Abstract
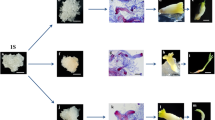
Somatic embryogenesis (SE) is a technique aimed at producing plant embryos in vitro and is considered a highly promising method for micropropagation. Here, we have established an efficient pathway for plant regeneration through somatic embryogenesis in S. chinensis. In the study, it was found that dark culture conditions significantly increased the rooting rate of plants through SE regeneration. Under dark conditions, radicle primordia were initiated during the globular embryo stage and developed from the heart-shaped to the torpedo-shaped embryo stages. The levels of IAA and ABA in somatic embryos subjected to the dark treatment were significantly lower (190.9 ng/g and 525.1 ng/g) from the globular to heart-shaped embryo stages compared to those in somatic embryos exposed to light (597.5 ng/g and 749.188 ng/g). Additionally, the concentrations of GA3 and ZR were lower at all stages under light treatment. Transcriptome sequencing and bioinformatics analysis revealed that the pathways and processes predominantly enriched in differentially expressed genes in somatic embryos under dark conditions were plant hormone signaling, circadian rhythm and phenylpropanoid biosynthesis. QRT-PCR was employed to validate the expression of genes related to plant hormone signaling transduction. The results were consistent with the transcriptome sequencing results. This work has laid the foundation for applied research and could prove useful in future programs aimed at improving reluctant woody plants. Furthermore, the findings can potentially be extended to other species as well.
Key Message
Dark culture significantly improved the radicle development of somatic embryos in S. chinensis and the differentially expressed genes were predominantly enriched in plant hormone signals, which corresponded to endogenous hormone levels.











Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this manuscript and are available from the corresponding author upon reasonable request.
References
Ai J, Wang ZX, Qin HY (2012) Research status and prospect of Schisandra chinensis in China. J Northeast Agri Univ 43(10):14–20. https://doi.org/10.3969/j.issn.1005-9369.2012.10.005
Ai J, Wang YP, Wang ZX, Sun D, Shi GL, Liu XY, Xu PL, Guo JH (2022) Chinese Schisandra germplasm resources. China Agricultural Publishing House, Bei**g
Arnold SV, Sabala I, Bozhkov P, Dyachok J, Filonova L (2002) Developmental pathways of somatic embryogenesis. Plant Cell Tiss Org 69(3):233–249. https://doi.org/10.1023/A:1015673200621
Bai R, Zhang XJ, Li YL, Liu JP, Zhang HB, **ao WL, Pu JX, Sun HD, Zheng YT, Liu LX (2015) SJP-L-5, a novel small-molecule compound, inhibits HIV-1 infection by blocking viral DNA nuclear entry. BMC Microbiol 15:274
Buchfink B, **e C, Huson DH (2015) Fast and sensitive protein alignment using diamond. Nat Methods 12(1):59. https://doi.org/10.1038/nmeth.3176
Chen AH, Yang JL, Niu YD, Yang CP, Liu GF, Yu CY, Li CH (2010) High-frequency somatic embryogenesis from germinated zygotic embryos of Schisandra chinensis and evaluation of the effects of medium strength, sucrose, GA3 and BA on somatic embryo development. Plant Cell Tiss Org 102(3):357–364. https://doi.org/10.1007/s11240-010-9740-6
Eisner N, Maymon T, Sanchez EC, Bar-Zvi D, Brodsky S, Finkelstein R, Bar-Zvi D (2021) Phosphorylation of Serine 114 of the transcription factor ABSCISIC ACID INSENSITIVE 4 is essential for activity. Plant Sci 3(05):1–10. https://doi.org/10.1016/j.plantsci.2021.110847
**g YJ, Lin RC (2017) Advances in plant light signal transduction in China. Chin Bull Botany 52(03):257–270. https://doi.org/10.11983/CBB16150
Kami C, Hersch M, Trevisan M, Genoud T, Hiltbrunner A, Bergmann S, Fankhauser C (2012) Nuclear phytochrome A signaling promotes phototropism in Arabidopsis. Plant Cell 24(02):566–576. https://doi.org/10.1105/tpc.111.095083
Kim TD, Anbazhagan VR, Park JI (2005) Somatic embryogenesis in Schisandra chinensis (Turcz) Baill. In Vitro Cell Dev Biol Plant 41:253–257. https://doi.org/10.1079/IVP2004630
Kim HJ, Ryu H, Hong SH, Woo HR, Lim PO, Lee IC, Sheen J, Nam HG, Hwang I (2006) Cytokinin-mediated control of leaf longevity by AHK3 through phosphorylation of ARR2 in Arabidopsis. PNAS 103(03):814–819. https://doi.org/10.1073/pnas.0505150103
Li J, Liu ZH, **ang FN (2015) Study on the role of cytokinin response regulators in mediating the growth and development of Arabidopsis thaliana. Life Sci 27(04):509–514. https://doi.org/10.13376/j.cbls/2015066
Li JT, Fan HY, Zhao QQ (2019) Regulation of abscisic acid on auxin synthesis and transport in rice roots. J **nyang Norm Univ (nat Sci Ed) 32(01):39–46. https://doi.org/10.3969/j.issn.1003-0972.2019.01.007
