Abstract
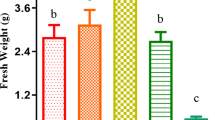
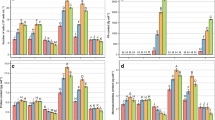
As a result of its high production, bisphenol A (BPA) has become ubiquitous in aquatic and terrestrial habitats. In this study, we investigated the toxicity of BPA at 10 mg L−1 on Lemna minor after 7 days of exposure under controlled conditions according to ISO 20079. BPA statistically reduced the total frond number and frond area, while frond number per colony was significantly elevated in BPA-treated group. However, no change was recorded in root number, while root length was significantly reduced by BPA. BPA also decreased the content of Chl a, Chl b, Chl a + b, and carotenoid by 36%, 44%, 38%, and 32%, respectively, versus the control leading to a decrease in the quantum yield of photosystem II. In addition, non-photochemical quenching (NPQ) values were 2.4- and 4.5-fold higher in light than in dark conditions for control and BPA-treated plants, respectively. Thus, there is a significant activation (61.8%; p<0.01) of PSII photoprotection mechanism (NPQ) in BPA-treated plants compared to control but without removing the negative effect of BPA on PSII. The total amount of soluble sugars was reduced by 40% compared to control, and starch accumulation was mainly observed in fronds exposed to BPA. Even if the response patterns of Lemna minor based on fresh and dry weight measurements were less sensitive in our experiment conditions, further studies should be addressed since BPA represents a threat to the dynamic equilibrium governing aquatic ecosystems.






Similar content being viewed by others
References
Adamakis IS, Malea P, Panteris E (2018) The effects of bisphenol A on the seagrass Cymodocea nodosa: leaf elongation impairment and cytoskeleton disturbance. Ecotoxicol Environ Saf 157:431–440. https://doi.org/10.1016/j.ecoenv.2018.04.005
Adamakis IS, Malea P, Sperdouli I, Panteris E, Kokkinidi D, Moustakas M (2021) Evaluation of the spatiotemporal effects of bisphenol A on the leaves of the seagrass Cymodocea nodosa. J Hazard Mater 404:124001. https://doi.org/10.1016/j.jhazmat.2020.124001
Almeida S, Raposo A, Almeida-González M, Carrascosa C (2018) Bisphenol A: food exposure and impact on human health. Compr Rev Food Sci Food Saf 17:1503–1517. https://doi.org/10.1111/1541-4337.12388
Amin MM, Ebrahim K, Hashemi M, ShoshtariYeganeh B, Rafiei N, Mansourian M, Kelishadi R (2019) Association of exposure to bisphenol A with obesity and cardiometabolic risk factors in children and adolescents. Int J Environ Health Res 29:94–106
Appenroth KJ, Krech K, Keresztes A, Fischer W, Koloczek H (2010) Effects of nickel on the chloroplasts of the duckweeds Spirodela polyrhiza and Lemna minor and their possible use in biomonitoring and phytoremediation. Chemosphere 78:216–233
Bahmani R, Kim D, Modareszadeh M, Thompson AJ, Park JH, Yoo HH, Hwang S (2020) The mechanism of root growth inhibition by the endocrine disruptor bisphenol A (BPA). Environ Pollut 257:113516. https://doi.org/10.1016/j.envpol.2019.113516
Bertolde FZ, Almeida AAF, Corrêa RX, Gomez FP, Gaiotto FA, Baligar VC, Loguercio LL (2010) Molecular, physiological and morphological analysis of waterlogging tolerance in clonal genotypes of Theobroma cacao L. Tree Physiol 30:56–67
Bourioug M, Mazzitelli J-Y, Marty P, Budzinski H, Aleya L, Bonnafé E, Geret F (2017) Assessment of Lemna minor (duckweed) and Corbicula fluminea (freshwater clam) as potential indicators of contaminated aquatic ecosystems: responses to presence of psychoactive drug mixtures. Environ Sci Pollut Res doi 25:11192–11204. https://doi.org/10.1007/s11356-017-8447-1
Chen LIS, Cheng L (2003) Carbon assimilation and carbohydrate metabolism of 'Concord' grape (Vitis labrusca L.) leaves in response to nitrogen supply. J Am Soc Hortic Sci 128:754–760
