Abstract
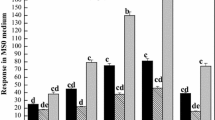
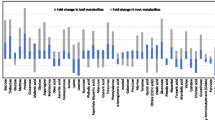
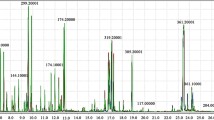
Influence of n-triacontanol (TRIA) and jasmonic acid (JA) on metabolic profiling during root morphogenesis was studied in Lycopersicon esculentum (cv. PKM-1). Proton nuclear magnetic resonance (1H NMR) based metabolomics was employed to investigate the variations in metabolic profile. Chenomx NMR suite v.8.1 was used to identify and quantify metabolites based on their respective signature spectra. The levels of 47 metabolites were monitored for 72 h at specific time intervals (0, 3, 6, 9, 12, 24, 36, 48 and 72 h). Principal component analysis was performed to determine the variations in the metabolic profile between control and treatments during in vitro rhizogenesis. TRIA was observed to promote early root emergence (24 h) and also influence the metabolic variation during rhizogenesis between 9 and 24 h post exposure. Compounds such as IAA, ATP, NADPH, UDP-N-acetylglucosamine and gallate predominated at 9 h. Unlike TRIA, JA was unable to promote an early root induction. However, it influenced the synthesis of a relatively higher concentration of IAA at 6 h when compared to ATP, NADPH and trigonelline at 9 h. In the presence of both TRIA and JA (TRIA + JA), significant changes in the metabolic profiles were observed 24 h post exposure and the rooting was observed only after 72 h. The study suggests that TRIA may accelerate in vitro rhizogenesis of cultured tomato tissues by mainly increasing the synthesis of other growth promoting metabolites. But in the presence of JA, TRIA’s effect appears to be reduced.






Similar content being viewed by others
References
Abdel-Farid I, Jahangir M, van den Hondel C, Kim H, Choi Y, Verpoorte R (2009) Fungal infection-induced metabolites in Brassica rapa. Plant Sci 176:608–615. https://doi.org/10.1016/j.plantsci.2009.01.017
Aftab T, Khan MMA, Idrees M, Naeem M, Singh M, Ram M (2010) Stimulation of crop productivity, photosynthesis and artemisinin production in Artemisia annua L. by triacontanol and gibberellic acid application. J Plant Interact 5:273–281. https://doi.org/10.1080/17429141003647137
Al-Abdallat AM, Ali-Sheikh-Omar MA, Alnemer LM (2015) Overexpression of two ATNAC3-related genes improves drought and salt tolerance in tomato (Solanum lycopersicum L.). Plant Cell Tissue Organ Cult 120:989–1001. https://doi.org/10.1007/s11240-014-0652-8
Arnao MB, Hernández-Ruiz J (2006) The physiological function of melatonin in plants. Plant Signal Behav 1:89–95
Arnao MB, Hernández-Ruiz J (2007) Melatonin promotes adventitious-and lateral root regeneration in etiolated hypocotyls of Lupinus albus L. J Pineal Res 42:147–152. https://doi.org/10.1111/j.1600-079X.2006.00396.x
Casimiro I, Marchant A, Bhalerao RP, Beeckman T, Dhooge S, Swarup R, Graham N, Inzé D, Sandberg G, Casero PJ (2001) Auxin transport promotes Arabidopsis lateral root initiation. Plant Cell 13:843–852. https://doi.org/10.1105/tpc.13.4.843
Chen X, Yuan H, Chen R, Zhu L, Du B, Weng Q, He G (2002) Isolation and characterization of triacontanol-regulated genes in rice (Oryza sativa L.): possible role of triacontanol as a plant growth stimulator. Plant Cell Physiol 43:869–876. https://doi.org/10.1093/pcp/pcf100
Chen G, Huo Y, Tan D-X, Liang Z, Zhang W, Zhang Y (2003) Melatonin in Chinese medicinal herbs. Life Sci 73:19–26. https://doi.org/10.1016/S0024-3205(03)00252-2
Csaba G, Pál K (1982) Effects of insulin, triiodothyronine, and serotonin on plant seed development. Protoplasma 110:20–22. https://doi.org/10.1007/BF01314677
de Siqueira Pinto M, Abeyratne CR, Benedito VA, Peres LEP (2017) Genetic and physiological characterization of three natural allelic variations affecting the organogenic capacity in tomato (Solanum lycopersicum cv. Micro-Tom). Plant Cell Tissue Organ Cult 129:89–103. https://doi.org/10.1007/s11240-016-1159-2
