Abstract
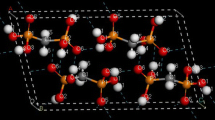
A computational study of dimers of heterosubstituted sumanenes has been carried out using a dispersion-corrected density functional theory method. In the heterosubstituted systems, the three bridging CH2 groups of sumanene have been replaced by O, NH, and S. For each dimer system, two motifs, staggered and eclipsed forms, were considered. The most stable geometry was the staggered stacked concave–convex motif where one monomer was rotated by 60° from the eclipsed configuration. The calculated binding energies and equilibrium distances of the staggered concave–convex dimers are predicted to be 20.1 kcal/mol and 3.7 Å for the parent sumanene molecule, 17.4 kcal/mol and 3.8 Å, 12.3 kcal/mol and 3.7 Å, and 16.6 kcal/mol and 3.7 Å for the NH-, O-, and S-substituted analogs, respectively. The binding energies of the dimers have been analyzed in terms of dipole–dipole contributions, dispersion contributions, and C–H···π interactions.






Similar content being viewed by others
References
Ruiz C, Garcia-Frutos EM, Hennrich G, Gomez-Lor BJ (2012) Phys Chem Lett 3:1428
Mas-Torrent M, Rovira C (2008) Chem Soc Rev 37:827
Segura JL, Martin N, Guldi DM (2005) Chem Soc Rev 34:31
Wang Q, Ma D (2010) Chem Soc Rev 39:2387
Nourmohammadian F, Wu T, Branda NR (2011) Chem Commun (Cambridge UK) 47:10954
McQuade DT, Pullen AE, Swager TM (2000) Chem Rev (Washington DC) 100:2537
Coropceanu V, Cornil J, Da SFDA, Olivier Y, Silbey R, Bredas J-L (2007) Chem Rev (Washington DC) 107:926
Sygula A, Fronczek FR, Sygula R, Rabideau PW, Olmstead MM (2007) J Am Chem Soc 129:3842
Mück-Lichtenfeld C, Grimme S, Kobryn L, Sygula A (2010) Phys Chem Chem Phys 12:7091
Sygula A, Collier WE (2011) In: Petrukhina LA, Scott ET (eds) Fragments of fullerenes and carbon nanotubes: design synthesis, unusual reactions, and coordination chemistry (Chapter 1). Wiley, New York, pp 1–40
Sygula A, Saebo S (2008) Int J Quantum Chem 109:65
Janowski T, Pulay P, Karrunarathna AAS, Sygula A, Saebo S (2011) Chem Phys Lett 512:155
Karaunarathna AAS, Saebo S (2014) Struct Chem 25:1831
Tang ML, Okamoto T, Bao Z (2006) J Am Chem Soc 128:16002
Briseno AL, Mannsfeld SCB, Lu X, **ong Y, Jenekhe SA, Bao Z, **a Y (2007) Nano Lett 7:668
Roberts JD, Streitwieser A Jr, Regan CM (1952) J Am Chem Soc 74:4579
Barth WE, Lawton RG (1966) J Am Chem Soc 88:380
Lawton RG, Barth WE (1971) J Am Chem Soc 93:1730
Sakurai T, Daiko T, Hirao T (2003) Science 187:1878
Sakurai T, Daiko T, Sakane H, Amaya T, Hirao T (2005) J Am Chem Soc 124:10887
Imamura K, Takimiya K, Aso Y, Otsubo T (1999) Chem Commun 1859
Higashibayashi S, Sakura H (2011) Chem Lett 40:122
Sastry GN, Priyakumar UD (2001) J Chem Soc Perkin Trans 2:30
Priyakumar UD, Sastry GN (2001) J Org Chem 66:6523
Jaafar R, Pignedoli CAQ, Bussi G, Ait-Mansour K, Groening O, Amaya T, Hirao T, Fasel R, Ruffleux P (2014) J Am Chem Soc 136:13666
Cauet E, Jacquemin D (2012) Chem Phys Lett 519–520:49
Armakovic S, Armakovic SJ, Setrajcic JP, Holodkov V (2014) J Mol Model 20:2538
Armakovic S, Armakovic SJ, Setrajcic JP, Dzambas D (2013) J Mol Model 19:1153
Josa D, Rodriguez-Otero J, Cabaleiro-Lago EM, Santos LA, Ramalho TC (2014) J Phys Chem A 118:9521
Grimme S (2006) J Comput Chem 27:1787
Grimme S (2004) J Comput Chem 25:146328
Godbout N, Salahub DR, Andzelm J, Wimmer E (1992) Can J Chem 70:560
Lee C, Yang W, Parr RG (1988) Phys Rev B Condens Matter 37:785
Becke AD (1993) J Chem Phys 98:5648
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650
McLean AD, Chandler GS (1980) J Chem Phys 72:5639
Dunning TH Jr (1989) J Chem Phys 90:1007
Woon DE, Dunning TH Jr (1995) J Chem Phys 103:4572
Boys SF, Bernardi F (1970) Mol Phys 19:553
Haddon RC, Scott LT (1986) Pure Appl Chem 58:137
Haddon RC (1988) Acc Chem Res 21:243
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford, GB
Denis PA (2011) Chem Phys Lett 516:82
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karunarathna, A.A.S., Saebo, S. Computational studies of π–π interactions in dimers of heterosubstituted sumanenes. Struct Chem 26, 1689–1695 (2015). https://doi.org/10.1007/s11224-015-0591-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-015-0591-y