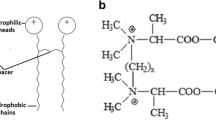
Abstract
Quaternary ammonium salts (QASs) are ubiquitous in nature, being found in organisms ranging from microorganisms to vertebrates (e.g., glycine betaine, carnitine) where they have important cellular functions. QASs are also obtained by chemical synthesis. These compounds, due to their diverse chemical structure (e.g. monomeric QAS or gemini) and their biological properties, are widely used in medicine (as disinfectants, drugs, and DNA carriers), industry, environmental protection and agriculture (as preservatives, biocides, herbicides and fungicides). Discussed chemical compounds reduce the adhesion of microorganisms to various biotic and abiotic surfaces and cause the eradication of biofilms produced by pathogenic microorganisms. The properties of these chemicals depend on their chemical structure (length of the alkyl chain, linker and counterion), which has a direct impact on the physicochemical and biological activity of these compounds. QASs by incorporation into the membranes, inhibit the activity of proteins (H+-ATPase) and disrupt the transport of substances to the cell. Moreover, in the presence of QASs, changes in lipid composition (qualitative and quantitative) of plasma membrane are observed. The widespread use of disinfectants in commercial products can induce resistance in microorganisms to these surfactants and even to antibiotics. In this article we discuss the biological activity of QASs as cationic surfactants against microorganisms and their resistance to these compounds.



Similar content being viewed by others
References
Ahn Y et al (2016) Intrinsic resistance of Burkholderia cepacia complex to benzalkonium chloride. mBio 7:e01716. https://doi.org/10.1128/mBio.01716-16
Al-Dulaymi M, Mohammed-Saeid W, El-Aneed A, Badea I (2019) Peptide-modified gemini surfactants: preparation and characterization for gene delivery. Methods Mol Biol (Clifton, NJ) 2000:203–225. https://doi.org/10.1007/978-1-4939-9516-5_14
Amsalu A et al (2020) Efflux pump-driven antibiotic and biocide cross-resistance in Pseudomonas aeruginosa isolated from different ecological niches: a case study in the development of multidrug resistance in environmental hotspots. Microorganisms 8:1647. https://doi.org/10.3390/microorganisms8111647
Babaei M, Sulong A, Hamat R, Nordin S, Neela V (2015) Extremely high prevalence of antiseptic resistant quaternary ammonium compound E gene among clinical isolates of multiple drug resistant Acinetobacter baumannii in Malaysia. Ann Clin Microbiol Antimicrob 14:11. https://doi.org/10.1186/s12941-015-0071-7
Baigelman W, Chodosh S (1977) Bronchodilator action of the anticholinergic drug, ipratropium bromide (Sch 1000), as an aerosol in chronic bronchitis and asthma. Chest 71:324–328. https://doi.org/10.1378/chest.71.3.324
Baker N, Williams A, Tropsha A, Ekins S (2020) Repurposing quaternary ammonium compounds as potential treatments for COVID-19. Pharm Res 37:104. https://doi.org/10.1007/s11095-020-02842-8
Block S (1991) Disinfection, sterilization, and preservation. Lea & Febiger, Philadelphia
Boruń A (2019) Conductance and ionic association of selected imidazolium ionic liquids in various solvents: a review. J Mol Liq 276:214–224. https://doi.org/10.1016/j.molliq.2018.11.140
Buck J, Grossen P, Cullis PR, Huwyler J, Witzigmann D (2019) Lipid-based DNA therapeutics: hallmarks of non-viral gene delivery. ACS Nano 13:3754–3782. https://doi.org/10.1021/acsnano.8b07858
