Abstract
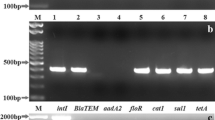
Salmonella species (spp) is the most important gastrointestinal pathogen present ubiquitously. Non typhoidal Salmonella (NTS) is commonly associated with gastroenteritis in humans. Layer birds once get infection with NTS, can become persistently infected with Salmonella Typhimurium and intermittently shed the bacteria. It results in a high risk of potential exposure of eggs to the bacteria. The current study was conducted to determine the serotype diversity, presence of virulence genes, antibiotic resistance pattern, and genes of NTS from poultry enteritis. Out of 151 intestinal swabs from poultry total 118 NTS were isolated, which were characterized serologically as S. Typhimurium (51 strains), S. Weltevreden (57 strains) and untypable (10 strains). Most effective antibiotics were amikacin, gentamycin and ceftriaxone (33.05%) followed by ampicillin, azithromycin and ciprofloxacin (16.69%), co-trimoxazole (13.55%), and tetracycline (6.78%). Multidrug resistance recorded in 17.70% (N = 21/118) strains. Antimicrobial-resistant genes i.e. blaTEM, blaSHV, blaCTX-M, tet(A), tet(B), tet(C), sul1, sul2, sul3. blaTEM and tet(A) were present in 95% (20/21). Eleven virulence genes i.e. invA, hilA, sivH, tolC, agfA, lpfA, spaN, pagC, spiA, iroN and fliC 2 were present in all the 30 isolates. While, sopE was present in only 2 isolates, NTS strains with characteristics of pathogenicity and multidrug resistance from poultry enteritis were detected. Multidrug resistance showed the necessity of prudent use of antibiotics in the poultry industry.



Similar content being viewed by others
References
Abdelfatah, E. N., Mohameda, A. S., & Abdellatif, S. S., 2018. Molecular differentiation and antimicrobial resistance patterns of Salmonella enteritidis and Salmonella typhimurium isolated from different food samples, Proceedings in 5th International Food Safety Conference Daman hour University,2018,165–183
Abdellah, C., Fouzia, R. F., Abdelkader, C., Rachida, S. B.& Mouloud, Z., 2009. Prevalence and anti-microbial susceptibility of Salmonella isolates from chicken carcasses and giblets in Mekns, Morocco, African Journal of Microbiology Research, 3(5), 215-219.
Ahmad, Y., 2020. Molecular screening of resistant and virulent genes in salmonella enteritidis and salmonella typhimurium from poultry in Khyber Pakhtunkhwa, Pakistan Veterinary Journal 40(3), 343–349. https://doi.org/10.29261/pakvetj/2020.044
Aldridge, P., Gnerer, J., Karlinsey, J. E., & Hughes, K. T. (2006). Transcriptional and translational control of the Salmonella fliC gene, Journal of Bacteriology, 188(12), 4487–4496. https://doi.org/10.1128/JB.00094-06
Arora, D., Kumar, S., Singh, D., **dal, N., & Mahajan, N. K. (2013). Isolation, characterization and antibiogram pattern of salmonella from poultry in parts of Haryana, India, Advances in Animal and Veterinary Sciences, 1(5), 161–163.
Awad, A., Gwida, M., Khalifa, E., & Sadat, A., 2020. Phenotypes, antibacterial-resistant profile, and virulence-associated genes of Salmonella serovars isolated from retail chicken meat in Egypt, Veterinary World, 13(3), 440–445. doi:https://doi.org/10.14202/vetworld.2020.440-445
Barua, H., Biswas, P. K., Olsen, K. E., Shil, S. K., & Christensen, J. P., 2013. Molecular characterization of motile serovars of Salmonella enterica from breeder and commercial broiler poultry farms in Bangladesh, PLOS ONE, 8(3), e57811. doi:https://doi.org/10.1371/journal.pone.0057811
Bauer, A. W., 1966. Antibiotic susceptibility testing by a standardized single disc method, American Journal of Clinical Pathology, 45, 149–158. https://doi.org/10.1093/ajcp/45.4_ts.493
Bertelloni, F., Tosi, G., Massi, P., Fiorentini, L., Parigi, M., Cerri, D., & Ebani, V. V., 2017. Some pathogenic characters of paratyphoid Salmonella enterica strains isolated from poultry, Asian Pacific Journal of Tropical Medicine, 10(12), 1161–1166. https://doi.org/10.1016/j.apjtm.2017.10.023
Borges, K. A., Furian, T. Q., Borsoi, A., Moraes, H. L. S., Salle, C. T. P., & Nascimento, V. P., 2013. Detection of virulence-associated genes in Salmonella enteritidis isolates from chicken in South of Brazil, Pesquisa Veterinaria Brasileira, 33(12), 1416–1422. https://doi.org/10.1590/S0100-736X2013001200004