Duan Y, Yao Y, Wang B, Han L, Wang L, Sun H, Chen L (2018) Association of urinary concentrations of bisphenols with type 2 diabetes mellitus: a case-control study. Environ Pollut 243:1719–1726
EFSA (2015) Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs: executive summary. EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids CEF. EFSA J 13((1):3978 https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2015.3978. Accessed 02 Nov. 2020
Errico S, Nicolucci C, Migliaccio M, Micale V, Mita DG, Diano N (2017) Analysis and occurrence of some phenol endocrine disruptors in two marine sites of the northern coast of Sicily (Italy). Mar Pollut Bull 120:68–74
Fekete-Kertész I, Kunglné-Nagy Z, Gruiz K, Magyar Á, Farkas É, Molnár M (2015) Assessing toxicity of organic aquatic micropollutants based on the total chlorophyll content of Lemna minor as a sensitive endpoint. Period Polytech Chem Eng 59:262–271. https://doi.org/10.3311/PPch.8077
Goldschmidt EE, Huber SC (1992) Regulation of photosynthesis by end-product accumulation in leaves of plants storing starch, sucrose and hexose sugars. Plant Physiol 99:1443–1448
Gorini F, Bustaffa E, Coi A, Iervasi G, Bianchi F (2020) Bisphenols as environmental triggers of thyroid dysfunction: clues and evidence. Int J Environ Res Public Health 17:2654. https://doi.org/10.3390/ijerph17082654
Han C, Hong YC (2016) Bisphenol A, hypertension, and cardiovascular diseases: epidemiological, laboratory, and clinical trial evidence. Curr Hypertens Rep 18:11
Hu H, Wang L, Wang Q, Jiao L, Hua W, Zhou Q, Huang X (2014) Photosynthesis, chlorophyll fluorescence characteristics, and chlorophyll content of soybean seedlings under combined stress of bisphenol A and cadmium. Environ Toxicol Chem 33:2455–2462. https://doi.org/10.1002/etc.2720
Huang YQ, Wong CKC, Zheng JS, Bouwman H, Barra R, Wahlström B, Neretin L, Wong MH (2012) Bisphenol A (BPA) in China: a review of sources, environmental levels, and potential human health impacts. Environ Int 42:91–99
ISO 20079 (2005) Water quality - Determination of the toxic effect of water constituents and waste water on duckweed (Lemna minor) - duckweed growth inhibition test. Switzerland, Geneva
Jiao L, Ding H, Wang L, Zhou Q, Huang X (2015) Effects of bisphenol a on chlorophyll synthesis in soybean seedlings. Environ Sci Pollut Res 22:5877–5886
Kim D, Kwak JI, An YJ (2018) Effects of bisphenol A in soil on growth, photosynthesis activity, and genistein levels in crop plants (Vigna radiata). Chemosphere 209:875–882
Li Y, Zhu G, Ng WJ, Tan SK (2014) A review on removing pharmaceutical contaminants from wastewater by constructed wetlands: design, performance and mechanism. Sci Total Environ 468:908–932
Li X, Wang L, Wang S, Yang Q, Zhou Q, Huang X (2018a) A preliminary analysis of the effects of bisphenol A on the plant root growth via changes in endogenous plant hormones. Ecotoxicol Environ Saf 150:152–158
Li YT, Liang Y, Li YN, Che XK, Zhao SJ, Zhang ZS, Gao HY (2018b) Mechanisms by which Bisphenol A affect the photosynthetic apparatus in cucumber (Cucumis sativus L.) leaves. Sci Rep. https://doi.org/10.1038/s41598-018-22486-4
Li X, Wu S, Yang C, Zeng G (2020) Microalgal and duckweed based constructed wetlands for swine wastewater treatment: a review. Bioresour Technol 318(4):123858. https://doi.org/10.1016/j.biortech.2020.123858
Lichtenthaler HK (1987) Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Lobiuc A, Olaru S, Hancu EI, Costica N, Fortuna ME, Zamfirache MM, Constantinescu G (2018) Toxicity and removal of direct red 28 diazo dye in living polymeric systems. Rev Chim -Bucharest 69:1628e1635
Malea P, Kokkinidi D, Kevrekidou A, Adamakis IS (2020) Environmentally relevant bisphenol A concentrations effects on the seagrass Cymodocea nodosa different parts elongation: perceptive assessors of toxicity. Environ Sci Pollut Res 27:7267–7279. https://doi.org/10.1007/s11356-019-07443-6
