Abstract
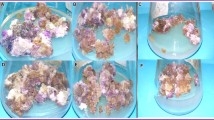
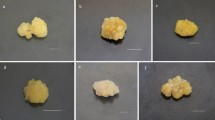
Light is one of the major abiotic factors which directly influence plant growth, development, and secondary metabolites accumulation in plants. The current study was designed to investigate the stimulatory effect of LEDs (light-emitting diodes), on biomass accumulation, pigment concentration, and bioactive metabolites production in Operculina turpethum callus culture. A 3.9-fold increase in the biomass accumulation was observed in callus culture grown under blue LED (FW: 13.1 ± 0.19 g/50 ml) whereas reduction in biomass was recorded for callus grown under dark conditions (FW: 2.1 ± 0.01 g/50 ml) when compared to the control (FW: 3.3 ± 0.04 g/50 ml). Furthermore, blue light was found to enhance the total phenolics (61.303 ± 0.32 mg GAE/g DW) and flavonoids (TFC: 26.48 ± 0.68 mg QE/g DW) synthesis in the callus culture among all the tested lights. Again, callus culture placed under blue light showed the highest in vitro antioxidant activities as compared to other lights. However, higher superoxide dismutase (SOD) and catalase (CAT) enzyme expression were observed for the callus culture grown under dark. Quantification of phytochemicals through HPLC revealed that optimum production of Gallic acid (3.3 ± 0.06 mg g−1 DW), Quercetin (0.92 ± 0.004 mg g−1 DW) Coumarin (5.3 ± 0.01 mg g−1 DW), and Salicylic acid (4.9 ± 0.03 mg g−1 DW) was observed under blue light treated callus cultures of O. turpethum. The results of our study demonstrated that the application of multispectral lights is a feasible alternative strategy for the enhanced biomass and secondary metabolite production in the callus culture of O. turpethum.
Key message
The application of abiotic elicitors such as light has been shown to be an effective alternative for the enhanced biomass production, stimulation of secondary metabolite content and in vitro antioxidant activity of callus culture of Operculina turpethum (L.)





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
Abidi F, Girault T, Douillet O et al (2013) Blue light on Rose photosynthesis and photomorphogenesis. Plant Biol. https://doi.org/10.1111/j.1438-8677.2012.00603.x
Acharya J, Rechner O, Neugart S et al (2016) Effects of light-emitting diode treatments on Brevicoryne brassicae performance mediated by secondary metabolites in Brussels sprouts. J Plant Dis Prot 123:321–330. https://doi.org/10.1007/s41348-016-0029-9
Adil M, Ren X, Jeong BR (2019) Light elicited growth, antioxidant enzymes activities and production of medicinal compounds in callus culture of Cnidium officinale Makino. J Photochem Photobiol B Biol 196:111509. https://doi.org/10.1016/j.jphotobiol.2019.05.006
Alam MJ, Alam I, Sharmin SA et al (2010) Micropropagation and antimicrobial activity of Operculina turpethum (syn. Ipomoea turpethum), an endangered medicinal plant. Plant Omics 3:40–46
Ali M, Abbasi BH (2014) Light-induced fluctuations in biomass accumulation, secondary metabolites production and antioxidant activity in cell suspension cu. J Photochem Photobiol B Biol 140:223–227. https://doi.org/10.1016/j.jphotobiol.2014.08.008
Ashry NA, Mohamed HI (2012) Impact of secondary metabolites and related enzymes in flax resistance and/or susceptibility to powdery mildew. Afr J Biotechnol 11
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Cioć M, Szewczyk A, Żupnik M et al (2018) LED lighting affects plant growth, morphogenesis and phytochemical contents of Myrtus communis L. in vitro. Plant Cell, Tissue Organ Cult 132:433–447. https://doi.org/10.1007/s11240-017-1340-2