Buffet-Bataillon S, Le Jeune A, Le Gall-David S, Bonnaure-Mallet M, Jolivet-Gougeon A (2012) Molecular mechanisms of higher MICs of antibiotics and quaternary ammonium compounds for Escherichia coli isolated from bacteraemia. J Antimicrob Chemother 67:2837–2842. https://doi.org/10.1093/jac/dks321
Calabrese V, Giuffrida Stella A, Calvani M, Butterfield D (2006) Acetylcarnitine and cellular stress response: roles in nutritional redox homeostasis and regulation of longevity genes. J Nutr Biochem 17:73–88. https://doi.org/10.1016/j.jnutbio.2005.03.027
Capita R, Vicente-Velasco M, Rodriguez-Melcon C, Garcia-Fernandez C, Carballo J, Alonso-Calleja C (2019) Effect of low doses of biocides on the antimicrobial resistance and the biofilms of Cronobacter sakazakii and Yersinia enterocolitica. Sci Rep 9:15905. https://doi.org/10.1038/s41598-019-51907-1
Cheng L, Zhang K, Weir M, Liu H, Zhou X, Xu H (2013) Effects of antibacterial primers with quaternary ammonium and nano-silver on Streptococcus mutans impregnated in human dentin blocks. Dent Mater 29:462–472. https://doi.org/10.1016/j.dental.2013.01.011
Cheng F, Zhou Y, Wang M, Guo C, Cao Z, Zhang R, Peng C (2020) A review of pharmacological and pharmacokinetic properties of stachydrine. Pharmacol Res 155:104755. https://doi.org/10.1016/j.phrs.2020.104755
Clancy M, Halford S, Walls R, Murphy M (2001) In patients with head injuries who undergo rapid sequence intubation using succinylcholine, does pretreatment with a competitive neuromuscular blocking agent improve outcome? A literature review. Emerg Med J 18:373–375. https://doi.org/10.1136/emj.18.5.373
Colley C, Zwaal R, Roelofsen B, van Deenen L (1973) Lytic and non-lytic degradation of phospholipids in mammalian erythrocytes by pure phospholipases. Biochim Biophys Acta Biomembr 307:74–82. https://doi.org/10.1016/0005-2736(73)90026-6
Cooper AL, Carrillo CD, Deschênes M, Blais B (2020) Quaternary ammonium compound resistance as a persistence marker for L. monocytogenes. J Food Prot. https://doi.org/10.4315/JFP-20-328
Cupkova V, Vinter V, Devinsky F, Lacko I, Mlynarcik D (1988) Inhibitory effect of 1-methyldodecyldimethylamine oxide and N,N-bis(dodecyldimethyl)-1,2-ethanediammonium dibromide on the spores of Bacillus cereus. Folia Microbiol (Praha) 33:433–439. https://doi.org/10.1007/BF02925767
Damen M, Groenen A, van Dongen S, Nolte R, Scholte B, Feiters M (2018) Transfection by cationic gemini lipids and surfactants. Med Chem Commun 9:1404–1425. https://doi.org/10.1039/c8md00249e
Debbasch C, de Saint JM, Pisella P, Rat P, Warnet J, Baudouin C (1999) Quaternary ammonium cytotoxicity in a human conjunctival cell line. J Fr Ophtalmol 22:950–958
Denyer S (1995) Mechanisms of action of antibacterial biocides. Int Biodeterior Biodegradation 36:227–245. https://doi.org/10.1016/0964-8305(96)00015-7
Dhawan V, Nagarsenker M (2017) Catanionic systems in nanotherapeutics—Biophysical aspects and novel trends in drug delivery applications. J Control Release 266:331–345. https://doi.org/10.1016/j.jconrel.2017.09.040
Dubnicková M, Rezanka T, Koscová H (2006) Adaptive changes in fatty acids of E. coli strains exposed to a quaternary ammonium salt and an amine oxide. Folia Microbiol (Praha) 51:371–374. https://doi.org/10.1007/bf02931578
Dubois-Brissonnet F, Malgrange C, Guerin-Mechin L, Heyd B, Leveau J (2001) Changes in fatty acid composition of Pseudomonas aeruginosa ATCC 15442 induced by growth conditions: consequences of resistance to quaternary ammonium compounds. Microbios 106:97–110
Elhanafi D, Dutta V, Kathariou S (2010) Genetic characterization of plasmid-associated benzalkonium chloride resistance determinants in a Listeria monocytogenes strain from the 1998–1999 outbreak. Appl Environ Microbiol 76:8231–8238. https://doi.org/10.1128/AEM.02056-10
