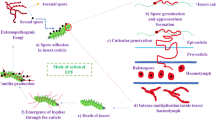
Abstract
There is a worldwide concern to achieve food security with a sustainable approach, including the generation and implementation of techniques for the production of high-quality chemical-free crops. This food revolution has promoted the development and consolidation of programmes for integrated pest management. Some of those programmes include the use of diverse organisms (biological control agents) to suppress populations of pests potentially harmful to the crops. Among these biological control agents are entomopathogenic fungi that are highly effective in suppressing a diversity of insects and have, therefore, been produced and marketed throughout the world. However, the bottleneck for applying entomopathogenic fungi is the production of propagules (blastospores and conidia) with resistance to environment conditions and abiotic factors, maintaining high quality in terms of virulence. Therefore, this manuscript presents recent studies related to increasing resistance and quality using different bioreactors to produce conidia. The above presents a global panorama related to current developments that contribute to improving the resistance, quality, and production of entomopathogenic fungal propagules.
Similar content being viewed by others
References
Abdel-Raheem MA, Reyad N, Alghandi H (2020) Virulence of nano – particle preparation of entomopathogenic fungi and entomopathogenic bacteria against red palm weevil Rhynchophorus ferrugineus (Olivier) (Coleoptera: Curculionidae). Rom Biotechnol Lett 25:1151–1159. https://doi.org/10.25083/rbl/25.1/1151.1159
Alcantara-Vargas E, Espitia-López J, Garza-López PM, Angel-Cuapio A (2020) Producción y calidad de conidios de cepas de entomopatógenos del género Metarhizium anisopliae, aislados en zonas agrícolas del Estado de México. Rev Mex Biodivers 91:912912. https://doi.org/10.22201/ib.20078706e.2020.91.2912
Arredondo-Bernal HC, González-Cabrera J (2020) El comercio del control biológico en México. In: Arredondo-Bernal HC, Tamayo-Mejía F, Rodriguez-del-Bosque L (eds) Fundamento y práctica del control biológico de plagas y enfermedades, 1st edn. bba, México, pp 599–629
Arthurs S, Thomas MB (2001) Effects of temperature and relative humidity on sporulation of Metarhizium anisopliae var. acridum in mycosed cadavers of Schistocerca gregaria. J Invertebr Pathol 78:59–65. https://doi.org/10.1006/jipa.2001.5050
Barrera JF (2020) Concepto, alcances e implicaciones del control biológico. In: Arredondo-Bernal HC, Tamayo-Mejia F, Rodriguez-del-Bosque LA (eds) Fundamento y práctica del control biológico de plagas y enfermedades, 1st edn. Colegio de Posgraduados, Texcoco, pp 15–40
Benenati RF, Brosilow CB (1962) Void fraction distribution in beds of spheres. AIChE J 8:359–361. https://doi.org/10.1002/aic.690080319
Birnbaum N, Reingold V, Matveev S et al (2021) Not only a formulation: The effects of pickering emulsion on the entomopathogenic action of Metarhizium brunneum. J Fungi 7:499. https://doi.org/10.3390/jof7070499
Biryol S, Güney E, Eski A et al (2021) Development of mycoinsecticide formulations with Beauveria bassiana and Metarhizium brunneum for the control of Orosanga japonica (Hemiptera: Ricaniidae). Ann Appl Biol 179:319–330. https://doi.org/10.1111/aab.12699
Brand D, Soccol CR, Sabu A, Roussos S (2010) Production of fungal biological control agents through solid state fermentation: a case study on Paecelomyces lilacinus againts root-knot nematodes. Micol Apl Int 22:31–48.