Mihaich EM, Friederich U, Caspers N, Hall AT, Klecka GM, Dimond SS, Staples CA, Ortego LS, Hentges SG (2009) Acute and chronic toxicity testing of bisphenol A with aquatic invertebrates and plants. Ecotox Environ Safe 72:1392–1399
Mínguez-Alarcón L, Hauser R, Gaskins AJ (2016) Effects of bisphenol A on male and couple reproductive health: a review. Fertil Steril 106:864–870
Mkandawire M, Dudel EG (2005) Assignment of Lemna gibba L. (duckweed) bioassay for in situ ecotoxicity assessment. Aquat Ecol 39:151–165
Mkandawire M, Dudel EG (2007) Are Lemna spp. Effective phytoremediation agents? Biorem Biodiv Bioavail 1:56–71
Nerín C, Aznar M, Carrizo D (2016) Food contamination during food process. Trends Food Sci Technol 48:63–68. https://doi.org/10.1016/j.tifs.2015.12.004
Pezeshki SR (2001) Wetland plant responses to soil flooding. Environ Exp Bot 46:299–312
Rapala M, Plucinski B, Jedynak P (2017) The effect of bisphenol A on growth, pigment composition and photosystem II activity of Arabidopsis thaliana. Acta Biochim Pol 64:407–413. https://doi.org/10.18388/abp.2017_1626
Regulation Commission 2018/213 (2018) on the use of bisphenol A in varnishes and coatings intended to come into contact with food and amending Regulation (EU) No 10/2011 as regards the use of that substance in plastic food contact materials. http://data.europa.eu/eli/reg/2018/213/oj. Accessed 2 Nov 2020
Research and Markets (2020) The Global Bisphenol A Market. https://www.researchandmarkets.com/research/hl86rz/global_bisphenol?w=5. Accessed 27 Oct. 2020
Ribeiro E, Ladeira C, Viegas S (2017) Occupational exposure to bisphenol A (BPA): A reality that still needs to be unveiled. Toxics 5(3):22. https://doi.org/10.3390/toxics5030022
Ruban AV (2016) Nonphotochemical chlorophyll fluorescence quenching: mechanism and effectiveness in protecting plants from photodamage. Plant Physiol 170:1903–1916
Sharma R, Kumari A, Rajput S, Nishu AS, Rampal R, Kaur R (2019) Accumulation, morpho-physiological and oxidative stress induction by single and binary treatments of fluoride and low molecular weight phthalates in Spirodela polyrhiza L. Schleiden Sci Rep 9:20006. https://doi.org/10.1038/s41598-019-56110-w
Sree KS, Keresztes A, Mueller-Roeber B, Brandt R, Eberius M, Fischer W, Appenroth KJ (2015) Phytotoxicity of cobalt ions on the duckweed Lemna minor morphology, ion uptake, and starch accumulation. Chemosphere 131:149–156
Üçüncü E, Tunca E, Fikirdeşici S, Özkan AD, Altındağ A (2013) Phytoremediation of Cu, Cr and Pb mixtures by Lemna minor. Bull Environ Contam Toxicol 91:600–604
Yamamoto T, Yasuhara A, Shiraishi H, Nakasugi O (2001) Bisphenol A in hazardous waste landfill leachates. Chemosphere 42:415–418
Zhang J, Wang L, Li M, Jiao L, Zhou Q, Huang X (2015) Effects of bisphenol A on chlorophyll fluorescence in five plants. Environ Sci Pollut Res 22:17724–17732. https://doi.org/10.1007/s11356-015-5003-8
Acknowledgements
The authors would like to thank Dr. Hervé Grisey for the helpful and fruitful discussion and the SICTOM (Syndicat Intercommunal de Collecte et de Traitement des Ordures Ménagères, Etueffont), for the financial help.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Funding
The authors would like to acknowledge the SICTOM for the financial help.
Author information
Authors and Affiliations
Contributions
BP and AE: Conceptualization, methodology, acquisition of data, and writing-original draft
ASL, HG, ASB, and BM: Supervision, resources, and project coordination
BM: Writing-review and editing
All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bourgeade, ., Aleya, E., Alaoui-Sosse, L. et al. Growth, pigment changes, and photosystem II activity in the aquatic macrophyte Lemna minor exposed to bisphenol A. Environ Sci Pollut Res 28, 68671–68678 (2021). https://doi.org/10.1007/s11356-021-15422-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15422-z