Fait M, Bakas L, Garrote G, Morcelle S, Saparrat M (2019) Cationic surfactants as antifungal agents. Appl Microbiol Biotechnol 103:97–112. https://doi.org/10.1007/s00253-018-9467-6
Ferreira V, Wiedmann M, Teixeira P, Stasiewicz M (2014) Listeria monocytogenes persistence in food-associated environments: epidemiology, strain characteristics, and implications for public health. J Food Prot 77:150–170. https://doi.org/10.4315/0362-028X.JFP-13-150
Forbes S, Morgan N, Humphreys G, Amezquita A, Mistry H, McBain A (2019) Loss of Function in Escherichia coli exposed to environmentally relevant concentrations of benzalkonium chloride. Appl Environ Microbiol 85:e02417. https://doi.org/10.1128/AEM.02417-18
Fox E, Leonard N, Jordan K (2011) Physiological and transcriptional characterization of persistent and nonpersistent Listeria monocytogenes isolates. Appl Environ Microbiol 77:6559–6569. https://doi.org/10.1128/AEM.05529-11
Fraser H (1956) Oxyphenonium bromide as a mydriatic. Br J Ophthalmol 40:751–753. https://doi.org/10.1136/bjo.40.12.751
Furi L et al (2013) Evaluation of reduced susceptibility to quaternary ammonium compounds and bisbiguanides in clinical isolates and laboratory-generated mutants of Staphylococcus aureus. Antimicrob Agents Chemother 57:3488–3497. https://doi.org/10.1128/AAC.00498-13
Futoma-Kołoch B et al (2015) Selection and electrophoretic characterization of Salmonella enterica subsp. enterica biocide variants resistant to antibiotics. Pol J Vet Sci 18:725–732. https://doi.org/10.1515/pjvs-2015-0094
Futoma-Kołoch B et al (2017) Relationship of triamine-biocide tolerance of Salmonella enterica serovar senftenberg to antimicrobial susceptibility, serum resistance and outer membrane proteins. Int J Mol Sci 18:1459. https://doi.org/10.3390/ijms18071459
Futoma-Kołoch B, Bugla-Płoskońska G, Dudek B, Dorotkiewicz-Jach A, Drulis-Kawa Z, Gamian A (2019) Outer membrane proteins of Salmonella as potential markers of resistance to serum, antibiotics and biocides. Curr Med Chem 26:1960–1978. https://doi.org/10.2174/0929867325666181031130851
Garcia M, Ribosa I, Kowalczyk I, Pakiet M, Brycki B (2019) Biodegradability and aquatic toxicity of new cleavable betainate cationic oligomeric surfactants. J Hazard Mater 371:108–114. https://doi.org/10.1016/j.jhazmat.2019.03.005
Gerba C (2015) Quaternary ammonium biocides: efficacy in application. Appl Environ Microbiol 81:464–469. https://doi.org/10.1128/AEM.02633-14
Giraud I, Rapp M, Maurizis J, Madelmont J (2002) Synthesis and in vitro evaluation of quaternary ammonium derivatives of chlorambucil and melphalan, anticancer drugs designed for the chemotherapy of chondrosarcoma. J Med Chem 45:2116–2119. https://doi.org/10.1021/jm010926x
Grant W (1956) A study of the actions of nonaromatic quaternary ammonium compounds on the eye. Trans Am Ophthalmol Soc 54:417–451
Grijalvo S et al (2019) Cationic niosomes as non-viral vehicles for nucleic acids: challenges and opportunities in gene delivery. Pharmaceutics 11:50. https://doi.org/10.3390/pharmaceutics11020050
Guérin-Méchin L, Dubois-Brissonnet F, Heyd B, Leveau JY (1999) Specific variations of fatty acid composition of Pseudomonas aeruginosa ATCC 15442 induced by quaternary ammonium compounds and relation with resistance to bactericidal activity. J Appl Microbiol 87:735–742. https://doi.org/10.1046/j.1365-2672.1999.00919.x
Guo Q et al (2017) Investigation on interaction of DNA and several cationic surfactants with different head groups by spectroscopy, gel electrophoresis and viscosity technologies. Chemosphere 168:599–605. https://doi.org/10.1016/j.chemosphere.2016.11.019
