Abstract
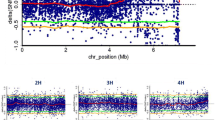
The pathogenic fungus, Alternaria porri (Ellis) Cifferi, that causes purple blotch (PB) disease, is a major constraint to production of onion and allied crops worldwide. In the present study, bulk segregant RNA sequencing (BSR-Seq) was used to analyze onion cultivar Arka Kalyan (resistant parent), Agrifound Rose (susceptible parent), and two sets of their bulks (20 homozygous resistant and 20 susceptible) from F6 RIL population to identify a potential region for resistance to PB. Transcript profiling resulted in 278.08 million clean reads from 8 libraries. Comparative expression analysis revealed 755 differentially expressed genes (DEGs) including 492 upregulated and 263 downregulated sequences. Bulk frequency ratio (BFR) was estimated between resistant and susceptible bulk, and 2963 common SNPs with BFR > 6 were detected on 1439 transcripts. Euclidean distance association analysis identified a 7.3 Mb resistance specific candidate region in the long arm of chromosome 6. Using RNA-Seq, 23 DEGs were reported in the candidate region in chromosome 6, including ACCL_20794 (Chr6: 187,639,724–187,643,297), a disease-resistant protein of the CC-NBS-LRR class, whose expression was elevated in the resistant pools following PB treatment. The ACCL_20794 gene was cloned and based on the sequences from the two parents, a single amino acid mutation—histidine (H) to serine (S) was detected in the resistance genotype Arka Kalyan. Quantitative reverse transcription (qRT)-PCR further demonstrated significantly differential expression of ACCL_20794 in the two parents as well as the RIL bulks. This indicates that ACCL_20794 might be the candidate resistance gene ApR1 and is implicated in the PB resistance response.






Similar content being viewed by others
Data of Availability
No datasets were generated or analysed during the current study.
References
Abubaker L, Ado SG (2008) Heterosis of purple blotch (Alternaria porri (Ellis) Cif.) resistance, yield and earliness in tropical onions (Allium cepa L.). Euphytica 164:63–74. https://doi.org/10.1007/s10681-007-9639-4
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11:R106. https://doi.org/10.1186/gb-2010-11-10-r106
Ascurra TY, Lin X, Wolters PJ, Vleeshouwers VGAA (2021) Identification of Solanum immune receptors by bulked segregant RNA-Seq and high-throughput recombinant screening. Methods Mol Biol 2354:315–330. https://doi.org/10.1007/978-1-0716-1609-3_15
Bezerra-Neto JP, Araújo FC, Ferreira-Neto JRC, Silva RLO, Borges ANC, Matos MKS, Silva JB, Silva MD, Kido EA (2020) Benko-Iseppon AM (2020) NBS-LRR genes-plant health sentinels: structure, roles, evolution and biotechnological applications. In: Poltronieri P, Hong Y (eds) Applied plant biotechnology for improving resistance to biotic stress. Academic Press, Cambridge, UK, pp 63–120
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 30(15):2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Cao Y, Diao Q, Chen Y, ** H, Zhang Y, Zhang H (2021) Development of KASP markers and identification of a QTL underlying powdery mildew resistance in melon (Cucumis melo L.) by bulked segregant analysis and RNA-Seq. Front Plant Sci 11:593207. https://doi.org/10.3389/fpls.2020.593207
Chagné D, Batley J, Edwards D, Forster JW (2007) Single nucleotide polymorphism genoty** in plants, In: Oraguzie, N.C., Rikkerink, E.H.A., Gardiner, S.E., De Silva, H.N. (eds) Association map** in plants. Springer, New York, NY. https://doi.org/10.1007/978-0-387-36011-9_5
Chand SK, Nanda S, Joshi RK (2018) Genetics and molecular map** of a novel purple blotch-resistant gene ApR1 in onion (Allium cepa L.) using STS and SSR markers. Mol Breed 38:109. https://doi.org/10.1007/s11032-018-0864-4