Cantas, L., Shah, S. Q. A., Cavaco, L. M., Manaia, C. M., Walsh, F., Popowska, M., Garelick, H., Bürgmann, H., & Sørum, H., 2013. A brief multi-disciplinary review on antimicrobial resistance in medicine and its linkage to the global environmental microbiota, Frontiers in Microbiology, 14(4), 96. https://doi.org/10.3389/fmicb.2013.00096
Card, R., Vaughan, K., Bagnall, M., Spiropoulos, J., Cooley, W., Strickland, T., & Anjum, M. F., 2016. Virulence characterization of Salmonella enterica isolates of differing antimicrobial resistance recovered from UK livestock and imported meat samples, Frontiers in Microbiology, 7(640), 1–11. doi:https://doi.org/10.3389/fmicb.2016.00640
Castilla, K. S., Ferreira, C. S. A., Moreno, A. M., Nunes, I. A., & Ferreira, A. J. P., 2006. Distribution of virulence genes sefC, pefA and spvC in Salmonella enteritidis phage type 4 strains isolated in Brazil, Brazilian Journal of Microbiology, 37(2), 135–139. https://doi.org/10.1590/S1517-83822006000200007
Chatterjee, R. N. and Rajkumar, U., 2015. An overview of poultry production in India, Indian journal of animal health, 54(2): 89-108.
Chen, Z., Bai, J., Wang, S., Zhang, X., Zhan, Z., Shen, H., Zhang, H., Wen, J., Gao, Y., Liao, M., & Zhang, J., 2020. Prevalence, Antimicrobial Resistance, Virulence Genes and Genetic Diversity of Salmonella Isolated from Retail Duck Meat in Southern China, Microorganisms, 8(3), 444. https://doi.org/10.3390/microorganisms8030444
CLSI. Autoverification of Medical Laboratory Results for Specific Disciplines, 1st ed. CLSI guideline AUTO15, Clinical and Laboratory Standards Institute; 2019.
Collinson, S. K., Liu, S. L., Clouthier, S. C., Banser, P. A., Doran, J. L., Sanderson, K. E., & Kay, W. W., 1996. The location of four fimbrin-encoding genes, Agfa, fimA, sefA and sefD, on the Salmonella enteritidis and/or S. typhimurium XbaI-BlnI genomic restriction maps, Gene, 169(1), 75–80. https://doi.org/10.1016/0378-1119(95)00763-6
Ferrari, R. G., Rosario, D. K. A., Cunha-Neto, A., Mano, S. B., Figueiredo, E. E. S., & Conte-Junior, C. A. 2019. Worldwide epidemiology of Salmonella serovars in animal-based foods: A meta-analysis, Applied and Environmental Microbiology, 85(14),e00591-619. https://doi.org/10.1128/AEM.00591-19
Fluit, A. C., 2005. Towards more virulent and antibiotic-resistant Salmonella?, FEMS Immunology and Medical Microbiology, 43(1), 1–11. https://doi.org/10.1016/j.femsim.2004.10.007
Galán, J. E., Ginocchio, C., & Costeas, P., 1992. Molecular and functional characterization of the Salmonella invasion gene invA: Homology of InvA to members of a new protein family, Journal of Bacteriology, 174(13), 4338–4349. https://doi.org/10.1128/jb.174.13.4338-4349.1992
Galanis, E., Lo Fo Wong, D. M. A., Patrick, M. E., Binsztein, N., Cieslik, A., Chalermchikit, T., Aidara-Kane, A., Ellis, A., Angulo, F. J., Wegener, H. C., & World Health Organization Global Salm-Surv., 2006. Web-based surveillance and global Salmonella distribution, 2000–2002, Emerging Infectious Diseases, 12(3), 381–388https://doi.org/10.3201/eid1205.050854
Hai, D., Yin, X., Lu, Z., Lv, F., Zhao, H., & Bie, X. (2020). Occurrence, drug resistance, and virulence genes of Salmonella isolated from chicken and eggs, Food Control, 113, 107-109. https://doi.org/10.1016/j.foodcont.2020.107109
Humphrey, T., 2000. Public-health aspects of Salmonella infection, Salmonella in domestic animals ( 245–263), CABI Publishing. U.K. http://sherekashmir.informaticspublishing.com
Ikeda, J. S., Schmitt, C. K., Darnell, S. C., Watson, P. R., Bispham, J., Wallis, T. S., Weinstein, D. L., Metcalf, E. S., Adams, P., O’Connor, C. D., & O’Brien, A. D., 2001. Flagellar phase variation of Salmonella enterica serovar Typhimurium contributes to virulence in the murine typhoid infection model but does not influence Salmonella-induced enteropathogenesis, Infection and Immunity, 69(5), 3021–3030. doi:https://doi.org/10.1128/IAI.69.5.3021-3030.2001
Ilyas B, Tsai CN and Coombes B.K., 2017. Evolution of Salmonella-Host Cell Interactions through a Dynamic Bacterial Genome, Frontiers in Cellular and infection Microbiology, 7, 428. doi: https://doi.org/10.3389/fcimb.2017.00428