Guo Y, Duan M, Wang X, Gao J, Guan Z, Zhang M (2019) Early events in rabies virus infection-attachment, entry, and intracellular trafficking. Virus Res 263:217–225. https://doi.org/10.1016/j.virusres.2019.02.006
Hussain M, Wilkinson A, Bramwell V, Christensen D, Perrie Y (2014) Th1 immune responses can be modulated by varying dimethyldioctadecylammonium and distearoyl-sn-glycero-3-phosphocholine content in liposomal adjuvants. J Pharm Pharmacol 66:358–366. https://doi.org/10.1111/jphp.12173
Inácio  et al (2016) Quaternary ammonium surfactant structure determines selective toxicity towards bacteria: mechanisms of action and clinical implications in antibacterial prophylaxis. J Antimicrob Chemother 71:641–654. https://doi.org/10.1093/jac/dkv405
Ito E et al (2009) Potential use of cetrimonium bromide as an apoptosis-promoting anticancer agent for head and neck cancer. Mol Pharmacol 76:969–983. https://doi.org/10.1124/mol.109.055277
Jacobs W, Heidelberger M, Bull C (1916) The bactericidal properties of the quaternary salts of hexamethylenetetramine: III. The relation between constitution and bactericidal action in the quaternary salts obtained from halogenacetyl compounds. J Exp Med 23:577–599. https://doi.org/10.1084/jem.23.5.577
Jennings M, Forman M, Duggan S, Minbiole K, Wuest W (2017) Efflux pumps might not be the major drivers of QAC resistance in methicillin-resistant Staphylococcus aureus. Chem Biochem 18:1573–1577. https://doi.org/10.1002/cbic.201700233
Karayiannis P (2017) Hepatitis B virus: virology, molecular biology, life cycle and intrahepatic spread. Hepatol Int 11:500–508. https://doi.org/10.1007/s12072-017-9829-7
Katharios-Lanwermeyer S, Rakic-Martinez M, Elhanafi D, Ratani S, Tiedje J, Kathariou S (2012) Coselection of cadmium and benzalkonium chloride resistance in conjugative transfers from nonpathogenic Listeria spp. to other Listeriae. Appl Environ Microbiol 78:7549–7556. https://doi.org/10.1128/aem.02245-12
Kirby A et al (2003) Gemini surfactants: new synthetic vectors for gene transfection. Angew Chem Int Ed Eng 42:1448–1457. https://doi.org/10.1002/anie.200201597
Klein S (2009) Polysaccharides in oral drug delivery—recent applications and future perspectives. In: Polysaccharide materials: performance by design, ACS Symposium Series, vol 1017. American Chemical Society, pp 13–30. https://doi.org/10.1021/bk-2009-1017.ch001
Knežević M et al (2020) Increased sensitivity of amoeba-grown Francisella species to disinfectants. Microorganisms 8:1260. https://doi.org/10.3390/microorganisms8091260
Koyama K, Shimazu Y (2005) Benzalkonium chloride. In: Suzuki O, Watanabe K (eds) Drugs and poisons in humans, vol 23. Springer, New York, pp 407–413
Kücken D, Feucht H, Kaulfers P (2000) Association of qacE and qacEDelta1 with multiple resistance to antibiotics and antiseptics in clinical isolates of gram-negative bacteria. FEMS Microbiol Lett 183:95–98. https://doi.org/10.1111/j.1574-6968.2000.tb08939.x
Kwaśniewska D, Chen Y, Wieczorek D (2020) Biological activity of quaternary ammonium salts and their derivatives. Pathogens (Basel, Switz) 9:459. https://doi.org/10.3390/pathogens9060459
Labena A, Hegazy M, Sami R, Hozzein W (2020) Multiple applications of a novel cationic gemini surfactant: anti-microbial, anti-biofilm, biocide, salinity corrosion inhibitor, and biofilm dispersion (Part II). Molecules (Basel, Switz) 25:1348. https://doi.org/10.3390/molecules25061348