Butt TM, Coates CJ, Dubovskiy IM, Ratcliffe NA (2016) Entomopathogenic fungi: new insights into host–pathogen interactions. Adv Genet 94:307–364. https://doi.org/10.1016/bs.adgen.2016.01.006
Carboué Q, Rébufa C, Dupuy N et al (2019) Solid state fermentation pilot-scaled plug flow bioreactor, using partial least square regression to predict the residence time in a semicontinuous process. Biochem Eng J 149:107248. https://doi.org/10.1016/j.bej.2019.107248
Casciatori FP, Thoméo JC (2018) Heat transfer in packed-beds of agricultural waste with low rates of air flow applicable to solid-state fermentation. Chem Eng Sci 188:97–111. https://doi.org/10.1016/j.ces.2018.05.024
Castillo-Minjarez JM, Garza-López PM, Barrios-González J, Loera O (2019) Free conidia of entomopathogenic fungi modify quality traits by changing glutathione levels after an oxidant stimulus. Biol Control 137:104011. https://doi.org/10.1016/j.biocontrol.2019.104011
Chantasingh D, Kitikhun S, Keyhani NO et al (2013) Identification of catalase as an early up-regulated gene in Beauveria bassiana and its role in entomopathogenic fungal virulence. Biol Control 67:85–93. https://doi.org/10.1016/j.biocontrol.2013.08.004
Chen X, Li L, Hu Q et al (2015) Expression of dsRNA in recombinant Isaria fumosorosea strain targets the TLR7 gene in Bemisia tabaci. BMC Biotechnol 15:64. https://doi.org/10.1186/s12896-015-0170-8
Cherry AJ, Jenkins NE, Heviefo G et al (1999) Operational and economic analysis of a west African pilot-scale production plant for aerial conidia of Metarhizium spp. for use as a mycoinsecticide against locusts and grasshoppers. Biocontrol Sci Technol 9:35–51. https://doi.org/10.1080/09583159929893
Comoé H, Siegrist M (2015) Relevant drivers of farmers’ decision behavior regarding their adaptation to climate change: a case study of two regions in Côte d’Ivoire. Mitig Adapt Strateg Glob Chang 20:179–199. https://doi.org/10.1007/s11027-013-9486-7
da Cunha LP, Casciatori FP, de Cenço Lopes I, Thoméo JC (2019) Production of conidia of the entomopathogenic fungus Metarhizium anisopliae ICB 425 in a tray bioreactor. Bioprocess Biosyst Eng 42:1757–1768. https://doi.org/10.1007/s00449-019-02172-z
da Cunha LP, Casciatori FP, Vicente IV et al (2020) Metarhizium anisopliae conidia production in packed-bed bioreactor using rice as substrate in successive cultivations. Process Biochem 97:104–111. https://doi.org/10.1016/j.procbio.2020.07.002
Dalla Santa HS, Sousa NJ, Brand D et al (2004) Conidia production of Beauveria sp. by solid-state fermentation for biocontrol of Ilex paraguariensis Caterpillars. Folia Microbiol (Praha) 49:418–422. https://doi.org/10.1007/BF02931603
De Vrije T, Antoine N, Buitelaar RM et al (2001) The fungal biocontrol agent Coniothyrium minitans: Production by solid-state fermentation, application and marketing. Appl Microbiol Biotechnol 56:58–68. https://doi.org/10.1007/s002530100678
Durand A (2003) Bioreactor designs for solid state fermentation. Biochem Eng J 13:113–125
Fan Y, Pei X, Guo S et al (2010) Increased virulence using engineered protease-chitin binding domain hybrid expressed in the entomopathogenic fungus Beauveria bassiana. Microb Pathog 49:376–380. https://doi.org/10.1016/j.micpath.2010.06.013
Fan Y, Borovsky D, Hawkings C et al (2012a) Exploiting host molecules to augment mycoinsecticide virulence. Nat Biotechnol 30:35–37. https://doi.org/10.1038/nbt.2080
Fan Y, Pereira RM, Kilic E et al (2012b) Pyrokinin β-neuropeptide affects necrophoretic behavior in fire nnts (S. invicta), and expression of β-NP in a mycoinsecticide increases its virulence. PLoS ONE 7:e26924. https://doi.org/10.1371/journal.pone.0026924
Fang W, Leng B, **ao Y et al (2005) Cloning of Beauveria bassiana chitinase gene Bbchit1 and its application to improve fungal strain virulence. Appl Environ Microbiol 71:363–370. https://doi.org/10.1128/AEM.71.1.363-370.2005
