Abstract
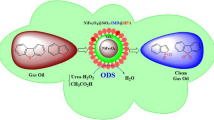
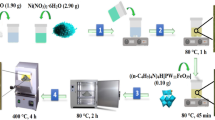
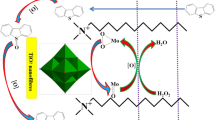
Magnetic nanoparticles surrounded with a silica shell are useful materials to immobilize active agents on their surface. Here, a heteropolyacid-functionalized hybrid nanomaterial (NiFe2O4@SiO2-DETA@POM) was prepared and characterized by X-ray powder diffraction patterns (XRD), Fourier-transform infrared spectroscopy (FT-IR), thermogravimetric analysis (TGA/DTG), vibrating sample magnetometer (VSM), the field emission scanning electron microscopy (FE-SEM), and the electron-dispersive X-ray spectroscopy (EDS). The synthesized hybrid nanostructure was used as a solid nanocatalyst in oxidative desulfurization (ODS) of real fuel and simulated gasoline samples. The ODS process of benzothiophene (BT) and dibenzothiophene (DBT) as model compounds in the presence of NiFe2O4@SiO2-DETA@POM and by using urea-hydrogen peroxide/acetic acid as a safer oxidizing agent was investigated. A good result was obtained by removing 97% of benzothiophene and 98% of dibenzothiophene. Also, 96% of the sulfur compounds were eliminated when the ODS process was tested on a real crude oil sample (600 ppm) under an optimized dosage of nanocatalyst, urea-hydrogen peroxide/acetic acid (0.1 g, 1 g/4 ml) at 50 ºC for 60 min. NiFe2O4@SiO2-DETA@POM could be recycled for five consecutive oxidation runs without significant deterioration in its catalytic activity. The UHP’s safety and efficiency as an oxidant, high removal efficacy, short transformation times, easy workup procedure, catalyst reusability, simple separation of nanocatalyst, green conditions, and environmental compatibility and sustainability. The obtained results prove that NiFe2O4@SiO2-DETA@POM is a suitable and efficient hybrid catalyst for the oxidative desulfurization of simulated and real fuels.
Graphical Abstract














Similar content being viewed by others
Data availability
There is no additional data.
References
Ammam M (2013) Polyoxometalates: formation, structures, principal properties, main deposition methods and application in sensing. J Mater Chem A 1:6291–6312. https://doi.org/10.1039/C3TA01663C
Ahadi N, Bodaghifard MA, Mobinikhaledi A (2020) Preparation and characterization of a novel organic–inorganic hybrid nanostructure: application in synthesis of spirocompounds. Res Chem Intermed 46:3277–3294
Bodaghifard MA, Hamidinasab M, Ahadi N (2018) Recent advances in the preparation and application of organic– inorganic hybrid magnetic nanocatalysts on multicomponent reactions. Curr Org Chem 22:234–267. https://doi.org/10.2174/1385272821666170705144854
Bodaghifard MA, Shafi S (2021) Ionic liquid-immobilized hybrid nanomaterial: an efficient catalyst in the synthesis of benzimidazoles and benzothiazoles via anomeric-based oxidation. J Iran Chem Soc 18:677–687. https://doi.org/10.1007/S13738-020-02055-1
Boniek D, Figueiredo D, Dos Santos AFB, De Resende Stoianoff MA (2015) Biodesulfurization: a mini review about the immediate search for the future technology. Clean Technol Environ Policy 17:29–37. https://doi.org/10.1007/S10098-014-0812-X
Bodaghifard MA (2019) Palladium-melamine complex anchored on magnetic nanoparticles: a novel promoter for CC cross coupling reaction. J Organomet Chem 886:57–64. https://doi.org/10.1016/j.jorganchem.2019.02.010