Lachowicz T, Witkowska R, Obłąk E (1990) Amino acid auxotrophy increases sensitivity of Saccharomyces cerevisiae to a quaternary ammonium salt IM. Acta Microbiol Pol 39:157–162
Lee C (2001) Structure, conformation, and action of neuromuscular blocking drugs. Br J Anaesth 87:755–769. https://doi.org/10.1093/bja/87.5.755
Lever M, Slow S (2010) The clinical significance of betaine, an osmolyte with a key role in methyl group metabolism. Clin Biochem 43:732–744. https://doi.org/10.1016/j.clinbiochem.2010.03.009
Li R, Wang Z, Xu Q, Yao S, Li Z, Song H (2020) Synthesis, characterization and physicochemical properties of new chiral quinuclidinol quaternary ammonium salts. J Mol Struct 1209:127918. https://doi.org/10.1016/j.molstruc.2020.127918
Ligowska-Marzęta M, Hancock V, Ingmer H, Aarestrup FM (2019) Comparison of gene expression profiles of uropathogenic Escherichia Coli CFT073 after prolonged exposure to subinhibitory concentrations of different biocides. Antibiotics (Basel, Switz) 8:167. https://doi.org/10.3390/antibiotics8040167
Longo N, Frigeni M, Pasquali M (2016) Carnitine transport and fatty acid oxidation. Biochim Biophys Acta 1863:2422–2435. https://doi.org/10.1016/j.bbamcr.2016.01.023
Lu N et al (2018) Bifunctional thiourea–ammonium salt catalysts derived from cinchona alkaloids: cooperative phase-transfer catalysts in the enantioselective aza-henry reaction of ketimines. J Organomet Chem 83:1486–1492. https://doi.org/10.1021/acs.joc.7b03078
Maertens H, De Reu K, Meyer E, Van Weyenberg S, Dewulf J, Van Coillie E (2019) Exposure of ciprofloxacin-resistant Escherichia coli broiler isolates to subinhibitory concentrations of a quaternary ammonium compound does not increase antibiotic resistance gene transfer. Poult Sci 98:2972–2976. https://doi.org/10.3382/ps/pez185
Makvandi P, Jamaledin R, Jabbari M, Nikfarjam N, Borzacchiello A (2018) Antibacterial quaternary ammonium compounds in dental materials: a systematic review. Dent Mater 34:851–867. https://doi.org/10.1016/j.dental.2018.03.014
Martínez-Suárez J, Ortiz S, López-Alonso V (2016) Potential impact of the resistance to quaternary ammonium disinfectants on the persistence of Listeria monocytogenes in food processing environments. Front Microbiol 7:638. https://doi.org/10.3389/fmicb.2016.00638
Mbithi J, Springthorpe V, Sattar S (1990) Chemical disinfection of hepatitis A virus on environmental surfaces. Appl Environ Microbiol 56:3601–3604
McDonnell G, Russell A (1999) Antiseptics and disinfectants: activity, action, and resistance. Clin Microbiol Rev 12:147–179
McGregor D, Dellow W, Robson R, Lever M, George P, Chambers S (2002) Betaine supplementation decreases post-methionine hyperhomocysteinemia in chronic renal failure. Kidney Int 61:1040–1046. https://doi.org/10.1046/j.1523-1755.2002.00199.x
Morrison K, Allen R, Minbiole K, Wuest W (2019) More QACs, more questions: recent advances in structure activity relationships and hurdles in understanding resistance mechanisms. Tetrahedron Lett 60:150935. https://doi.org/10.1016/j.tetlet.2019.07.026
Myers D (2005) Surfactant science and technology. John Wiley & Sons. https://doi.org/10.1002/047174607x
Nikunj D, Tejas J (2017) A concise review on surfactants and tts significance. IJAC 13:663–672
Nor A’shimi M et al (2019) Biocide susceptibilities and biofilm-forming capacities of Acinetobacter baumannii clinical isolates from Malaysia. J Infect Dev Countries 13:626–633. https://doi.org/10.3855/jidc.11455
Obłąk E, Krasowska A (2010) The influence of organic nitrogen compounds on melanoma, bacterial, and fungal cells. Adv Clin Exp Med 19:65–75
