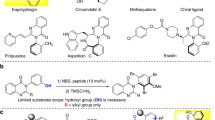
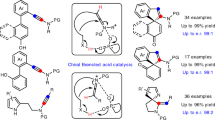
Catalytic asymmetric transformations of ynamides have attracted considerable attention in recent years. However, most of them were limited to intramolecular reactions or required metal catalysts. Herein, a chiral Brønsted acid-catalyzed asymmetric intermolecular [4 + 2] annulation of ynamides with para-quinone methides (p-QMs) is disclosed, which not only represents the first metal-free protocol for catalytic asymmetric nucleophilic addition of ynamides to electrophiles, but also constitutes the first enantioselective annulation between p-QMs and alkynes. This methodology leads to the practical synthesis of biologically important chiral 4-aryl-3,4-dihydrocoumarins and 4-aryl-coumarins. Preliminary control experiments indicate that the ortho-hydroxyphenyl substituted p-QMs could isomerize into ortho-quinone methides (o-QMs) in the presence of chiral catalyst, which further react with ynamides via enantioselective [4 + 2] annulation, to generate the chiral product.

Similar content being viewed by others
References
For selected examples for 4-aryl-3,4-dihydrocoumarins in natural products and bioactive molecules, see: (a) Li GT, Li ZK, Gu Q, You SL. Org Lett, 2017, 19: 1318–1321
Neugebauer RC, Uchiechowska U, Meier R, Hruby H, Valkov V, Verdin E, Sippl W, Jung M. J Med Chem, 2008, 51: 1203–1213
Yao CS, Lin M, Wang L. Chem Pharm Bull, 2006, 54: 1053–1057
Zhang X, Wang H, Song Y, Nie L, Wang L, Liu B, Shen P, Liu Y. Bioorg Med Chem Lett, 2006, 16: 949–953
Iinuma M, Tanaka T, Takenaka M, Mizuno M, Asai F. Phytochemistry, 1992, 31: 2487–2490
Nonaka G, Kawa-hara O, Nishioka I. Chem Pharm Bull, 1982, 30: 4277–4282
Leitis Z. Chem Heterocycl Comp, 2016, 52: 527–529
Kamat DP, Tilve SG, Kamat VP, Kirtany JK. Org Prep Proc Int, 2015, 47: 1–79
Sudo A, Uenishi K, Endo T. Polym Int, 2009, 58: 970–975
For selected reviews on p-QMs, see: (a) Singh G, Pandey R, Pankhade YA, Fatma S, Anand RV. Chem Record, 2021, 21: 4150–4173
Lima CGS, Pauli FP, Costa DCS, de Souza AS, Forezi LSM, Ferreira VF, de Carvalho da Silva F. Eur J Org Chem, 2020, 2020: 2650–2692
Wang JY, Hao WJ, Tu SJ, Jiang B. Org Chem Front, 2020, 7: 1743–1778
Li W, Xu X, Zhang P, Li P. Chem Asian J, 2018, 13: 2350–2359
Chauhan P, Kaya U, Enders D. Adv Synth Catal, 2017, 359: 888–912
Parra A, Tortosa M. ChemCatChem, 2015, 7: 1524–1526
Caruana L, Fochi M, Bernardi L. Molecules, 2015, 20: 11733–11764
For selected examples on catalytic asymmetric reactions involving p-QMs: (a) Zurro M, Ge L, Harutyunyan SR. Org Lett, 2022, 24: 6686–6691
Santra S, Porey A, Jana B, Guin J. Chem Sci, 2018, 9: 6446–6450
Li W, Xu X, Liu Y, Gao H, Cheng Y, Li P. Org Lett, 2018, 20: 1142–1145
Chen M, Sun J. Angew Chem Int Ed, 2017, 56: 4583–4587
Li S, Liu Y, Huang B, Zhou T, Tao H, **ao Y, Liu L, Zhang J. ACS Catal, 2017, 7: 2805–2809
Zhao K, Zhi Y, Wang A, Enders D. ACS Catal, 2016, 6: 657–660
Ma C, Huang Y, Zhao Y. ACS Catal, 2016, 6: 6408–6412