Liu XT (2020) Establishment of peony embryo regeneration system. Shenyang Agricultural University, Shenyang
Liu J, Mu X, Liang J, Zhang J, Qiang T, Li H, Li B, Liu H, Zhang B (2022) Metabolic profiling on the analysis of different parts of Schisandra chinensis based on UPLC-QTOF-MS with comparative bioactivity assays. Front Plant Sci 13:970535. https://doi.org/10.3389/fpls.2022.970535
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Map** and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5(7):621–628. https://doi.org/10.1038/nmeth.1226
Muller B, Sheen J (2007) Advances in cytokinin signaling. Science 318(5847):68–69. https://doi.org/10.1126/science.1145461
Patro R, Duggal G, Love MI, Irizarry RA, Kingsford C (2017) Salmon provides fast and bias-aware quantification of transcript expression. Nat Methods 14(4):417–419. https://doi.org/10.1038/nmeth.4197
Robinson MD, McCarthy DJ, Smyth GK (2010) EdgeR: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1):139–140. https://doi.org/10.1093/bioinformatics/btp616
Rui HY, Zhang XX, Shinwari KI, Zheng LQ, Shen ZG (2018) Comparative transcriptomic analysis of two Vicia sativa L. varieties with contrasting responses to cadmium stress reveals the important role of metal transporters in cadmium tolerance. Plant Soil 423(01):241–255. https://doi.org/10.1007/s11104-017-3501-9
Scully ED, Gries T, Sarath G, Palmer NA, Baird L, Serapiglia MJ, Dien BS, Boateng AA, Ge Z, Funnell-Harris DL, Twigg P, Clemente TE, Sattler SE (2016) Overexpression of SbMyb60 impacts phenylpropanoid biosynthesis and alters secondary cell wall composition in Sorghum bicolor. Plant J Cell Mol Biol 85(3):378–395. https://doi.org/10.1111/tpj.13112
Song Y, Shan B, Zeng S, Zhang J, ** C, Liao Z, Wang T, Zeng Q, He H, Wei F, Ai Z, Su D (2021) Raw and wine processed Schisandra chinensis attenuate anxiety like behavior via modulating gut microbiota and lipid metabolism pathway. J Ethnopharmacol 266:113426. https://doi.org/10.1016/j.jep.2020.113426
Sun D, Li HB, Li Q, Piao ZY (2013) Dynamic changes of endogenous IAA, ABA and GA 3 contents during somatic embryogenesis of Schisandra chinensis. Plant Physiol J 49(01):70–74. https://doi.org/10.13592/j.cnki.ppj.2013.01.008
Wang ZZ (2010) Study on the mechanism of underground transverse stalk of Schisandra chinensis. Chinese Academy of Agricultural Sciences, Bei**g
Wei DX (2017) Study on somatic embryo induction and callus redifferentiation of Paeonia lactiflora. Bei**g Forestry University, Bei**g
Xu FG (2021) The mechanism of OsARF18 regulating rice root development and phosphorus uptake. Henan Agricultural University, Zhengzhou
Yan S, Zhang SY, Xu K, Yuan JC, Li XG, Zhou JT, Cheng CG, Zhao DY (2019) Changes of polyamines, endogenous hormones and related oxidase activities in the rooting process of Pyrus betulaefolia. J Fruit Sci 36(03):318–326. https://doi.org/10.13925/j.cnki.gsxb.20180372
Yang JL, Niu YD, Yang CP, Liu GF, Li CH (2011) Induction of somatic embryogenesis from female flower buds of elite Schisandra chinensis. Plant Cell Tiss Org 106(3):391–399. https://doi.org/10.1007/s11240-011-9935-5
Yu B, Huang LL, Zhu Y, Zhu GF, Sun YB (2020) Embryogenic callus induction and efficient plant regeneration of young pedicel of Hippeastrum vittatum. Acta Hort Sinica 47(05):907–915. https://doi.org/10.16420/j.issn.0513-353x.2019-0672
Zhou XM (2008) Study on somatic embryogenesis of tree peony. Bei**g Forestry University, Bei**g
Funding
This work was supported by the Talent Introduction Fund of Jilin Agricultural University (Grant number 0214–202022920) and the Research Project in the Jilin Provincial Science and Technology Department (Grant number 20210204083YY). We thank LC-Bio Technology Co. Ltd. (Hangzhou, China) for transcriptome sequencing.
Author information
Authors and Affiliations
Contributions
DS: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing—original draft, Writing—review and editing. SZ: Data curation, Formal analysis, Investigation. JA: Conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, resources. ZW: Conceptualization, Investigation, Methodology, Project administration. GS: Investigation, Data curation, Formal analysis, Methodology. JG: Conceptualization, formal analysis, methodology. XS: Investigation, Project administration. ML and YL: Investigation, Formal analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Qiao-Chun Wang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, D., Zhang, S., Ai, J. et al. Physiological and molecular mechanisms of radicle development of somatic embryos in Schisandra chinensis cultured in the dark. Plant Cell Tiss Organ Cult 157, 1 (2024). https://doi.org/10.1007/s11240-023-02662-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-023-02662-9