Cingolani P, Platts A, le Wang L, Coon M, Nguyen T, Wang L, Land SJ, Lu X, Ruden DM (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 6(2):80–92. https://doi.org/10.4161/fly.19695
Dar AA, Sharma S, Mahajan R, Mushtaq M, Salathia A, Ahamad S, Sharma JP (2020) Overview of purple blotch disease and understanding its management through chemical, biological and genetic approaches. J Int Agri 19:3013–3024. https://doi.org/10.1016/S2095-3119(20)63285-3
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Evoor S, Veeregowda R, Ganeshan G (2007) Genetics of resistance to purple blotch disease in onion (Allium cepa L.). Karnataka J Agric Sci 20:810–812
Finkers R, Kaauwen MV, Ament K, Burger-Meijer K, Egging R, Huits H, Kodde L, Kroon L, Shigyo M, Sato S, Vosman B, Workum WV, Scholten O (2021) Insights from the first genome assembly of onion (Allium cepa). G3 Genes Genomes Genetics 11:jkab243. https://doi.org/10.1093/g3journal/jkab243
Fleming MB, Miller T, Fu W, Li Z, Gasic K, Saski C (2022) Ppe.XapF: high throughput KASP assays to identify fruit response to Xanthomonas arboricola pv. pruni (Xap) in peach. PLoS One 17:e0264543. https://doi.org/10.1371/journal.pone.0264543
Fujito S, Akyol TY, Mukae T, Wako T, Yamashita KI, Tsukazaki H, Hirakawa H, Tanaka K, Mine Y, Sato S, Shigyo M (2021) Construction of a high-density linkage map and graphical representation of the arrangement of transcriptome-based unigene markers on the chromosomes of onion. Allium Cepa l BMC Genomics 22:481. https://doi.org/10.1186/s12864-021-07803-y
Gothandapani S, Boopalakrishnan G, Prabhakaran N, Chethana BS, Aravindhan M, Saravanakumar M (2015) Evaluation of entomopathogenic fungus against Alternaria porri (Ellis) causing purple blotch disease of onion. Arch Phytopathol Plant Prot 48:135–144. https://doi.org/10.1080/03235408.2014.884532
Hu J, Li J, Wu P, Li Y, Qiu D, Qu Y, **e J, Zhang H, Yang L, Fu T, Yu Y, Li M, Liu H, Zhu T, Zhou Y, Liu Z, Li H (2019) Development of SNP, KASP, and SSR markers by BSR-Seq technology for saturation of genetic linkage map and efficient detection of wheat powdery mildew resistance gene Pm61. Int J Mol Sci 20:750. https://doi.org/10.3390/ijms20030750
Khandagale K, Roylawar P, Kulkarni O, Khambalkar P, Ade A, Kulkarni A, Singh M, Gawande S (2022) Comparative transcriptome analysis of onion in response to infection by Alternaria porri (Ellis) Cifferi. Front Plant Sci 13:857306. https://doi.org/10.3389/fpls.2022.857306
Kourelis J, van der Hoorn RAL (2018) Defended to the nines: 25 years of resistance gene cloning identifies nine mechanisms for R protein function. Plant Cell 30:285–299. https://doi.org/10.1105/tpc.17.00579
Kumar S, Banks TW, Cloutier S (2012) SNP Discovery through next-generation sequencing and its applications. Int J Plant Genomics 2012:831460. https://doi.org/10.1155/2012/831460
Labate JA, Glaubitz JC, Havey MJ (2020) Genoty** by sequencing for SNP marker development in onion. Genome 63:607–613. https://doi.org/10.1139/gen-2020-0011
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) 1000 genome project data processing subgroup. The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Liu S, Yeh CT, Tang HM, Nettleton D, Schnable P (2012) Gene map** via bulked segregant RNA-Seq (BSR-Seq). PLoS ONE 7:e36406. https://doi.org/10.1371/journal.pone.0036406
Liu G, Zhao T, You X, Jiang J, Li J, Xu X (2019) Molecular map** of the Cf-10 gene by combining SNP/InDel-index and linkage analysis in tomato (Solanum lycopersicum). BMC Plant Biol 19:15. https://doi.org/10.1186/s12870-018-1616-7