Fang W, Feng J, Fan Y et al (2009) Expressing a fusion protein with protease and chitinase activities increases the virulence of the insect pathogen Beauveria bassiana. J Invertebr Pathol 102:155–159. https://doi.org/10.1016/j.jip.2009.07.013
Fang W, St. Leger RJ (2012) Enhanced UV resistance and improved killing of malaria mosquitoes by photolyase transgenic entomopathogenic fungi. PLoS ONE 7:e43069. https://doi.org/10.1371/journal.pone.0043069
García Riaño JL, Gómez-Valderrama JA, Araque- Echeverry GA et al (2021) Insecticidal activity and persistance of formulation a base Beauveria bassiana for Diatraea saccharalis control. Chil J Agric Anim Sci 37:257–269. https://doi.org/10.29393/CHJAAS37-27AIJL50027
Gutiérrez-Ramirez A, Robles-Bermúdez A, Santillan-Ortega C et al (2013) Control biológico como herramienta sustentable en el manejo de plagas y su uso en el estado de Nayarit, México. Rev Biociencias 2:102–112
Ibrahim L, Butt TM, Beckett A, Clark SJ (1999) The germination of oil-formulated conidia of the insect pathogen, Metarhizium anisopliae. Mycol Res 103:901–907. https://doi.org/10.1017/S0953756298007849
Jaronski ST (2014) Mass production of entomopathogenic fungi: State of the art. In: Morales-Ramos JA, Rojas GM, Shapiro-Ilan DI (eds) Mass Production of Beneficial Organisms. Elsevier, pp 357–413
Jenkins NE, Grzywacz D (2000) Quality control of fungal and viral biocontrol agents - Assurance of product performance. Biocontrol Sci Technol 10:753–777. https://doi.org/10.1080/09583150020011717
Kamp AM, Bidochka MJ (2002) Conidium production by insect pathogenic fungi on commercially available agars. Lett Appl Microbiol 35:74–77. https://doi.org/10.1046/j.1472-765X.2002.01128.x
Lee JY, Kang SW, Yoon CS et al (2006) Verticillium lecanii spore formulation using UV protectant and wetting agent and the biocontrol of cotton aphids. Biotechnol Lett 28:1041–1045. https://doi.org/10.1007/s10529-006-9036-4
Lewandowski I (ed) (2018) Bioeconomy. Springer International Publishing, Cham
Liao X, Lu H-L, Fang W, St. Leger RJ (2014) Overexpression of a Metarhizium robertsii HSP25 gene increases thermotolerance and survival in soil. Appl Microbiol Biotechnol 98:777–783. https://doi.org/10.1007/s00253-013-5360-5
Liu Y-J, Liu J, Ying S-H et al (2013) A fungal insecticide engineered for fast per os killing of caterpillars has high field efficacy and safety in full-Season control of cabbage insect pests. Appl Environ Microbiol 79:6452–6458. https://doi.org/10.1128/AEM.01594-13
Lopes-Perez C, Casciatori FP, Thoméo JC (2019) Strategies for scaling-up packed-bed bioreactors for solid-state fermentation: The case of cellulolytic enzymes production by a thermophilic fungus. Chem Eng J 361:1142–1151. https://doi.org/10.1016/j.cej.2018.12.169
Lopes-Perez C, Casciatori FP, Thoméo JC (2021) Improving enzyme production by solid-state cultivation in packed-bed bioreactors by changing bed porosity and airflow distribution. Bioprocess Biosyst Eng 44:537–548. https://doi.org/10.1007/s00449-020-02466-7
Lovett B, St. Leger RJ (2015) Stress is the rule rather than the exception for Metarhizium. Curr Genet 61:253–261. https://doi.org/10.1007/s00294-014-0447-9
Maïga Y, Carboué Q, Hamrouni R et al (2021) Development and Evaluation of a Disposable Solid-State Culture Packed-Bed Bioreactor for the Production of Conidia from Trichoderma asperellum Grown Under Water Stress. Waste Biomass Valoriz 12:3223–3231. https://doi.org/10.1007/s12649-020-01210-2
Marciano AF, Mascarin GM, Franco RFF et al (2021) Innovative granular formulation of Metarhizium robertsii microsclerotia and blastospores for cattle tick control. Sci Rep 11:4972. https://doi.org/10.1038/s41598-021-84142-8
Matthiessen P, Sheahan D, Harrison R et al (1995) Use of a Gammarus pulex bioassay to measure the effects of transient carbofuran runoff from farmland. Ecotoxicol Environ Saf 30:111–119. https://doi.org/10.1006/eesa.1995.1013