Coelho AD, de Souza CK, Bertolucci SKV et al (2021) Wavelength and light intensity enhance growth, phytochemical contents and antioxidant activity in micropropagated plantlets of Urtica dioica L. Plant Cell Tissue Organ Cult 145:59–74. https://doi.org/10.1007/s11240-020-01992-2
Csepregi K, Hideg É (2018) Phenolic compound diversity explored in the context of photo-oxidative stress protection. Phytochem Anal 29:129–136
Dutta Gupta S, Jatothu B (2013) Fundamentals and applications of light-emitting diodes (LEDs) in in vitro plant growth and morphogenesis. Plant Biotechnol. Rep. 7
Fazal H, Abbasi BH, Ahmad N et al (2016) Correlation of different spectral lights with biomass accumulation and production of antioxidant secondary metabolites in callus cultures of medicinally important Prunella vulgaris L. J Photochem Photobiol B Biol 159:1–7. https://doi.org/10.1016/j.jphotobiol.2016.03.008
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Gurav A, Mhase A, Sinha R et al (2018) Micropropagation of Operculina turpethum (L.) Silva Manso through Nodal Segment, Apical Bud and Cotyledon. J Drug Res Ayurvedic Sci 3:71–76. https://doi.org/10.5005/jp-journals-10059-0038
Hasan D-M, Bashir T, Ghosh R et al (2017) An overview of LEDs’ effects on the production of bioactive compounds and crop quality. Molecules. https://doi.org/10.3390/molecules22091420
Heo J-W, Kang D-H, Bang H-S et al (2012) Early growth, pigmentation, protein content, and phenylalanine ammonia-lyase activity of red curled lettuces grown under different lighting conditions. Korean Soc Hortic Sci 30:6–12
Jena B, Biswal B, Giri AK et al (2020) Rapid in vitro propagation and genetic fidelity evaluation of medicinally important mexican coriander (Eryngium Foetidum L.)”. Plant Cell Biotechnol Mol Biol 21:79–88
Kamiya A, Ikegami I, Hase E (1983) Effects of light on chlorophyll formation in cultured tobacco cells, I. I. blue light effect on 5-aminolevulinic acid formation. Plant Cell Physiol 24:799–809
Kapoor S, Raghuvanshi R, Bhardwaj P et al (2018) Influence of light quality on growth, secondary metabolites production and antioxidant activity in callus culture of Rhodiola imbricata Edgew. J Photochem Photobiol B Biol 183:258–265. https://doi.org/10.1016/j.jphotobiol.2018.04.018
Kar D, Pattanaik PK, Acharya L et al (2014) Assessment of genetic diversity among some elite cultivars of ginger (Zingiber officinale Rosc.) using isozyme and protein markers. Braz J Bot 37:469–479. https://doi.org/10.1007/s40415-014-0097-7
Khan M, Abbasi B, Ali H et al (2015) Temporal variations in metabolite profiles at different growth phases during somatic embryogenesis of Silybum marianum L. Plant Cell Tissue Organ Cult. https://doi.org/10.1007/s11240-014-0587-0
Khurshid R, Ullah MA, Tungmunnithum D et al (2020) Lights triggered differential accumulation of antioxidant and antidiabetic secondary metabolites in callus culture of Eclipta alba L. PLoS ONE 15:e0233963
Kim D-O, Chun O, Kim Y et al (2003) Quantification of polyphenolics and their antioxidant capacity in fresh plums. J Agric Food Chem 51(22):615–6509
Kim YJ, Kim YB, Li X et al (2015) Accumulation of phenylpropanoids by white, blue, and red light irradiation and their organ-specific distribution in chinese cabbage (Brassica rapa ssp. pekinensis). J Agric Food Chem 63:6772–6778. https://doi.org/10.1021/acs.jafc.5b02086
Kong S-G, Wada M (2016) Molecular basis of chloroplast photorelocation movement. J Plant Res 129:159–166. https://doi.org/10.1007/s10265-016-0788-1
Lee S-W, Seo JM, Lee M-K et al (2014) Influence of different LED lamps on the production of phenolic compounds in common and Tartary buckwheat sprouts. Ind Crops Prod 54:320–326. https://doi.org/10.1016/j.indcrop.2014.01.024
Li Q, Kubota C (2009) Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Environ Exp Bot 67:59–64