Dong N, Zhang ZP, Xue XS, Li X, Cheng JP. Angew Chem Int Ed, 2016, 55: 1460–1464
Wang Z, Wong YF, Sun J. Angew Chem Int Ed, 2015, 54: 13711–13714
Lou Y, Cao P, Jia T, Zhang Y, Wang M, Liao J. Angew Chem Int Ed, 2015, 54: 12134–12138
Caruana L, Kniep F, Johansen TK, Poulsen PH, Jørgensen KA. J Am Chem Soc, 2014, 136: 15929–15932
Chu WD, Zhang LF, Bao X, Zhao XH, Zeng C, Du JY, Zhang GB, Wang FX, Ma XY, Fan CA. Angew Chem Int Ed, 2013, 52: 9229–9233
For recent selected examples on catalytic asymmetric annulation for ortho-hydroxyphenyl substituted p-QMs, see: (a) **ang M, Li CY, Song XJ, Zou Y, Huang ZC, Li X, Tian F, Wang LX. Chem Commun, 2020, 56: 14825–14828
Wang JR, Jiang XL, Hang QQ, Zhang S, Mei GJ, Shi F. J Org Chem, 2019, 84: 7829–7839
Yang GH, Zhao Q, Zhang ZP, Zheng HL, Chen L, Li X. J Org Chem, 2019, 84: 7883–7893
Liu Q, Li S, Chen XY, Rissanen K, Enders D. Org Lett, 2018, 20: 3622–3626
Liu L, Yuan Z, Pan R, Zeng Y, Lin A, Yao H, Huang Y. Org Chem Front, 2018, 5: 623–628
Jiang XL, Wu SF, Wang JR, Mei GJ, Shi F. Adv Synth Catal, 2018, 360: 4225–4235
Li W, Yuan H, Liu Z, Zhang Z, Cheng Y, Li P. Adv Synth Catal, 2018, 360: 2460–2464
Zhang ZP, Chen L, Li X, Cheng JP. J Org Chem, 2018, 83: 2714–2724
Zhang ZP, **e KX, Yang C, Li M, Li X. J Org Chem, 2018, 83: 364–373
Zhang L, Liu Y, Liu K, Liu Z, He N, Li W. Org Biomol Chem, 2017, 15: 8743–8747
Zhang L, Zhou X, Li P, Liu Z, Liu Y, Sun Y, Li W. RSC Adv, 2017, 7: 39216–39220
Zhao K, Zhi Y, Shu T, Valkonen A, Rissanen K, Enders D. Angew Chem Int Ed, 2016, 55: 12104–12108
For recent reviews on ynamide reactivity, see: (a) Hu YC, Zhao Y, Wan B, Chen QA. Chem Soc Rev, 2021, 50: 2582–2625
Chen YB, Qian PC, Ye LW. Chem Soc Rev, 2020, 49: 8897–8909
Hong FL, Ye LW. Acc Chem Res, 2020, 53: 2003–2019
Lynch CC, Sripada A, Wolf C. Chem Soc Rev, 2020, 49: 8543–8583
Luo J, Chen GS, Chen SJ, Yu JS, Li ZD, Liu YL. ACS Catal, 2020, 10: 13978–13992
Zhou B, Tan TD, Zhu XQ, Shang M, Ye LW. ACS Catal, 2019, 9: 6393–6406
Evano G, Theunissen C, Lecomte M. Aldrichimica Acta, 2015, 48: 59–70
Wang XN, Yeom HS, Fang LC, He S, Ma ZX, Kedrowski BL, Hsung RP. Acc Chem Res, 2014, 47: 560–578
DeKorver KA, Li H, Lohse AG, Hayashi R, Lu Z, Zhang Y, Hsung RP. Chem Rev, 2010, 110: 5064–5106
Evano G, Coste A, Jouvin K. Angew Chem Int Ed, 2010, 49: 2840–2859
Zhou B, Zhang YQ, Zhang K, Yang MY, Chen YB, Li Y, Peng Q, Zhu SF, Zhou QL, Ye LW. Nat Commun, 2019, 10: 3234
Zhang YQ, Chen YB, Liu JR, Wu SQ, Fan XY, Zhang ZX, Hong X, Ye LW. Nat Chem, 2021, 13: 1093–1100
Chen PF, Zhou B, Wu P, Wang B, Ye LW. Angew Chem Int Ed, 2021, 60: 27164–27170
Li HH, Zhang YP, Zhai TY, Liu BY, Shi CY, Zhou JM, Ye LW. Org Chem Front, 2022, 9: 3709–3717
Wang ZS, Zhu LJ, Li CT, Liu BY, Hong X, Ye LW. Angew Chem Int Ed, 2022, 61: e202201436
Zhang ZX, Wang X, Jiang JT, Chen J, Zhu XQ, Ye LW. Chin Chem Lett, 2023, 34: 107647
Moskowitz M, Wolf C. Angew Chem IntEd, 2019, 58: 3402–3406
Cook AM, Wolf C. Angew Chem Int Ed, 2016, 55: 2929–2933
Cook AM, Wolf C. Chem Commun, 2014, 50: 3151–3154