Méndez-González F, Loera O, Favela-Torres E (2018b) Conidia production of Metarhizium anisopliae in bags and packed column bioreactors. Curr Biotechnol 7:65–69. https://doi.org/10.2174/2211550105666160926123350
Méndez-González F, Loera-Corral O, Saucedo-Castañeda G, Favela-Torres E (2018a) Bioreactors for the production of biological control agents produced by solid-state fermentation. In: Pandey A, Larroche C, Soccol CR (eds) Current Developments in Biotechnology and Bioengineering. Elsevier, pp 109–121
Méndez-González F, Loera O, Saucedo-Castañeda G, Favela-Torres E (2020) Forced aeration promotes high production and productivity of infective conidia from Metarhizium robertsii in solid-state fermentation. Biochem Eng J 156:107492. https://doi.org/10.1016/j.bej.2020.107492
Méndez-González F, Figueroa‐Montero A, Saucedo‐Castañeda G et al (2021) Addition of spherical‐style packing improves the production of conidia by Metarhizium robertsii in packed column bioreactors. J Chem Technol Biotechnol. https://doi.org/10.1002/jctb.6993
Miranda-Hernández F, Saucedo-Castañeda G, Alatorre-Rosas R, Loera O (2014) Oxygen-rich culture conditions enhance the conidial infectivity and the quality of two strains of Isaria fumosorosea for potentially improved biocontrol processes. Pest Manag Sci 70:661–666. https://doi.org/10.1002/ps.3605
Miranda-Hernández F, Garza-López PM, Loera O (2016) Cellular signaling in cross protection: An alternative to improve mycopesticides. Biol Control 103:196–203. https://doi.org/10.1016/j.biocontrol.2016.09.007
Mondal S, Baksi S, Koris A, Vatai G (2016) Journey of enzymes in entomopathogenic fungi. Pac Sci Rev Nat Sci Eng 18:85–99. https://doi.org/10.1016/j.psra.2016.10.001
Muñiz-Paredes F, Miranda-Hernández F, Loera O (2017) Production of conidia by entomopathogenic fungi: from inoculants to final quality tests. World J Microbiol Biotechnol 33:57–66. https://doi.org/10.1007/s11274-017-2229-2
Oliveira AS, Braga GUL, Rangel DEN (2018) Metarhizium robertsii illuminated during mycelial growth produces conidia with increased germination speed and virulence. Fungal Biol 122:555–562. https://doi.org/10.1016/j.funbio.2017.12.009
Ortiz-Urquiza A, Keyhani NO (2015) Stress response signaling and virulence: insights from entomopathogenic fungi. Curr Genet 61:239–249. https://doi.org/10.1007/s00294-014-0439-9
Paixão FRS, Fernandes ÉKK, Pedrini N (2019) Thermotolerance of fungal conidia. In: Khan M, Ahmad W (eds) Microbes for Sustainable Insect Pest Management. Springer, Cham, pp 185–196
Posada-Flórez FJ (2008) Production of Beauveria bassiana fungal spores on rice to control the coffee berry borer, Hypothenemus hampei, in Colombia. J Insect Sci 8:1–13. https://doi.org/10.1673/031.008.4101
Qiu L, Wang J-J, Chu Z-J et al (2014) Phytochrome controls conidiation in response to red/far-red light and daylight length and regulates multistress tolerance in B eauveria bassiana. Environ Microbiol 16:2316–2328. https://doi.org/10.1111/1462-2920.12486
Qiu L, Wei X-Y, Wang S-J, Wang J-J (2020) Characterization of trehalose-6-phosphate phosphatase in trehalose biosynthesis, asexual development, stress resistance and virulence of an insect mycopathogen. Pestic Biochem Physiol 163:185–192. https://doi.org/10.1016/j.pestbp.2019.11.016
Ramírez-Guzmán NK, Torres-León C, Martínez-Terrazas E et al (2018) Biocontrol as an efficient tool for food control and biosecurity. In: Grumezescu AM, Holban AM (eds) Food Safety and Preservation, 1st edn. Academic Press, pp 167–193
Rangel DEN, Alston DG, Roberts DW (2008a) Effects of physical and nutritional stress conditions during mycelial growth on conidial germination speed, adhesion to host cuticle, and virulence of Metarhizium anisopliae, an entomopathogenic fungus. Mycol Res 112:1355–1361. https://doi.org/10.1016/j.mycres.2008.04.011
Rangel DEN, Anderson AJ, Roberts DW (2008b) Evaluating physical and nutritional stress during mycelial growth as inducers of tolerance to heat and UV-B radiation in Metarhizium anisopliae conidia. Mycol Res 112:1362–1372. https://doi.org/10.1016/j.mycres.2008.04.013
