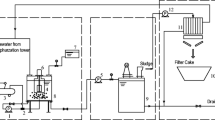
Abstract
Most of the existing oxidation denitrification methods need longer residence time to obtain higher NOx removal efficiency. In this study, urea peroxide (CO(NH2)2·H2O2) was first used for removing SO2 and NOx on diesel engine bench. The addition of ferrous sulfate can enhance the oxidant capacity of the solution. The better removal efficiency and lower nitrate content in liquid can be achieved in short exhaust gas residence time. The raw gas flow and residence time contained the actual application situation in ships and have high reference value. The removal efficiency decreased with the increase of gas flow, and the reaction temperature, urea peroxide concentration, liquid–gas ratio were the main factors. The optimal Fe2+ concentration of 50 mmol/L and pH value of 4 were determined. The urea peroxide concentration, reaction temperature, and liquid–gas ratio were 9%, 70 ℃, and 10 L/m3 respectively. The maximum gas treatment capacity was about 100 L/min, and residence time was close to 10 s for the scrubber. The pre-agglomerating method were used to improve the particle capturing efficiency combined with spray technology. The composited method can realize the synchronous and efficient removal of multiple pollutants in a single scrubber. The possibility of application on ship was further increased.















Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. And the datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- PM:
-
Particulate matter
- NOx:
-
Nitrogen oxides
- SOx:
-
Sulfur oxides
- SCR:
-
Selective catalytic reduction
- EGC:
-
Exhaust gas cleaning
- OC:
-
Organic carbon
- EC:
-
Elemental carbon
- CO:
-
Carbon monoxide
- CO2 :
-
Carbon dioxide
- HFO:
-
Heavy fuel oil
- FSC:
-
Fuel sulfur content
- IMO:
-
International Maritime Organization
References
Adewuyi TG, Sakyi NY, Khan MA (2018) Simultaneous removal of NO and SO2 from flue gas by combined heat and Fe2+ activated aqueous persulfate solutions. Chemosphere 193:1216–1225
Ahile UJ, Wuana RA, Itodo AU, Sha’Ato R, Dantas RF (2020) A review on the use of chelating agents as an alternative to promote photo-Fenton at neutral pH: current trends, knowledge gap and future studies. Sci Total Environ 710:134872
Erik F, Kent S (2016) Measurements of abatement of particles and exhaust gases in a marine gas scrubber. J Eng Marit Environ 1:154–162
Flagiello D, Erto A, Lancia A, Di Natale F (2022) Advanced flue-gas cleaning by wet oxidative scrubbing (WOS) using NaClO2 aqueous solutions. Chem Eng J 447:137585
Herdzik J (2021) Decarbonization of marine fuels-the future of ship**. Energies 14:4311
Kang X, Ma X, Ja Y, Gao X (2018) A study on simultaneous removal of NO and SO2 by using sodium persulfate aqueous scrubbing. Chin J Chem Eng 26:1536–1544
Koytsoumpa EI, Bergins C, Kakaras E (2018) The CO2 economy: review of CO2 capture and reuse technologies. Journal of Supercritical Fluid 132:3–16
Liu Y, Wang Y, Wang Q, Pan J, Zhang Y, Zhou J, Zhang J (2015a) A study on removal of elemental mercury in flue gas using fenton solution. J Hazard Mater 292:164–172
Liu Y, Zhou J, Zhang Y, Pan J, Zhang J (2015b) Removal of Hg0 and simultaneous removal of Hg0/SO2/NO in flue gas using two Fenton-like reagents in a spray reactor. Fuel 145:180–188
Liu A, Wang L, Wu J, **ao L, **g X, Wang L, Ma L, Wang H (2022a) Simultaneous removal of SO2 and NO from the flue gas of marine ships with a gas cyclone−liquid jet absorption separator. Ind Eng Chem Res 61:10233–10242
Liu W, Kong W, Jiang H, Cheng C, Yang Z, Huang R, Zhang R, Yang L (2022b) Application research on deep removal of multi pollutants in condensation and coagulation system. Electric Power Technol Environ Prot 38:135–141
Morosini DF, Baltar CAM, Coelho ACD (2014) Iron removal by precipitate flotation. Rem Revista Escola De Minas 6:203–207
Wang Z, Ye Y, Chen N, Li H (2016) Study on the removal of nitric oxide (NO) by dual oxidant (H2O2/Na2S2O8) system. Chem Eng Sci 145:133–140
Wang Z, Lun L, Tan Z, Zhang Y, Li Q (2019) Simultaneous wet desulfurization and denitration by an oxidant absorbent of NaClO2/CaO2. Environ Sci Pollut Res 26:29032–29040
Wang C, Chen Z, Deng J, Wang L (2021a) Progress on combustion and emission reduction technology of marine modified heavy fuel oil. Energy Eng 1:63–69
Wang Y, Wang ZH, Liu YX (2021b) Review on removal of SO2, NOx, Mercury, and Arsenic from flue gas using green oxidation absorption technology. Energy Fuels 35:9775–9794
Wilailak S, Yoo BH, Kim Y, Lee CJ (2021) Parametric analysis and design optimization of wet SOx scrubber system in marine industry. Fuel 304:121369
** HY, Zhou S, Zhang Z (2020) A novel method for the synchronous absorption of SO2 and NO from marine diesel engines. Fuel Process Technol 210:106560
Yang BC, Ma SX, Cui RG, Wang J (2019) Novel low-cost simultaneous removal of NO and SO2 with ∙OH from decomposition of H2O2 catalyzed by alkali-magnetic modified fly ash. Ind Eng Chem Res 58:5339–5347
Yang SL, An JX, **ang XB, Han ZT, Pan XX (2021) UV enhanced denitrification using chlorine from seawater electrolysis for hydrogen production. Int J Hydrogen Energy 46:16836–16846
Yoo BY (2017) Economic assessment of liquefied natural gas (LNG) as a marine fuel for CO2 carriers compared to marine gas oil (MGO). Energy 121:772–780
Yuan P, Egedy A, Miskolczi N, Shen B, Wang J, Zhou W, Pan Y, Zhang H (2018a) Oxidation removal of NO by in situ Fenton system: factors and optimization. Fuel 233:519–528
Yuan P, Mei X, Shen B, Lu F, Zhou W, Si M (2018b) Chakraborty, S. Oxidation of NO by in situ Fenton reaction system with dual ions as reagents. Chem Eng J 351:660–667
Zhang Z, ** HY, Zhou S, Zhou WJ, Shreka M (2022) Efficient removal of NOx from simulated marine exhaust by using O3-Na2SO3: Experimental factors and optimization analysis. Fuel 313:122659
Zhao Y, Wen WX, Guo TX, Zhou JH (2014) Desulfurization and denitrogenation from flue gas using Fenton reagent. Fuel Process Technol 128:54–60
Zhou JX, Wang H (2020) Study on efficient removal of SOx and NOx from marine exhaust gas by wet scrubbing method using urea peroxide solution. Chem Eng J 390:124567
Acknowledgements
We gratefully acknowledge the financial support of WUST Research Fund (KJRC2022010).
Author information
Authors and Affiliations
Contributions
**xi Zhou: resources, supervision, project administration, methodology, investigation, writing-review and editing, funding acquisition, investigation, data curation, visualization. Guoxian Jiang: resources, supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
We declare that we have no human participants or human data.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, J., Jiang, G. Study on removing marine multiple pollutants in raw exhaust gas with a novel composited method combined with pre-agglomeration and wet scrubbing technology. Environ Sci Pollut Res 30, 47262–47273 (2023). https://doi.org/10.1007/s11356-023-25660-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-25660-y