Jayaweera, T. S. P., Ruwandeepika, H. A. D., Deekshit, V. K., Vidanarachchi, J. K., Kodithuwakku, S. P., Karunasagar, I., & Cyril, H. W., 2020. Isolation and identification of Salmonella spp from broiler chicken meat in Sri Lanka and their antibiotic resistance, Journal of Agricultural Sciences – Sri Lanka, 15(3), 395. doi:https://doi.org/10.4038/jas.v15i3.9031
**dal, N., Patnayak, D. P., Ziegler, A. F., Lago, A., & Goyal, S. M., 2009. A retrospective study on poult enteritis syndrome in Minnesota, Avian Diseases, 53(2), 268–275.doi:https://doi.org/10.1637/8513-110308-Reg.1
Keelara, S., Scott, H. M., Morrow, W. M., Hartley, C. S., Griffin, D. L., Gebreyes, W. A., & Thakur, S., 2014. Comparative phenotypic and genotypic characterization of temporally related nontyphoidal Salmonella isolated from human clinical cases, pigs, and the environment in North Carolina, Foodborne Pathogens and Disease, 11(2), 156–164. doi:https://doi.org/10.1089/fpd.2013.1630
Kingsley, R. A., Humphries, A. D., Weening, E. H., De Zoete, M. R., Winter, S., Papaconstantinopoulou, A., Dougan, G., & Bäumler, A. J., 2003. Molecular and phenotypic analysis of the CS54 island of Salmonella enterica serotype typhimurium: Identification of intestinal colonization and persistence determinants, Infection and Immunity, 71(2), 629–640. doi:https://doi.org/10.1128/IAI.71.2.629-640.2003
Kauffman, F., 1972.Serological diagnosis of Salmonella species, (Munksgaard, Copenhagen).
Li, F., Li, F., Chen, B., Zhou, B., Yu, P., Yu, S., Lai, W., & Xu, H., 2017. Sextuplex PCR combined with immunomagnetic separation and PMA treatment for rapid detection and specific identification of viable Salmonella spp, Salmonella enterica serovars Paratyphi B, Salmonella Typhimurium, and Salmonella enteritidis in raw meat, Food Control, 73, 587–594. doi:2016.09.009
Livermore, D. M., Canton, R., Gniadkowski, M., Nordmann, P., Rossolini, G. M., Arlet, G., Ayala, J., Coque, T. M., Kern-Zdanowicz, I., Luzzaro, F., Poirel, L., & Woodford, N. (2007). CTX-M: Changing the face of ESBLs in Europe, Journal of Antimicrobial Chemotherapy, 59(2), 165–174. doi:https://doi.org/10.1093/jac/dkl483
Lobani, A.M., Gharaibeh, S.M. and Al-Majali, A.M., 2016. Relationship between different enteric viral infections and the occurrence of diarrhea in broiler flocks in Jordan, Poultry Science. 95(6), 1257-1261.
Markey, B., Leonard, F., Archambault, M. Cullinane, A. and Magnuire, D., 2013. Clinical Veterinary Microbiology, (Mosby-Wolfe).
Mir I. A., Kashyap S. K. and Maherchandani S., 2015. Isolation, serotype diversity and antibiogram of Salmonella enterica isolated from different species of poultry in India, Asian Pacific Journal of Tropical Biomedicine, 5(7), 561-567 doi: /https://doi.org/10.1016/j.apjtb.2015.03.010
Odoch, T., Sekse, C., L’Abee-Lund, T. M., Høgberg Hansen, H. C., Kankya, C., & Wasteson, Y., 2018. Diversity and antimicrobial resistance genotypes in non-typhoidal salmonella isolates from poultry farms in Uganda, International Journal of Environmental Research and Public Health, 15(2), 324. doi:https://doi.org/10.3390/ijerph15020324
Pandey, M., & Sharath Kumar Goud, E., 2021. Non-Typhoidal Salmonellosis: A Major Concern for Poultry Industry, ( IntechOpen). https://doi.org/10.5772/intechopen.96400
Randall, L. P., Cooles, S. W., Osborn, M. K., Piddock, L. J. V., & Woodward, M. J., 2004. Antibiotic resistance genes, integrons and multiple antibiotic resistance in thirty-five serotypes of Salmonella enterica isolated from humans and animals in the UK, Journal of Antimicrobial Chemotherapy, 53(2), 208–216. https://doi.org/10.1093/jac/dkh070
Saikia, L., Sharma, A., Nath, R., Choudhury, G., & Borah, A. K., 2015. Salmonella Weltevreden food poisoning in a tea garden of Assam: An outbreak investigation, Indian Journal of Medical Microbiology, 33(4), 503–506. doi:https://doi.org/10.4103/0255-0857.167347
Singh, N. P., Bhatt, N., Usman, S. M. and Chaudhary, P., 2020. A detailed review on backyard poultry production and management in India, Journal of Entomology and Zoology Studies, 8(2), 1411-1415.