Obłąk E, Ułaszewski S, Morawiecki A, Witek S, Witkowska R, Majcher K, Lachowicz TM (1989) Quaternary ammonium salt resistant mutants in yeast Saccharomyces cerevisiae. Yeast 5:273–278
Obłąk E, Bacal J, Lachowicz T (2000) A quaternary ammonium salt as an inhibitor of plasma membrane H+-ATPase in yeast Saccharomyces cerevisiae. Cell Mol Biol Lett 5:315–324
Obłąk E, Lachowicz T, Łuczyński J, Witek S (2002) Lysosomotropic N,N-dimethyl alpha-aminoacid N-alkyl esters and their quaternary ammonium salts as plasma membrane and mitochondrial ATPases inhibitors. Cell Mol Biol Lett 7:1121–1129
Obłąk E, Adamski R, Lachowicz T (2003) pH-dependent influence of a quaternary ammonium salt and an aminoester on the yeast Saccharomyces cerevisiae ultrastructure. Cell Mol Biol Lett 8:105–110
Obłąk E, Gamian A, Adamski R, Ułaszewski S (2010) The physiological and morphological phenotype of a yeast mutant resistant to the quaternary ammonium salt N-(dodecyloxycarboxymethyl)-N,N,N-trimethyl ammonium chloride. Cell Mol Biol Lett 15:215–233. https://doi.org/10.2478/s11658-010-0002-8
Obłąk E, Piecuch A, Krasowska A, Łuczyński J (2013) Antifungal activity of gemini quaternary ammonium salts. Microbiol Res 168:630–638. https://doi.org/10.1016/j.micres.2013.06.001
Obłąk E, Piecuch A, Guz-Regner K, Dworniczek E (2014) Antibacterial activity of gemini quaternary ammonium salts. FEMS Microbiol Lett 350:190–198. https://doi.org/10.1111/1574-6968.12331
Obłąk E, Piecuch A, Dworniczek E, Olejniczak T (2015) The influence of biodegradable gemini surfactants, N,N′-bis(1-decyloxy-1-oxopronan-2-yl)-N,N,N′,N′ tetramethylpropane-1,3-diammonium dibromide and N,N′-bis(1-dodecyloxy-1-oxopronan-2-yl) N,N,N′,N′-tetramethylethane-1,2-diammonium dibromide, on fungal biofilm and adhesion. J Oleo Sci 64:527–537. https://doi.org/10.5650/jos.ess14195
Obłąk E, Piecuch A, Maciaszczyk-Dziubińska E, Wawrzycka D (2016) Quaternary ammonium salt N-(dodecyloxycarboxymethyl)-N,N,N-trimethyl ammonium chloride induced alterations in Saccharomyces cerevisiae physiology. J Biosci 41:601–614. https://doi.org/10.1007/s12038-016-9644-7
Obłąk E, Piecuch A, Rewak-Soroczyńska J, Paluch E (2019) Activity of gemini quaternary ammonium salts against microorganisms. Appl Microbiol Biotechnol 103:625–632. https://doi.org/10.1007/s00253-018-9523-2
Ohkawa K, Kim H, Lee K (2004) Biodegradation of electrospun poly(e-caprolactone) non-woven fabrics by pure-cultured soil filamentous fungi. J Polym Environ 12:211–219. https://doi.org/10.1007/s10924-004-8148-y
Orelle C, Mathieu K, Jault JM (2019) Multidrug ABC transporters in bacteria. Res Microbiol 170:381–391. https://doi.org/10.1016/j.resmic.2019.06.001
Paluch E, Piecuch A, Obłąk E, Lamch Ł, Wilk K (2018) Antifungal activity of newly synthesized chemodegradable dicephalic-type cationic surfactants. Colloids Surf B: Biointerfaces 164:34–41. https://doi.org/10.1016/j.colsurfb.2018.01.020
Picó Y, Font G, Moltó J, Mañes J (2000) Solid-phase extraction of quaternary ammonium herbicides. J Chromatogr A 885:251–271. https://doi.org/10.1016/s0021-9673(99)01145-0
Piecuch A, Lamch Ł, Paluch E, Obłąk E, Wilk K (2016a) Biofilm prevention by dicephalic cationic surfactants and their interactions with DNA. J Appl Microbiol 121:682–692. https://doi.org/10.1111/jam.13204
Piecuch A, Obłąk E, Guz-Regner K (2016b) Antibacterial activity of alanine-derived gemini quaternary ammonium compounds. J Surfactant Deterg 19:275–282. https://doi.org/10.1007/s11743-015-1778-3
Prado H, Matulewicz M (2014) Cationization of polysaccharides: a path to greener derivatives with many industrial applications. Eur Polym J 52:53–75. https://doi.org/10.1016/j.eurpolymj.2013.12.011