Lian TT, Cha S-Y, Moe MM et al (2019) Effects of different colored LEDs on the enhancement of biologically active ingredients in callus cultures of Gynura procumbens (Lour.) Merr. Molecules 24:4336. https://doi.org/10.3390/molecules24234336
Loi M, Villani A, Paciolla F et al (2021) Challenges and opportunities of light-emitting diode (LED) as key to modulate antioxidant compounds in plants. A review. Antioxidants. https://doi.org/10.3390/antiox10010042
Ma G, Zhang L, Kato M et al (2012) Effect of blue and red LED light irradiation on β-cryptoxanthin accumulation in the flavedo of citrus fruits. J Agric Food Chem 60:197–201. https://doi.org/10.1021/jf203364m
Ma G, Zhang L, Kato M et al (2015) Effect of the combination of ethylene and red LED light irradiation on carotenoid accumulation and carotenogenic gene expression in the flavedo of citrus fruit. Postharvest Biol Technol 99:99–104. https://doi.org/10.1016/j.postharvbio.2014.08.002
Manivannan A, Soundararajan P, Halimah N et al (2015) Blue LED light enhances growth, phytochemical contents, and antioxidant enzyme activities of Rehmannia glutinosa cultured in vitro. Hortic Environ Biotechnol 56:105–113. https://doi.org/10.1007/s13580-015-0114-1
Matkowski A (2008) Plant in vitro culture for the production of antioxidants–a review. Biotechnol Adv 26:548–560. https://doi.org/10.1016/j.biotechadv.2008.07.001
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7(9):10–405
Mohammad S, Khan MA, Ali A et al (2019) Feasible production of biomass and natural antioxidants through callus cultures in response to varying light intensities in olive (Olea europaea. L) cult. Arbosana. J Photochem Photobiol B Biol 193:140–147. https://doi.org/10.1016/j.jphotobiol.2019.03.001
Nazir M, Nazir M, Ullah MA et al (2020) Light-mediated biosynthesis of phenylpropanoid metabolites and antioxidant potential in callus cultures of purple basil (Ocimum basilicum L. var. purpurascens). Plant Cell, Tissue, Organ Cult 142:107–120. https://doi.org/10.1007/s11240-020-01844-z
Petrillo E, Godoy Herz MA, Fuchs A et al (2014) A chloroplast retrograde signal regulates nuclear alternative splicing. Science 344:427–430. https://doi.org/10.1126/science.1250322
Richter G, Wessel K (1985) Red light inhibits blue light-induced chloroplast development in cultured plant cells at the mRNA level. Plant Mol Biol 5:175–182. https://doi.org/10.1007/BF00015681
Silva ST, Silva ST, Bertolucci SKV et al (2017) Effect of light and natural ventilation systems on the growth parameters and carvacrol content in the in vitro cultures of Plectranthus amboinicus (Lour) Spreng. Plant Cell, Tissue, Organ Cult 129:501–510. https://doi.org/10.1007/s11240-017-1195-6
Singleton V, Rossi J (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagent. Am J Enol Vitic 16:144–158
Soundararajan P, Manivannan A, Park YG et al (2015) Silicon alleviates salt stress by modulating antioxidant enzyme activities in Dianthus caryophyllus ‘Tula.’ Hortic Environ Biotechnol 56:233–239. https://doi.org/10.1007/s13580-015-0111-4
Szopa A, Ekiert H (2016) The importance of applied light quality on the production of lignans and phenolic acids in Schisandra chinensis (Turcz.) Baill. cultures in vitro. Plant Cell, Tissue Organ Cult 127:115–121. https://doi.org/10.1007/s11240-016-1034-1
Szopa A, Starzec A, Ekiert H (2018) The importance of monochromatic lights in the production of phenolic acids and flavonoids in shoot cultures of Aronia melanocarpa, Aronia arbutifolia and Aronia × prunifolia. J Photochem Photobiol B 179:91–97. https://doi.org/10.1016/j.jphotobiol.2018.01.005