Schotes C, Mezzetti A. Angew Chem Int Ed, 2011, 50: 3072–3074
Aikawa K, Hioki Y, Shimizu N, Mikami K. J Am Chem Soc, 2011, 133: 20092–20095
Enomoto K, Oyama H, Nakada M. Chem Eur J, 2015, 21: 2798–2802
For alternative examples on transition metal-catalyzed asymmetric cycloaddition of ynamides via cyclometallation type approach, see: (a) Straker RN, Peng Q, Mekareeya A, Paton RS, Anderson EA. Nat Commun, 2016, 7: 10109
Oppenheimer J, Hsung RP, Figueroa R, Johnson WL. Org Lett, 2007, 9: 3969–3972
Tanaka K, Takeishi K, Noguchi K. J Am Chem Soc, 2006, 128: 4586–4587
Qi LJ, Li CT, Huang ZQ, Jiang JT, Zhu XQ, Lu X, Ye LW. Angew Chem Int Ed, 2022, 61: e202210637
Zhu GY, Zhou JJ, Liu LG, Li X, Zhu XQ, Lu X, Zhou JM, Ye LW. Angew Chem Int Ed, 2022, 61: e202204603
Hong FL, Shi CY, Hong P, Zhai TY, Zhu XQ, Lu X, Ye LW. Angew Chem Int Ed, 2022, 61: e202115554
Zhang YQ, Zhang YP, Zheng YX, Li ZY, Ye LW. Cell Rep Phys Sci, 2021, 2: 100448–100463
Zhu XQ, Hong P, Zheng YX, Zhen YY, Hong FL, Lu X, Ye LW. Chem Sci, 2021, 12: 9466–9474
Zhu BH, Zheng YX, Kang W, Deng C, Zhou JM, Ye LW. Sci China Chem, 2021, 64: 1985–1989
Wang ZS, Chen YB, Zhang HW, Sun Z, Zhu C, Ye LW. J Am Chem Soc, 2020, 142: 3636–3644
Liu X, Wang ZS, Zhai TY, Luo C, Zhang YP, Chen YB, Deng C, Liu RS, Ye LW. Angew Chem Int Ed, 2020, 59: 17984–17990
Hong FL, Chen YB, Ye SH, Zhu GY, Zhu XQ, Lu X, Liu RS, Ye LW. J Am Chem Soc, 2020, 142: 7618–7626
For selected examples about 4-aryl-coumarins in natural products and bioactive molecules, see: (a) Yun Y, Yang J, Miao Y, Wang X, Sun J. Bioorg Med Chem Lett, 2021, 30: 126900–126906
Patil AD, Freyer AJ, Eggleston DS, Haltiwanger RC, Bean MF, Taylor PB, Caranfa MJ, Breen AL, Bartus HR. J Med Chem, 1993, 36: 4131–4138
Brenzan MA, Nakamura CV, Dias Filho BP, Ueda-Naka-mura T, Young MCM, Côrrea AG, Júnior JA, dos Santos AO, Cortez DAG. Biomed Pharmacother, 2008, 62: 651–658
Acknowledgements
This work was supported by the Ministry of Science and Technology (MOST) (2021YFC2100100), the National Natural Science Foundation of China (22125108, 22121001, 92056104), the President Research Funds from **amen University (20720210002), the Natural Science Foundation of Jiangsu Province (BK20211059), the Project of Science and Technology of Xuzhou Government (KC22080), and the National Fund for Fostering Talents of Basic Science (NFFTBS) (J1310024).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of interest
The authors declare no conflict of interest.
Supporting information
The supporting information is available online at chem.scichina.com and springer.longhoe.net/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Rights and permissions
About this article
Cite this article
Li, HH., Meng, YN., Chen, CM. et al. Chiral Brønsted acid-catalyzed asymmetric intermolecular [4 + 2] annulation of ynamides with para-quinone methides. Sci. China Chem. 66, 1467–1473 (2023). https://doi.org/10.1007/s11426-022-1536-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1536-9