Rasmussen C, Kaufman P, Kiland J (2014) Benzalkonium chloride and glaucoma. J Ocul Pharmacol Ther 30:163–169. https://doi.org/10.1089/jop.2013.0174
Regis R, Zanini A, Della Vecchia M, Silva-Lovato C, Oliveira Paranhos H, de Souza R (2011) Physical properties of an acrylic resin after incorporation of an antimicrobial monomer. J Prosthodont 20:372–379. https://doi.org/10.1111/j.1532-849X.2011.00719.x
Rewak-Soroczyńska J, Paluch E, Siebert A, Szałkiewicz K, Obłąk E (2019) Biological activity of glycine and alanine derivatives of quaternary ammonium salts (QASs) against micro-organisms. Lett Appl Microbiol 69:212–220. https://doi.org/10.1111/lam.13195
Rietschel R (1995) Fisher’s contact dermatitis. Williams & Wilkins, Baltimore
Rinaudo M (2008) Main properties and current applications of some polysaccharides as biomaterials. Polym Int 57:397–430. https://doi.org/10.1002/pi.2378
Rogers B, Decottignies A, Kolaczkowski M, Carvajal E, Balzi E, Goffeau A (2001) The pleitropic drug ABC transporters from Saccharomyces cerevisiae. J Mol Microbiol Biotechnol 3:207–214
Russell A (2001) Mechanisms of bacterial insusceptibility to biocides. Am J Infect Control 29:259–261. https://doi.org/10.1067/mic.2001.115671
Russell A, Furr J (1986) Susceptibility of porin- and lipopolysaccharide-deficient strains of Escherichia coli to some antiseptics and disinfectants. J Hosp Infect 8:47–56. https://doi.org/10.1016/0195-6701(86)90104-0
Schrank C, Minbiole K, Wuest W (2020) Are quaternary ammonium compounds, the workhorse disinfectants, effective against severe acute respiratory syndrome-Coronavirus-2? ACS Infect Dis 6:1553–1557. https://doi.org/10.1021/acsinfecdis.0c00265
Schwartz L, Wooda A, Gibson D (2014) Trigonelline accumulation in leaves of Panicum virgatum seedlings. Nat Prod Commun 9:1163–1166. https://doi.org/10.1177/1934578X1400900826
Shaban S, Fouda A, Elmorsi M, Fayed T, Azazy O (2016) Adsorption and micellization behavior of synthesized amidoamine cationic surfactants and their biological activity. J Mol Liq 216:284–292. https://doi.org/10.1016/j.molliq.2015.12.111
Shao H, Jiang L, Meng W, Qing F (2003) Synthesis and antimicrobial activity of a perfluoroalkyl-containing quaternary ammonium salt. J Fluor Chem 124:89–91. https://doi.org/10.1016/S0022-1139(03)00193-3
Shao J, Zhang F, Bai Z, Wang C, Yuan Y, Wang W (2012) Synthesis and antitumor activity of emodin quaternary ammonium salt derivatives. Eur J Med Chem 56:308–319. https://doi.org/10.1016/j.ejmech.2012.07.047
Siatra-Papastaikoudi T, Papadaki-Valiraki A, Tsantili-Kakoulidou A, Tzouvelekis L, Mentis A (1994) Synthesis, lipophilicity studies and antibacterial properties of some novel quaternary ammonium salts. Chem Pharm Bull (Tokyo) 42:392–394. https://doi.org/10.1248/cpb.42.392
Teixeira R et al (2015) Novel serine-based gemini surfactants as chemical permeation enhancers of local anesthetics: a comprehensive study on structure-activity relationships, molecular dynamics and dermal delivery. Eur J Pharm Biopharm 93:205–213. https://doi.org/10.1016/j.ejpb.2015.02.024
To M, Favrin S, Romanova N, Griffiths M (2002) Postadaptational resistance to benzalkonium chloride and subsequent physicochemical modifications of Listeria monocytogenes. Appl Environ Microbiol 68:5258–5264. https://doi.org/10.1128/aem.68.11.5258-5264.2002
Trinchant J, Boscari A, Spennato G, Van de Sype G, Le Rudulier D (2004) Proline betaine accumulation and metabolism in alfalfa plants under sodium chloride stress. Exploring its compartmentalization in nodules. Plant Physiol 135:1583–1594. https://doi.org/10.1104/pp.103.037556