Rangel DEN (2011) Stress induced cross-protection against environmental challenges on prokaryotic and eukaryotic microbes. World J Microbiol Biotechnol 27:1281–1296. https://doi.org/10.1007/s11274-010-0584-3
Rangel DEN, Braga GUL, Fernandes ÉKK et al (2015) Stress tolerance and virulence of insect-pathogenic fungi are determined by environmental conditions during conidial formation. Curr Genet 61:383–404. https://doi.org/10.1007/s00294-015-0477-y
Rangel DEN, Finlay RD, Hallsworth JE et al (2018) Fungal strategies for dealing with environment- and agriculture-induced stresses. Fungal Biol 122:602–612. https://doi.org/10.1016/j.funbio.2018.02.002
Roberts DW, St. Leger RJ (2004) Metarhizium spp., cosmopolitan insect-pathogenic fungi: mycological aspects. In: Laskin A, Bennett J, Gadd G (eds) Advances in Applied Microbiology. Academic Press, pp 1–70
Rodriguez-Gomez D, Garate-Velez L, Morales-Piña AL et al (2020) Increase in the mortality of the entomopathogenic fungus Metarhizium anisopliae due to the application of an electric field during conidiation. Rev Mex Ing Química 19:123–133. https://doi.org/10.24275/rmiq/Bio1601
Rodriguez-Romero J, Hedtke M, Kastner C et al (2010) Fungi, hidden in soil or up in the air: light makes a difference. Annu Rev Microbiol 64:585–610. https://doi.org/10.1146/annurev.micro.112408.134000
Sala A, Barrena R, Artola A, Sánchez A (2019) Current developments in the production of fungal biological control agents by solid-state fermentation using organic solid waste. Crit Rev Environ Sci Technol 49:655–694. https://doi.org/10.1080/10643389.2018.1557497
Sekhar KR, Spitz DR, Harris S et al (2002) Redox-sensitive interaction between KIAA0132 and Nrf2 mediates indomethacin-induced expression of γ-glutamylcysteine synthetase. Free Radic Biol Med 32:650–662. https://doi.org/10.1016/S0891-5849(02)00755-4
Shang Y, Duan Z, Huang W et al (2012) Improving UV resistance and virulence of Beauveria bassiana by genetic engineering with an exogenous tyrosinase gene. J Invertebr Pathol 109:105–109. https://doi.org/10.1016/j.jip.2011.10.004
St Leger R, Joshi L, Bidochka MJ, Roberts DW (1996) Construction of an improved mycoinsecticide overexpressing a toxic protease. Proc Natl Acad Sci 93:6349–6354. https://doi.org/10.1073/pnas.93.13.6349
Suárez-Vergel G, Figueroa-Martinez F, Garza-López PM et al (2021) DOPA-melanin, component and tolerance factor to heat and UV-B radiation in the conidia of two species of Cordyceps. Biocontrol Sci Technol 1–18. https://doi.org/10.1080/09583157.2021.2016626
Sutanto KD, Husain M, Rasool KG et al (2022) Persistency of indigenous and exotic entomopathogenic fungi isolates under ultraviolet B (UV-B) irradiation to enhance field application efficacy and obtain sustainable control of the red palm weevil. Insects 13:103. https://doi.org/10.3390/insects13010103
Tamayo-Mejía F, Alatorre-Rosas R, Delgado-Fernández S, Ocampo-Hernández JA (2020) Principios de aplicación de entomopatógenos. In: Arredondo-Bernal HC, Tamayo-Mejía F, Rodríguez-del-Bosque LA (eds) Fundamento y práctica del control biológico de plagas y enfermedades, 1st edn. Colegio de Posgraduados, Texcoco, pp 405–426
Tong S-M, Zhang A-X, Guo C-T et al (2018) Daylight length-dependent translocation of VIVID photoreceptor in cells and its essential role in conidiation and virulence of Beauveria bassiana. Environ Microbiol 20:169–185. https://doi.org/10.1111/1462-2920.13951
Tong S-M, Feng M-G (2020) Phenotypic and molecular insights into heat tolerance of formulated cells as active ingredients of fungal insecticides. Appl Microbiol Biotechnol 104:5711–5724. https://doi.org/10.1007/s00253-020-10659-z
Tong S, Feng M (2022) Molecular basis and regulatory mechanisms underlying fungal insecticides’ resistance to solar ultraviolet irradiation. Pest Manag Sci 78:30–42. https://doi.org/10.1002/ps.6600