Dettmer J, Ursache R, Campilho A, Miyashima S, Belevich I, O’Regan S, Mullendore DL, Yadav SR, Lanz C, Beverina L, Papagni A, Schneeberger K, Weigel D, Stierhof YD, Moritz T, Knoblauch M, Jokitalo E, Helariutta Y (2014) CHOLINE TRANSPORTER-LIKE1 is required for sieve plate development to mediate long-distance cell-to-cell communication. Nat Commun 5:4276. https://doi.org/10.1038/ncomms5276
Evans LS (1978) Cell cycle kinetics of endoreduplication in gamma-irradiated root meristems of Pisum sativum. Am J Bot 65:1084–1090. https://doi.org/10.2307/2442325
Evans LS, Tramontano WA (1981) Is trigonelline a plant hormone? Am J Bot 68:1282–1289. https://doi.org/10.2307/2443053
Fan X, Zhang R, Cheng H (2010) Use of triacontanol in preparation of medicaments for treatment of cancers. US 7863337 B2
Fiehn O (2002) Metabolomics—the link between genotypes and phenotypes. Plant Mol Biol 48:155–171. https://doi.org/10.1023/A:1013713905833
Fliniaux O, Mesnard F, Raynaud-Le Grandic S, Baltora-Rosset S, Bienaimé C, Robins RJ, Fliniaux MA (2004) Altered nitrogen metabolism associated with de-differentiated suspension cultures derived from root cultures of Datura stramonium studied by heteronuclear multiple bond coherence (HMBC) NMR spectroscopy. J Exp Bot 55:1053–1060. https://doi.org/10.1093/jxb/erh119
Fujiwara T, Mitsuya S, Miyake H, Hattori T, Takabe T (2010) Characterization of a novel glycinebetaine/proline transporter gene expressed in the mestome sheath and lateral root cap cells in barley. Planta 232:133–143. https://doi.org/10.1007/s00425-010-1155-4
Georgiev MI, Radziszewska A, Neumann M, Marchev A, Alipieva K, Ludwig-Müller J (2015) Metabolic alterations of Verbascum nigrum L. plants and SAArT transformed roots as revealed by NMR-based metabolomics. Plant Cell Tissue Organ Cult 123:349–356. https://doi.org/10.1007/s11240-015-0840-1
Gerszberg A, Hnatuszko-Konka K, Kowalczyk T, Kononowicz AK (2015) Tomato (Solanum lycopersicum L.) in the service of biotechnology. Plant Cell Tissue Organ Cult 120:881–902. https://doi.org/10.1007/s11240-014-0664-4
Gong B, Wang X, Wei M, Yang F, Li Y, Shi Q (2016) Overexpression of S-adenosylmethionine synthetase 1 enhances tomato callus tolerance to alkali stress through polyamine and hydrogen peroxide cross-linked networks. Plant Cell Tissue Organ Cult 124:377–391. https://doi.org/10.1007/s11240-015-0901-5
Grzegorczyk I, Bilichowski I, Mikicuik-Olasik E, Wysokinska H (2005) In vitro cultures of Salvia officinalis L. as a source of antioxidant compounds. Acta Soc Bot Pol Pol Tow Bot 74:17–21. https://doi.org/10.5586/asbp.2005.003
Hangarter R, Ries SK, Carlson P (1978) Effect of triacontanol on plant cell cultures in vitro. Plant Physiol 61:855–857. https://doi.org/10.1104/pp.61.5.855
Hanover JA (2001) Glycan-dependent signaling: O-linked N-acetylglucosamine. FEBS J 15:1865–1876. https://doi.org/10.1096/fj.01-0094rev
Ivanchenko MG, Muday GK, Dubrovsky JG (2008) Ethylene–auxin interactions regulate lateral root initiation and emergence in Arabidopsis thaliana. Plant J 55:335–347. https://doi.org/10.1111/j.1365-313X.2008.03528.x
Kang S, Kang K, Lee K, Back K (2007) Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants. Plant Cell Rep 26:2009–2015. https://doi.org/10.1007/s00299-007-0405-9
Khan MIR, Asgher M, Khan NA (2014) Alleviation of salt-induced photosynthesis and growth inhibition by salicylic acid involves glycinebetaine and ethylene in mungbean (Vigna radiata L.). Plant Physiol Biochem 80:67–74. https://doi.org/10.1016/j.plaphy.2014.03.026
Khandaker MM, Faruq G, Rahman MM, Sofian-Azirun M, Boyce AN (2013) The influence of 1-triacontanol on the growth, flowering, and quality of potted bougainvillea plants (Bougainvillea glabra var. “Elizabeth Angus”) under natural conditions. Sci World J. https://doi.org/10.1155/2013/308651