Vekariya R (2017) A review of ionic liquids: applications towards catalytic organic transformations. J Mol Liq 227:44–60. https://doi.org/10.1016/j.molliq.2016.11.123
Vijayakumar R et al (2018) Distribution of biocide resistant genes and biocides susceptibility in multidrug-resistant Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter baumannii—a first report from the Kingdom of Saudi Arabia. J Infect Public Health 11:812–816. https://doi.org/10.1016/j.jiph.2018.05.011
Wang W, Bai Z, Zhang F, Wang C, Yuan Y, Shao J (2012) Synthesis and biological activity evaluation of emodin quaternary ammonium salt derivatives as potential anticancer agents. Eur J Med Chem 56:320–331. https://doi.org/10.1016/j.ejmech.2012.07.051
Wassenaar T, Ussery D, Nielsen L, Ingmer H (2015) Review and phylogenetic analysis of qac genes that reduce susceptibility to quaternary ammonium compounds in Staphylococcus species. Eur J Microbiol Immunol (Bp) 5:44–61. https://doi.org/10.1556/eujmi-d-14-00038
**ao Y, Chen J, Fang M, **ng X, Wang H, Wang Y, Li F (2008) Antibacterial effects of three experimental quaternary ammonium salt (QAS) monomers on bacteria associated with oral infections. J Oral Sci 50:323–327. https://doi.org/10.2334/josnusd.50.323
Xu D, Li Y, Zahid M, Yamasaki S, Shi L, Li J, Yan H (2014) Benzalkonium chloride and heavy-metal tolerance in Listeria monocytogenes from retail foods. Int J Food Microbiol 190:24–30. https://doi.org/10.1016/j.ijfoodmicro.2014.08.017
Yan N (2015) Structural biology of the major facilitator superfamily transporters. Annu Rev Biophys 44:257–283. https://doi.org/10.1146/annurev-biophys-060414-033901
Zakharova L, Pashirova T, Fernandes A, Doktorovova S, Martins-Gomes C, Silva A, Souto E (2018) Self-assembled quaternary ammonium surfactants for pharmaceuticals and biotechnology. In: Grumezescu AM (ed) Organic materials as smart nanocarriers for drug delivery. William Andrew Publishing, pp 601–618. https://doi.org/10.1016/B978-0-12-813663-8.00014-2
Zakharova L et al (2019) Cationic surfactants: self-assembly, structure-activity correlation and their biological applications. Int J Mol Sci 20:5534. https://doi.org/10.3390/ijms20225534
Zayed S (2011) Flow injection potentiometric determination of pancuronium bromide in fharmaceutical preparation and urine samples using modified carbon paste electrodes. Chem Pharm Bull (Tokyo) 59:254–259. https://doi.org/10.1248/cpb.59.254
Zhang Y et al (2015) Synthesis and antibacterial characterization of waterborne polyurethanes with gemini quaternary ammonium salt. Sci Bull 60:1114–1121. https://doi.org/10.1007/s11434-015-0811-2
Zhi D, Bai Y, Yang J, Cui S, Zhao Y, Chen H, Zhang S (2018) A review on cationic lipids with different linkers for gene delivery. Adv Colloid Interf Sci 253:117–140. https://doi.org/10.1016/j.cis.2017.12.006
Zubris D, Minbiole K, Wuest W (2017) Polymeric quaternary ammonium compounds: versatile antimicrobial materials. Curr Top Med Chem 17:305–318. https://doi.org/10.2174/1568026616666160829155805
Acknowledgements
We are sincerely grateful Alicja Seniuk for kind help with the fluorescence study.
Funding
The work is financed by the National Science Center from the Opus 16 research Project No. 2018/31/B/NZ9/03878.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Obłąk, E., Futoma-Kołoch, B. & Wieczyńska, A. Biological activity of quaternary ammonium salts and resistance of microorganisms to these compounds. World J Microbiol Biotechnol 37, 22 (2021). https://doi.org/10.1007/s11274-020-02978-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-020-02978-0