Tseng MN, Chung PC, Tzean SS (2011) Enhancing the stress tolerance and virulence of an entomopathogen by metabolic engineering of dihydroxynaphthalene melanin biosynthesis genes. Appl Environ Microbiol 77:4508–4519. https://doi.org/10.1128/AEM.02033-10
Van Breukelen FR, Haemers S, Wijffels RH, Rinzema A (2011) Bioreactor and substrate selection for solid-state cultivation of the malaria mosquito control agent Metarhizium anisopliae. Process Biochem 46:751–757. https://doi.org/10.1016/j.procbio.2010.11.023
Wang C, St Leger RJ (2007) A scorpion neurotoxin increases the potency of a fungal insecticide. Nat Biotechnol 25:1455–1456. https://doi.org/10.1038/nbt1357
Wang S, Fang W, Wang C, St. Leger RJ (2011) Insertion of an esterase gene into a specific locust pathogen (Metarhizium acridum) enables it to infect caterpillars. PLoS Pathog 7:e1002097. https://doi.org/10.1371/journal.ppat.1002097
Wang D-Y, Fu B, Tong S-M et al (2019) Two photolyases repair distinct DNA lesions and reactivate UVB-inactivated conidia of an insect mycopathogen under visible light. Appl Environ Microbiol 85. https://doi.org/10.1128/AEM.02459-18
Wang J-J, Yin Y-P, Song J-Z et al (2021) A p53-like transcription factor, BbTFO1, contributes to virulence and oxidative and thermal stress tolerances in the insect pathogenic fungus, Beauveria bassiana. PLoS ONE 16:e0249350. https://doi.org/10.1371/journal.pone.0249350
Wraight SP, Jackson MA, Kokc SL (2001) Production, stabilization and formulation of fungal biocontrol agents. Fungi as biocontrol agents: progress, problems and potential. CABI, Wallingford, pp 253–287
**e X-Q, Wang J, Huang B-F et al (2010) A new manganese superoxide dismutase identified from Beauveria bassiana enhances virulence and stress tolerance when overexpressed in the fungal pathogen. Appl Microbiol Biotechnol 86:1543–1553. https://doi.org/10.1007/s00253-010-2437-2
**e L, Chen HM, Yang J Bin (2012) Conidia production by Beauveria bassiana on rice in solids-state fermentation using tray bioreactor. Adv Mater Res 610–613:3478–3482. https://doi.org/10.4028/www.scientific.net/AMR.610-613.3478
Yaakov N, Ananth Mani K, Felfbaum R et al (2018) Single cell encapsulation via pickering emulsion for biopesticide applications. ACS Omega 3:14294–14301. https://doi.org/10.1021/acsomega.8b02225
Yang L, Keyhani NO, Tang G et al (2014) Expression of a toll signaling regulator serpin in a mycoinsecticide for increased virulence. Appl Environ Microbiol 80:4531–4539. https://doi.org/10.1128/AEM.01197-14
Ye SD, Ying SH, Chen C, Feng MG (2006) New solid-state fermentation chamber for bulk production of aerial conidia of fungal biocontrol agents on rice. Biotechnol Lett 28:799–804. https://doi.org/10.1007/s10529-006-9004-z
Ying SH, Feng MG (2011) Integration of Escherichia coli thioredoxin (trxA) into Beauveria bassiana enhances the fungal tolerance to the stresses of oxidation, heat and UV-B irradiation. Biol Control 59:255–260. https://doi.org/10.1016/j.biocontrol.2011.07.005
Zhang YJ, Feng MG, Fan YH et al (2008) A cuticle-degrading protease (CDEP-1) of Beauveria bassiana enhances virulence. Biocontrol Sci Technol 18:543–555. https://doi.org/10.1080/09583150802082239
Zhang Y, Zhao J, Fang W et al (2009) Mitogen-activated protein kinase hog1 in the entomopathogenic fungus Beauveria bassiana regulates environmental stress responses and virulence to insects. Appl Environ Microbiol 75:3787–3795. https://doi.org/10.1128/AEM.01913-08
Author information
Authors and Affiliations
Contributions
FMG and JMCM wrote the manuscript with support from OL and EFT. All authors discussed and contributed to the final version of the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Méndez-González, F., Castillo-Minjarez, J.M., Loera, O. et al. Current developments in the resistance, quality, and production of entomopathogenic fungi. World J Microbiol Biotechnol 38, 115 (2022). https://doi.org/10.1007/s11274-022-03301-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03301-9