Kissimon J, Tantos Á, Mészáros A, Jámbor-Benczúr E, Horváth G (1999) Stress alterations in growth parameters, pigment content and photosynthetic functions of in vitro cultured plants. Z Naturforsch C Bio Sci 54:834–839. https://doi.org/10.1515/znc-1999-9-1033
Kumaravelu G, Livingstone VD, Ramanujam M (2000) Triacontanol-induced changes in the growth, photosynthetic pigments, cell metabolites, flowering and yield of green gram. Biol Plant 43:287–290. https://doi.org/10.1023/A:1002724831619
Laskowski MJ, Williams ME, Nusbaum HC, Sussex IM (1995) Formation of lateral root meristems is a two-stage process. Development 121:3303–3310
Li S-W, Xue L, Xu S, Feng H, An L (2009) Mediators, genes and signaling in adventitious rooting. Bot Rev 75:230–247. https://doi.org/10.1007/s12229-009-9029-9
Mira MM, Wally OSD, Elhiti M, El-Shanshory A, Reddy DS, Hill RD, Stasolla C (2016) Jasmonic acid is a downstream component in the modulation of somatic embryogenesis by Arabidopsis class 2 phytoglobin. J Exp Bot 67:2231–2246. https://doi.org/10.1093/jxb/erw022
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Naeem M, Khan MMA, Siddiqui MH (2009) Triacontanol stimulates nitrogen-fixation, enzyme activities, photosynthesis, crop productivity and quality of hyacinth bean (Lablab purpureus L.). Sci Hortic 121:389–396. https://doi.org/10.1016/j.scienta.2009.02.030
Naeem M, Khan MMA, Idrees M, Aftab T (2011) Triacontanol-mediated regulation of growth and other physiological attributes, active constituents and yield of Mentha arvensis L. Plant Growth Regul 65:195–206. https://doi.org/10.1007/s10725-011-9588-8
Naeem M, Khan MMA, Moinuddin (2012) Triacontanol: a potent plant growth regulator in agriculture. J Plant Interact 7:129–142. https://doi.org/10.1080/17429145.2011.619281
Negi AS, Darokar MP, Chattopadhyay SK, Garg A, Bhattacharya AK, Srivastava V, Khanuja SP (2005) Synthesis of a novel plant growth promoter from gallic acid. Bioorg Med Chem Lett 15:1243–1247. https://doi.org/10.1016/j.bmcl.2004.11.079
Palama TL, Menard P, Fock I, Choi YH, Bourdon E, Govinden-Soulange J, Bahut M, Payet B, Verpoorte R, Kodja H (2010) Shoot differentiation from protocorm callus cultures of Vanilla planifolia (Orchidaceae): proteomic and metabolic responses at early stage. BMC Plant Biol 10:82. https://doi.org/10.1186/1471-2229-10-82
Ramakrishna A, Giridhar P, Ravishankar GA (2009) Indoleamines and calcium channels influence morphogenesis in in vitro cultures of Mimosa pudica L. Plant Signal Behav 4:1136–1141. https://doi.org/10.4161/psb.4.12.10101
Ramanarayan K, Swamy GS (2004) Triacontanol negatively modulates the jasmonic acid-stimulated proteinase inhibitors in tomato (Lycopersicon esculentum). J Plant Physiol 161:489–492. https://doi.org/10.1078/0176-1617-01260
Ravnikar M, Rode J, Gogala N, Benedicic D (1990) Regulation of organogenesis with jasmonic acid. Acta Hort 169–172. https://doi.org/10.17660/ActaHortic.1990.280.29
Reddy BO, Giridhar P, Ravishankar G (2002) The effect of triacontanol on micropropagation of Capsicum frutescens and Decalepis hamiltonii W & A. Plant Cell Tissue Organ Cult 71:253–258. https://doi.org/10.1023/A:1020342127386
Ries S (1991) Triacontanol and its second messenger 9-beta-l(+)-adenosine as plant growth substances. Plant Physiol 95:986–989. https://doi.org/10.1104/pp.95.4.986
Ries S, Wert V (1977) Growth responses of rice seedlings to triacontanol in light and dark. Planta 135:77–82. https://doi.org/10.1007/BF00387979
Ries SK, Wert VF (1982) Rapid in vivo and in vitro effects of triacontanol. J Plant Growth Regul 1:117–127