Chen TC, Shen YH, Lee WJ et al (2010) The study of ultrasound-assisted oxidative desulfurization process applied to the utilization of pyrolysis oil from waste tires. J Clean Prod 18:1850–1858. https://doi.org/10.1016/J.JCLEPRO.2010.07.019
Cherevan AS, Nandan SP, Roger I et al (2020) Polyoxometalates on functional substrates: concepts, synergies, and future perspectives. Adv Sci 7:1903511. https://doi.org/10.1002/ADVS.201903511
Craven M, **ao D, Kunstmann-Olsen C et al (2018) Oxidative desulfurization of diesel fuel catalyzed by polyoxometalate immobilized on phosphazene-functionalized silica. Appl Catal B 231:82–91. https://doi.org/10.1016/j.apcatb.2018.03.005
Gawande MB, Branco PS, Varma RS (2013) Nano-magnetite (Fe3O4) as a support for recyclable catalysts in the development of sustainable methodologies. Chem Soc Rev 42:3371–3393. https://doi.org/10.1039/C3CS35480F
Gu Q, Wen G, Ding Y et al (2017) Reduced graphene oxide: a metal-free catalyst for aerobic oxidative desulfurization. Green Chem 19:1175–1181. https://doi.org/10.1039/C6GC02894B
Hamidinasab M, Bodaghifard MA, Mobinikhaledi A (2020a) Green synthesis of 1H-pyrazolo[1,2-b]phthalazine-2-carbonitrile derivatives using a new bifunctional base–ionic liquid hybrid magnetic nanocatalyst. Appl Organomet Chem 34. https://doi.org/10.1002/AOC.5386
Hamidinasab M, Bodaghifard MA, Mobinikhaledi A (2020b) Synthesis of new, vital and pharmacologically important bis phthalazine-triones using an efficient magnetic nanocatalyst and their HF and NBO investigation. J Mol Struct 1200:127091. https://doi.org/10.1016/J.MOLSTRUC.2019.127091
HozhabrAraghi S, Entezari MH (2015) Amino-functionalized silica magnetite nanoparticles for the simultaneous removal of pollutants from aqueous solution. Appl Surf Sci 333:68–77. https://doi.org/10.1016/j.apsusc.2015.01.211
Injumpa W, Ritprajak P, Insin N (2017) Size-dependent cytotoxicity and inflammatory responses of PEGylated silica-iron oxide nanocomposite size series. J Magn Magn Mater 427:60–66. https://doi.org/10.1016/j.jmmm.2016.11.015
Jiang W, Jia H, Fan X et al (2019) Ionic liquid immobilized on magnetic mesoporous microspheres with rough surface: application as recyclable amphiphilic catalysts for oxidative desulfurization. Appl Surf Sci 484:1027–1034. https://doi.org/10.1016/j.apsusc.2019.03.341
Jiang W, Li H, Wang C et al (2016) Synthesis of ionic-liquid-based deep eutectic solvents for extractive desulfurization of fuel. Energy Fuels 30:8164–8170. https://doi.org/10.1021/ACS.ENERGYFUELS.6B01976/SUPPL_FILE/EF6B01976_SI_001.PDF
Kim HS, Kim D, Kwak BS et al (2014) Synthesis of magnetically separable core at shell structured NiFe2O4 at TiO2 nanomaterial and its use for photocatalytic hydrogen production by methanol/water splitting. Chem Eng J 243:272–279. https://doi.org/10.1016/j.cej.2013.12.046
Liu Y, Zuo P, Wang R et al (2020) Covalent immobilization of Dawson polyoxometalates on hairy particles and its catalytic properties for the oxidation desulfurization of tetrahydrothiophene. J Clean Prod 274:122774. https://doi.org/10.1016/j.jclepro.2020.122774
Nejati K, Zabihi R (2012) Preparation and magnetic properties of nano size nickel ferrite particles using hydrothermal method. Chem Cent J 6:1–6. https://doi.org/10.1186/1752-153X-6-23/TABLES/2
Rezvani MA, Fereyduni M (2019) Synthesis of organic−inorganic hybrid nanocomposite polyoxometalate/metal oxide/CS polymer (PMnW11@TiO2@CS): nanocatalyst for oxidative desulfurization of real fuel. ChemistrySelect 4:11467–11474. https://doi.org/10.1002/SLCT.201902654