Liu Z, Lu T, Feng C, Zhang H, Xu Z, Correl JC, Qian W (2021) Fine map** and molecular marker development of the Fs gene controlling fruit spines in spinach (Spinacia oleracea L.). Theor Appl Genet 134:1319–1328. https://doi.org/10.1007/s00122-021-03772-8
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2CT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu H, Lin T, Klein J, Wang S, Qi J, Zhou Q, Sun J, Zhang Z, Weng Y, Huang S (2014) QTL-seq identifies an early flowering QTL located near flowering locus T in cucumber. Theor Appl Genet 127:1491–1499. https://doi.org/10.1007/s00122-014-2313-z
Mammadov J, Aggarwal R, Buyyarapu R, Kumpatla S (2012) SNP markers and their impact on plant breeding. Int J Plant Genom 728398. https://doi.org/10.1155/2012/728398
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303. https://doi.org/10.1101/gr.107524.110
Nanda S, Chand SK, Mandal P, Tripathi P, Joshi RK (2016) Identification of novel source of resistance and differential response of Allium genotypes to purple blotch pathogen, Alternaria porri (Ellis) Ciferri. Plant Pathol J 32:519–527. https://doi.org/10.5423/PPJ.OA.02.2016.0034
Nguyen K, Grondin A, Courtois B, Gantet P (2018) Next generation sequencing accelerates crop gene discovery. Trends in Plant Sci 24:263–274. https://doi.org/10.1016/j.tplants.2018.11.008
Ning X, Yunyu W, Aihong L (2020) Strategy for use of rice blast resistance genes in rice molecular breeding. Rice Sci 27:263–277. https://doi.org/10.1016/j.rsci.2020.05.003
Prakasham V, Sharma P (2012) Trichoderma harzianum (Th-3), a potential strain to manage the purple blotch of onion (Allium cepa L.) caused by Alternaria porri under north Indian plains. J Agric Sci 4:266–272. https://doi.org/10.5539/jas.v4n10p266
Ramirez-Gonzalez RH, Segovia V, Bird N, Fenwick P, Holdgate S, Berry S, Jack P, Caccamo M, Uauy C (2015) RNA-Seq bulked segregant analysis enables the identification of high-resolutiongenetic markers for breeding in hexaploid wheat. Plant Biotechnol J 13:613–624. https://doi.org/10.1111/pbi.12281
Ray S (2014) Next generation sequencing technologies for next generation plant breeding. Front Plant Sci. https://doi.org/10.3389/fpls.2014.00367
Rout E, Nanda S, Nayak S, Joshi RK (2014) Molecular characterization of NBS encoding resistance genes and induction analysis of a putative candidate gene linked to Fusarium basal rot resistance in Allium sativum. Physiol Mol Plant Pathol 85:15–24. https://doi.org/10.1016/j.pmpp.2013.11.003
Ruff TM, Marston EJ, Eagle JD, Sthapit SR, Hooker MA, Skinner DZ, See DR (2020) Genoty** by multiplexed sequencing (GMS): a customizable platform for genomic selection. PLoS ONE 15:e0229207. https://doi.org/10.1371/journal.pone.0229207
Sahoo J, Mishra R, Joshi RK (2023) Development of SNP markers linked to purple blotch resistance for marker-assisted selection in onion (Allium cepa L.) breeding, 3 Biotech. https://doi.org/10.1007/s13205-023-03562-7
Sahu PK, Sao R, Mondal S, Vishwakarma G, Gupta SK, Kumar V, Singh S, Sharma D, Das BK (2020) Next generation sequencing based forward genetics approaches for identification and map** of causal mutations in crop plants: a comprehensive review. Plants 9:1355. https://doi.org/10.3390/plants9101355
Saxesena RR, Mishra VK, Chand R, Kumar U, Chowdhury AK, Bhati J, Budhlakoti N, Joshi AK (2022) SNP discovery using BSR-Seq approach for spot blotch resistance in wheat (Triticum aestivum L.), an essential crop for food security. Front Genet 13:859676. https://doi.org/10.3389/fgene.2022.859676
Shao ZQ, Xue JY, Wu P, Zhang YM, Wu Y, Hang YY, Wang B, Chen JQ (2016) Large-scale analyses of angiosperm nucleotide-binding site-leucine-rich repeat genes reveal three anciently diverged classes with distinct evolutionary patterns. Plant Physiol 170:2095–2109. https://doi.org/10.1104/pp.15.01487