Rotem R, Heyfets A, Fingrut O, Blickstein D, Shaklai M, Flescher E (2005) Jasmonates: novel anticancer agents acting directly and selectively on human cancer cell mitochondria. Cancer Res 65:984–1993. https://doi.org/10.1158/0008-5472.can-04-3091
Ryan D, Robards K (2006) Metabolomics: the greatest omics of them all? Anal Chem 78:7954–7958. https://doi.org/10.1021/ac0614341
Saiman MZ, Mustafa NR, Choi YH, Verpoorte R, Schulte AE (2015) Metabolic alterations and distribution of five-carbon precursors in jasmonic acid-elicited Catharanthus roseus cell suspension cultures. Plant Cell Tissue Organ Cult 122:351–362. https://doi.org/10.1007/s11240-015-0773-8
Shah SH, Ali S, Jan SA, Jalal-Ud-Din, Ali GM (2015) Piercing and incubation method of in planta transformation producing stable transgenic plants by overexpressing DREB1A gene in tomato (Solanum lycopersicum Mill.). Plant Cell Tissue Organ Cult 120:1139–1157. https://doi.org/10.1007/s11240-014-0670-6
Smith AM, Coupland G, Dolan L, Harberd N, Jones J, Martin C, Sablowski R, Amey A (2009) Plant biology. Garland Science, New York
Swamy SG, Ramanarayan K, Inamdar LS, Inamdar SR (2009) Triacontanol and jasmonic acid differentially modulate the lipid organization as evidenced by the fluorescent probe behavior and 31P nuclear magnetic resonance shifts in model membranes. J Membr Biol 228:165–177. https://doi.org/10.1007/s00232-009-9198-9
Tiburcio AF, Altabella T, Bitrián M, Alcázar R (2014) The roles of polyamines during the lifespan of plants: from development to stress. Planta 240:1–18. https://doi.org/10.1007/s00425-014-2055-9
Trygg J, Holmes E, Lundstedt T (2007) Chemometrics in metabonomics. J Proteome Res 6:469–479. https://doi.org/10.1021/pr060594q
Wasternack C, Hause B (2002) Jasmonates and octadecanoids: signals in plant stress responses and development. Prog Nucleic Acid Res Mol Biol 72:165–221. https://doi.org/10.1016/S0079-6603(02)72070-9
Yaseen M, Tajuddin K (1998) Effect of plant growth regulators on yield, oil composition and artemisinin of Artemisia annua under temperate conditions. J Med Aromat Plant Sci 20:1038–1041
Zaragoza-Martínez F, Lucho-Constantino GG, Ponce-Noyola T, Esparza-García F, Poggi-Varaldo H, Cerda-García-Rojas CM, Trejo-Tapia G, Ramos-Valdivia AC (2016) Jasmonic acid stimulates the oxidative responses and triterpene production in Jatropha curcas cell suspension cultures through mevalonate as biosynthetic precursor. Plant Cell Tissue Organ Cult 127:47. https://doi.org/10.1007/s11240-016-1028-z
Zhao J, Davis LC, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23:283–333. https://doi.org/10.1016/j.biotechadv.2005.01.003
Zulak KG, Weljie AM, Vogel HJ, Facchini PJ (2008) Quantitative 1H NMR metabolomics reveals extensive metabolic reprogramming of primary and secondary metabolism in elicitor-treated opium poppy cell cultures. BMC Plant Biol 8:5. https://doi.org/10.1186/1471-2229-8-5
Acknowledgements
We would like to thank Dr. Leela Iyengar (Chief Scientific Officer (Retd), I.I.T. Kanpur) for her constant support and encouragement. We acknowledge the help of the NMR facility supported by DST at Indian Institute of Science, Bangalore, where all the NMR spectra were recorded.
Author information
Authors and Affiliations
Contributions
Study conception and design: GSS and SD. Acquisition of data: MS and SKG. Analysis and interpretation of data: MS. Drafting of manuscript: MS. Critical revision: GSS and SD.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sergio J. Ochatt.
Rights and permissions
About this article
Cite this article
Soundararajan, M., Swamy, G.S., Gaonkar, S.K. et al. Influence of triacontanol and jasmonic acid on metabolomics during early stages of root induction in cultured tissue of tomato (Lycopersicon esculentum) . Plant Cell Tiss Organ Cult 133, 147–157 (2018). https://doi.org/10.1007/s11240-017-1369-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-017-1369-2