Tariq U, Ali M, Abbasi BH (2014) Morphogenic and biochemical variations under different spectral lights in callus cultures of Artemisia absinthium L. J Photochem Photobiol B 130:264–271. https://doi.org/10.1016/j.jphotobiol.2013.11.026
Thwe AA, Kim YB, Li X et al (2014) Effects of light-emitting diodes on expression of phenylpropanoid biosynthetic genes and accumulation of phenylpropanoids in Fagopyrum tataricum sprouts. J Agric Food Chem 62:4839–4845. https://doi.org/10.1021/jf501335q
Tokarz K, Makowski W, Banasiuk R et al (2018) Response of Dionaea muscipula J. Ellis to light stress in in vitro: physiological study. Plant Cell, Tissue Organ Cult 134:65–77. https://doi.org/10.1007/s11240-018-1400-2
Usman H, Ullah MA, Jan H, et al (2020) Interactive Effects of Wide-Spectrum Monochromatic Lights on Phytochemical Production, Antioxidant and Biological Activities of Solanum xanthocarpum Callus Cultures. Mol 25
Wang M, **e B, Fu Y et al (2015) Effects of different elevated CO2 concentrations on chlorophyll contents, gas exchange, water use efficiency, and PSII activity on C3 and C4 cereal crops in a closed artificial ecosystem. Photosynth Res 126:351–362. https://doi.org/10.1007/s11120-015-0134-9
Wang P, Chen S, Gu M et al (2020) Exploration of the effects of different blue LED light intensities on flavonoid and lipid metabolism in tea plants via transcriptomics and metabolomics. Int J Mol Sci. https://doi.org/10.3390/ijms21134606
Weis JS, Jaffe MJ (1969) Photoenhancement by blue light of organogenesis in tobacco pith cultures. Physiol Plant 22:171–176
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Yang L, Stöckigt J (2010) Trends for diverse production strategies of plant medicinal alkaloids. Nat Prod Rep 27:1469–1479. https://doi.org/10.1039/C005378C
Yeo HJ, Park CH, Lee KB et al (2018) Metabolic analysis of Vigna unguiculata sprouts exposed to different light-emitting diodes. Nat Prod Commun. https://doi.org/10.1177/1934578X1801301029
Yue W, Ming Q, Lin B et al (2016) Medicinal plant cell suspension cultures: pharmaceutical applications and high-yielding strategies for the desired secondary metabolites. Crit Rev Biotechnol 36:215–232. https://doi.org/10.3109/07388551.2014.923986
Acknowledgements
All authors thank Prof. (Dr.) Manojranjan Nayak, Founder & President, Siksha ‘O’ Anusandhan (Deemed to be University), Bhubaneswar, Odisha, India for providing the required infrastructure for the study. The authors are grateful to the Head, Centre for Biotechnology and Dean, School of Pharmaceutical Sciences, Siksha O Anusandhan (Deemed to be University) for the support.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Media standardization and establishment of callus culture was performed by BB. The biochemical analysis was performed by BB, BJ. Data collection and statistical analysis were performed by BJ and AKG. Drafting and finalisation of the manuscript was carried out by BB, BJ and AKG. Supervision and critical revision were done by LA. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical approval
This research involved no human and/or animal participants.
Consent for publication
This study has not been published before and is not under consideration for publication anywhere else. All authors consent to the publication of the manuscript in PCTOC.
Additional information
Communicated by Konstantin V. Kiselev.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Biswal, B., Jena, B., Giri, A.K. et al. Monochromatic light elicited biomass accumulation, antioxidant activity, and secondary metabolite production in callus culture of Operculina turpethum (L.). Plant Cell Tiss Organ Cult 149, 123–134 (2022). https://doi.org/10.1007/s11240-022-02274-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-022-02274-9