Skyberg, J. A., Logue, C. M., & Nolan, L. K., 2006. Virulence genoty** of Salmonella spp with multiplex PCR, Avian Diseases, 50(1), 77–81.doi:https://doi.org/10.1637/7417.1
Suez, J., Porwollik, S., Dagan, A., Marzel, A., Schorr, Y. I., Desai, P. T., Agmon, V., McClelland, M., Rahav, G., & Gal-Mor, O., 2013. Virulence gene profiling and pathogenicity characterization of non-typhoidal Salmonella accounted for invasive disease in humans, PLOS ONE, 8(3), e58449. doi:https://doi.org/10.1371/journal.pone.0058449
Suresh, Y., Bindukiranmayi, Ch., Rao, T. S., Srivani, M., Sankar, A. U. S. N., 2019. Prevalence and virulence gene profiles of Salmonella serovars isolated from animal origin, The Pharma Innovation Journal, 8(12), 139–145.
Webber, B., Borges, K. A., Furian, T. Q., Rizzo, N. N., Tondo, E. C., Santos, L. R. D., Rodrigues, L. B., & Nascimento, V. P. D., 2019. Detection of virulence genes in Salmonella Heidelberg isolated from chicken carcasses, Revista do Instituto de Medicina Tropical de São Paulo, 61, e36. doi:https://doi.org/10.1590/S1678-9946201961036
White, D. G., Zhao, S., Sudler, R., Ayers, S., Friedman, S., Chen, S., McDermott, P. F., McDermott, S., Wagner, D. D., & Meng, J., 2001. The isolation of antibiotic-resistant salmonella from retail ground meats, New England Journal of Medicine, 345(16), 1147–1154. doi:https://doi.org/10.1056/NEJMoa010315
Yoo, A. Y., Yu, J. E., Yoo, H., Lee, T. H., Lee, W. H., Oh, J. I., & Kang, H. Y., 2013. Role of sigma factor E in regulation of Salmonella AGF expression, Biochemical and Biophysical Research Communications, 430(1), 131–136.doi:https://doi.org/10.1016/j.bbrc.2012.11.025
Zhu, Y., Lai, H., Zou, L., Yin, S., Wang, C., Han, X., **a, X., Hu, k., He, L., Zhou, K., Chen, S., Ao, X. and Liu, S., 2017. Antimicrobial resistance and resistance genes in Salmonella strains isolated from broiler chickens along the slaughtering process in China, International journal of food microbiology, 259, 43-51
Acknowledgements
Authors want to acknowledge Vice Chancellor RAJUVAS, Bikaner and Dean, CVAS, Navania for financial support for the study
Funding
The authors declare that no funds, grant or other support were received during preparation of this manuscript. The study was conducted with institutional support (Dean, CVAS, Navania Vallabhnagar Udaipur, RAJUVAS Bikaner Rajasthan, India).
Author information
Authors and Affiliations
Contributions
Arpita Sain and Deepak Kumar Sharma: Concept and design of study or acquisition of data or analysis and interpretation of data.
Arpita Sain, Chaman Patidar, PrabuRam Suthar and Karishma Rathore: Carried out lab work as per requirement of the manuscript.
Rajesh Singathia, Abhishek Gaurav and Rohit Juneja: Drafting the article or revising it critically for important intellectual content.
Deepak Kumar Sharma, Rajesh Singathia, Abhishek Gaurav and Rohit Juneja: Final approval of the version to be published.
Corresponding author
Ethics declarations
Data availability statement
All data generated or analysed during this study are included in this published article.
Conflict of interest
Authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Department of Veterinary Microbiology, CVAS Navania, Vallabhnagar, Udaipur. Rajasthan University of Veterinary and Animal Sciences (RAJUVAS), Bikaner.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sain, A., Sharma, D.K., Singathia, R. et al. Antibiotic resistance and virulence genes profile of Non typhodial Salmonella species isolated from poultry enteritis in India. Trop Anim Health Prod 56, 91 (2024). https://doi.org/10.1007/s11250-024-03932-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-024-03932-0