Abstract
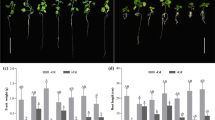
Solanum nigrum (S. nigrum), a newly discovered Cd-hyperaccumulator, has attracted the attention of many scholars. The present experiment was aimed to explore the difference of Cd accumulation ability among different S. nigrum genotypes and reveal the underlying mechanism. Three different genotypes of S. nigrum were grown in a hydroponic system with different Cd levels (0, 10,25, 50, 100 mg L−1, respectively); the Cd content, subcellular distribution of Cd, net Cd fluxes of in S. nigrum roots, and organic acid content in S. nigrum leaves were investigated. The results showed that Cd was more abundant in cell sap and cell wall fraction than that in organelle fraction. The content of organic acids in three species of S. nigrum was citric acid > tartaric acid > acetic acid > malic acid > oxalic acid. Non-destructive micrometry (NMT) revealed that the net Cd fluxes of the same species of S. nigrum first increased and then decreased with increasing of Cd concentration. And among all tested genotypes, the net Cd fluxes were stronger in the genotype of S. nigrum (HZ).







Similar content being viewed by others

Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Dai, F. W., Luo, G. Q., Li, Z. Y., Wei, X., & Wang, Z. J. (2020). Physiological and transcriptomic analyses of mulberry (Morus atropurpurea) response to cadmium stress. Ecotoxicology and Environmental and Safety, 205, 111–298. https://doi.org/10.1016/j.ecoenv.2020.111298
Dong, J., Mao, W. H., Zhang, G. P., Wu, F. B., & Cai, Y. (2007). Root excretion and plant tolerance to cadmium toxicityea review. Plant, Soil and Environment, 53(5), 193–200. https://doi.org/10.1007/s11104-007-9257-x
Elizabeth, P. S. (2005). Phytoremediation. Annual Review of Plant Biology, 56, 15–39. https://doi.org/10.1146/annurev.arplant.56.032604.144214
Elobeid, M., Göbel, C., Feussner, I., & Polle, A. (2012). Cadmium interferes with auxin physiology and lignification in poplar. Journal of Experimental Botany, 63, 1413–1421. https://doi.org/10.1093/jxb/err384
Fidalgo, F., Freitas, R., Ferreira, R., Pessoa, A. M., & Teixeira, J. (2011). Solanum nigrum L. antioxidant defense system isozymes are regulated transcriptionally and posttranslationally in Cd-induced stress. Environmental and Experimental Botany., 72, 312–319. https://doi.org/10.1016/j.envexpbot.2011.04.007
Fu, X., Dou, C., Chen, Y., Chen, X., Shi, J., Yu, M., & Xu, J. (2011). Subcellular distribution and chemical forms of cadmium in Phytolaccaamericana L. Journal of Hazardos Materials, 186, 103–107. https://doi.org/10.1016/j.jhazmat.2010.10.122
Gao, Y., Zhou, P., & Mao, L. (2010). Improvement of phytoextraction and antioxidative defense of Solanum nigrum L. under cadmium stress by application of cadmium-resistant strain and citric acid synergy. Journal of Hazardos Materials, 181, 771–777. https://doi.org/10.1016/j.jhazmat.2010.05.080
Hernández, L. E., Sobrino-Plata, J., Montero-Palmero, M. B., Carrasco-Gil, S., Flores-Cáceres, M. L., Ortega-Villasante, C., & Escobar, C. (2015). Contribution of glutathione to the control of cellular redox homeostasis under toxic metal and metalloid stress. Journal of Experimental Botany, 66(10), 2901–2911. https://doi.org/10.1093/jxb/erv063
Jia, D. D., Liu, A. Q., Li, H. T., Yu, Y. Y., Wei, Z. C., Wang, J. N., & Zhou, L. L. (2017). Applications and advances of non-invasive micro-test technology in plant physiologicalecology research. Chin Journal of Applied Environmental and Biology, 1, 177–184. https://doi.org/10.3724/SP.J.1145.2016.01031
Krzesłowska, M. (2010). The cell wall in plant cell response to trace metals: Polysaccharide remodeling and its role in defense strategy. Acta Physiologiae Plantarum, 33(1), 35–51. https://doi.org/10.1007/s11738-010-0581-ze
Larsen, P. B., Degenhardt, J., Tai, C. Y., Stenzler, L. M., Howell, S. H., & Kochian, L. V. (1997). Aluminum-resistant Arabidopsis mutants that exhibit altered patterns of aluminum accumulation and organic acid release from rots. Plantarum Physiology, 117(1), 19–27. https://doi.org/10.2307/4278249
Li, W., Xu, B., Song, Q., Liu, X., Xu, J., & Brookes, P. C. (2014). The identification of ‘hotspots’ of heavy metal pollution in soil-rice systems at a regional scale in eastern China. Science of the Total Environment, 472, 407–420. https://doi.org/10.1016/j.scitotenv.2020.125834
Li, W. C., Deng, H., & Wong, M. H. (2017). Effects of Fe plaque and organic acids on metal uptake by wetland plants under drained and waterlogged conditions. Environmental Pollution, 231, 732–741. https://doi.org/10.1016/j.envpol.2017.08.012
Li, X., Ma, H., Li, L. L., Gao, Y., Li, Y. Z., & Xu, H. (2019). Subcellular distribution, chemical forms and physiological responses involved in cadmium tolerance and detoxification in Agrocybe Aegerita. Ecotoxicology and Environmental Safety, 171, 66–74. https://doi.org/10.1016/j.ecoenv.2018.12.063