Rezvani MA, Hadi M, Mirsadri SA (2020a) Synthesis of new nanocomposite based on nanoceramic and mono substituted polyoxometalate, PMo11Cd@MnFe2O4, with superior catalytic activity for oxidative desulfurization of real fuel. Appl Organomet Chem 34:e5882. https://doi.org/10.1002/AOC.5882
Rezvani MA, Shaterian M, Aghmasheh M (2020b) Catalytic oxidative desulphurization of gasoline using amphiphilic polyoxometalate@polymer nanocomposite as an efficient, reusable, and green organic–inorganic hybrid catalyst. Environ Technol 41:1219–1231. https://doi.org/10.1080/09593330.2018.1526217
Rezvani MA, Imani A (2021) Ultra-deep oxidative desulfurization of real fuels by sandwich-type polyoxometalate immobilized on copper ferrite nanoparticles, Fe6W18O70⊂ CuFe2O4, as an efficient heterogeneous nanocatalyst. J Environ Chem Eng 9. https://doi.org/10.1016/j.jece.2020.105009
Rezvani MA, Mirsadri SA (2020) Synthesis and characterization of new hybrid inorganic–organic polymer nanocomposite as efficient catalyst for oxidative desulfurization of real fuel. Appl Organomet Chem 34:e5585. https://doi.org/10.1002/AOC.5585
Rezvani MA, Shaterian M, Akbarzadeh F, Khandan S (2018) Deep oxidative desulfurization of gasoline induced by PMoCu@MgCu2O4-PVA composite as a high-performance heterogeneous nanocatalyst. Chem Eng J 333:537–544. https://doi.org/10.1016/J.CEJ.2017.09.184
Safa M, Mokhtarani B, Mortaheb HR et al (2017) Oxidative desulfurization of diesel fuel using a brønsted acidic ionic liquid supported on silica gel. Energy Fuels 31:10196–10205. https://doi.org/10.1021/ACS.ENERGYFUELS.6B03505
Sen R, Jain P, Patidar R et al (2015) Synthesis and characterization of nickel ferrite (NiFe2O4) nanoparticles prepared by sol- gel method. Mater Today: Proceedings 2:3750–3757. https://doi.org/10.1016/j.matpr.2015.07.165
Taghizadeh M, Mehrvarz E, Taghipour A (2020) Polyoxometalate as an effective catalyst for the oxidative desulfurization of liquid fuels: a critical review. Rev Chem Eng 36:831–858. https://doi.org/10.1515/REVCE-2018-0058/XML
Yang E, Yao C, Liu Y et al (2018) Bamboo-derived porous biochar for efficient adsorption removal of dibenzothiophene from model fuel. Fuel 211:121–129. https://doi.org/10.1016/j.fuel.2017.07.099
Ye JJ, De Wu C (2016) Immobilization of polyoxometalates in crystalline solids for highly efficient heterogeneous catalysis. Dalton Trans 45:10101–10112. https://doi.org/10.1039/C6DT01378C
Zhou Y, Chen G, Long Z, Wang J (2014) Recent advances in polyoxometalate-based heterogeneous catalytic materials for liquid-phase organic transformations. RSC Adv 4:42092–42113. https://doi.org/10.1039/C4RA05175K
Funding
The authors acknowledge the financial support of this work by the Research Council of Arak University.
Author information
Authors and Affiliations
Contributions
M.A Bodaghifard conceived, planned, and supervised the project. P. Bayat carried out the experiments and analyses. M. Hamidinasab advised the project and wrote the manuscript with support from M.A. Bodaghifard. All authors discussed the results and contributed to the final manuscript. M.A. Bodaghifard revised the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bodaghifard, M.A., Hamidinasab, M. & Bayat, P. Deep oxidative desulfurization of simulated and real gas oils by NiFe2O4@SiO2-DETA@POM as a retrievable hybrid nanocatalyst. Environ Sci Pollut Res 30, 57821–57832 (2023). https://doi.org/10.1007/s11356-023-26614-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26614-0