López-Bucio, J., Nieto-Jacobo, M. F., Ramı́rez-Rodrı́guez, V., Herrera-Estrella, L., (2000) Organic acid metabolism in plants: From adaptive physiology to transgenic varieties for cultivation in extreme soils. Plant Science 160(1): 0-13. https://doi.org/10.1016/s0168-9452(00)00347-2
Ma, Y. L., He, J. L., Ma, C. F., Luo, J., Li, H., Liu, T. X., Polle, A., Peng, C. H., & Luo, Z. B. (2014). Ectomycorrhizas with Paxillus involutus enhance cadmium uptake and tolerance in Populus × canescens. Plant Cell Environment, 37, 627–642. https://doi.org/10.1111/pce.12183
Marques, A. P. G. C., Oliveira, R. S., Samardjieva, K. A., Pissarra, J., Rangel, A. O. S. S., & Castro, P. M. L. (2007). Solanum nigrum grown in contaminated soil: Effect of arbuscular mycorrhizal fungi on zinc accumulation and histolocalisation. Environment Pollution., 145, 691–699. https://doi.org/10.1016/j.envpol.2006.06.029
Rizwan, M., Ali, S., Zia Ur Rehman, M., Rinklebe, J., Tsang, D. C. W., Bashir, A., Maqbool, A., Tack, F. M. G., & Ok, Y. S. (2018). Cadmium phytoremediation potential of Brassica crop species: A review. Science of the Total Environmental, 631e632, 1175e1191. https://doi.org/10.1016/j.scitotenv.2018.03.104
Sun, J., Wang, R., Liu, Z., Ding, Y., & Li, T. (2013). Non-invasive microelectrode cadmium flux measurements reveal the spatial characteristics and real-time kinetics of cadmium transport in hyperaccumulator and nonhyperaccumulator ecotypes of Sedum alfredii. Plant Physiology, 170, 355–359. https://doi.org/10.1016/j.jplph.2012.10.014
Wei, S. H., Li, Y. M., Zhou, Q. X., & Srivastava, M. (2010). Effect of fertilizer amendments on phytoremediation of Cd-contaminated soilby a newly discovered hyperaccumulator Solanum nigrum L. Journal of Hazardous Materials, 176, 269–273. https://doi.org/10.1016/j.jhazmat.2009.11.023
Wei, S. H., Zhou, Q. X., Wang, X., Zhang, K. S., Guo, G. L., & Ma, L. Q. Y. (2005). A newly discovered cadmium superaccumulation plant Solanum nigrum L. Chinese. Science Bulletin, 49(24), 2568–2573. https://doi.org/10.1360/982004-292
Wei, T., Liu, X., Dong, M. F., **n, Lv., Hua, L., & Jia, H. L. (2021). Rhizosphere iron and manganese-oxidizing bacteria stimulate root iron plaque formation and regulate Cd uptake of rice plants (Oryza sativa L). Journal of Environmental Management, 278, 111533. https://doi.org/10.1016/j.jenvman.2020.111533
Wei, T., Lv, X., Jia, H., Hua, L., Xu, H., Zhou, R., Zhao, J., Ren, X., & Guo, J. (2018). Effects of salicylic acid, Fe (II) and plant growth-promoting bacteria on Cd accumulation and toxicity alleviation of Cd tolerant and sensitive tomato genotypes. Journal of Environmental Management, 214, 164–171. https://doi.org/10.1016/j.jenvman.2018.02.100
Xu, J., Sun, J., Du, L., & Liu, X. (2012a). Comparative transcriptome analysis of cadmium responses in Solanum nigrum and Solanum torvum. New Phytologist, 196(1), 110–124. https://doi.org/10.1111/j.1469-8137.2012.04235.x
Xu, J., Yin, H. X., & Li, X. (2009). Protective effects of proline against cadmium toxicity in micropropagated hyperaccumulator, Solanum nigrum L. Plant Cell Reports Journal, 28(2), 325–333. https://doi.org/10.1007/s00299-008-0643-5
Xu, J., Zhu, Y., Ge, Q., Li, Y., Sun, J., Zhang, Y., & Liu, X. (2012b). Comparative physiological responses of Solanum nigrum and Solanum torvum to cadmium stress. New Phytologist, 196(1), 125–138. https://doi.org/10.1111/j.1469-8137.2012.04236.x
Xu, L., Dai, H. P., Skuza, L., & Wei, S. H. (2020). Optimal voltage and treatment time of electric field with assistant Solanum nigrum L cadmium hyperaccumulation in soil. Chemosphere, 253, 126575.
Yang, W. J., Wang, Q. Y., & Sun, J. Y. (2018). Alleviation mechanism of exogenous silicon in cabbages under cadmium stress. Acta Botanica Boreali-Occident Sinica, 38(6), 1088–1098. https://doi.org/10.7606/J.issn.1000-4025.2018.06.1088
Zhu, X. F., Wang, Z. W., Dong, F., Lei, G. J., Shi, Y. Z., Li, G. X., & Zheng, X. J. (2013). Exogenous auxin alleviates cadmium toxicity in Arabidopsis thaliana by stimulating synthesis of hemicellulose 1 and increasing the cadmium fixation capacity of root cell walls. Journal of Hazardous Materials, 263, 398–403. https://doi.org/10.1016/j.jhazmat.2013.09.018
Acknowledgements
Key Research & Development programs of Weiyang District, **an (202043), and Projects of **’an Science & Technology (GXYD1.9).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hua, L., Liang, Z. & Wei, T. Cadmium Tolerance Mechanism of Solanum nigrum Based on Subcellular Distribution and Organic Acid Content. Water Air Soil Pollut 233, 318 (2022). https://doi.org/10.1007/s11270-022-05803-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05803-6