Singh VK, Khan AW, Saxena RK, Kumar V, Kale SM, Sinha P, Chitikineni A, Pazhamala LT, Garg V, Sharma M, Sameer Kumar CV, Parupalli S, Vechalapu S, Patil S, Muniswamy S, Ghanta A, Yamini KN, Dharmaraj PS, Varshney RK (2016) Next-generation sequencing for identification of candidate genes for Fusarium wilt and sterility mosaic disease in pigeonpea (Cajanus cajan). Plant Biotechnol J 14:1183–1194. https://doi.org/10.1111/pbi.12470
Singh L, Anderson JA, Chen J, Gill BS, Tiwari VK, Rawat N (2019) Development and validation of a perfect KASP marker for Fusarium head blight resistance gene Fhb1 in wheat. Plant Pathol J 35:200–207. https://doi.org/10.5423/PPJ.OA.01.2019.0018
Singh H, Khar A (2021) Perspectives of onion hybrid breeding in India: an overview. Indian J Agri Sci 91:1426–1432. https://doi.org/10.56093/ijas.v9li10.117404
Steele JFC, Hughes RK, Banfield MJ (2019) Structural and biochemical studies of an NB-ARC domain from a plant NLR immune receptor. PLoS ONE 14:e0221226. https://doi.org/10.1371/journal.pone.0221226
Takagi H, Abe A, Yoshida K, Kosugi S, Natsume S, Mitsuoka C, Uemura A, Utsushi H, Tamiru M, Takuno S, Innan H, Cano LM, Kamoun S, Terauchi R (2013) QTL-seq: rapid map** of quantitative trait loci in rice by whole genome resequencing of DNA from two bulked populations. Plant J 74:174–183. https://doi.org/10.1111/tpj.12105
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515. https://doi.org/10.1038/nbt.1621
Trick M, Adamski NM, Mugford SG, Jiang CC, Febrer M, Uauy C (2012) Combining SNP discovery from next-generation sequencing data with bulked segregant analysis (BSA) to fine-map genes in polyploid wheat. BMC Plant Biol 12:14. https://doi.org/10.1186/1471-2229-12-14
Tripathi P, Priyadarshini A, Das SK, Sahoo BB, Dash DK (2013) Evaluation of onion (Allium cepa L.) genotypes for tolerance to thrips (Thrips tabaci L.) and purple blotch (Alternaria porri (E) Ciferri). Int J Biores Stress Manage 4:561–564. https://doi.org/10.13140/RG.2.2.11126.63042
Veeraghanti KS, Naik BG, Hegde KT (2017) Management of purple blotch disease of onion under field condition. J Pharmacogn Phytochem 6:1768–1769
Wang Z, Gerstein M, Snyder M (2009) RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10(1):57–63. https://doi.org/10.1038/nrg2484
Wang J, Li Y, Xu F, Xu H, Han Z, Liu L, Song Y (2022a) Candidate powdery mildew resistance gene in wheat landrace cultivar Hongyoumai discovered using SLAF and BSR-seq. BMC Plant Biol 22:83. https://doi.org/10.1186/s12870-022-03448-5
Wang S, Wang X, Zhang R, Liu Q, Sun X, Wang J, Wang Y, **ng J, Liu Y, Zhao Y, Shi Z, Su A, Li C, **ao S, Jiao Y, Li Z, Wang R, Song W, Zhao J (2022b) RppM, encoding a typical CC-NBS-LRR protein, confers resistance to southern corn rust in maize. Front Plant Sci 13:951318. https://doi.org/10.3389/fpls.2022.951318
Wu P, **e J, Hu J, Qiu D, Liu Z, Li J, Li M, Zhang H, Yang L, Liu H, Zhou Y, Zhang Z, Li H (2018) Development of molecular markers linked to powdery mildew resistance gene Pm4b by combining SNP discovery from transcriptome sequencing data with bulked segregant analysis (BSR-Seq) in wheat. Front Plant Sci 9:95. https://doi.org/10.3389/fpls.2018.00095
Wu Q, Su Y, Pan YB, Xu F, Zou W, Que B, Lin P, Sun T, Grisham MP, Xu L, Que Y (2022) Genetic identification of SNP markers and candidate genes associated with sugarcane smut resistance using BSR-Seq. Front Plant Sci 13:1035266. https://doi.org/10.3389/fpls.2022.1035266
**e J, Guo G, Wang Y, Hu T, Wang L, Li J, Qiu D, Li Y, Wu Q, Lu P, Chen Y, Dong L, Li M, Zhang H, Zhang P, Zhu K, Li B, Deal KR, Huo N, Liu Z (2020) A rare single nucleotide variant in Pm5e confers powdery mildew resistance in common wheat. New Phytol 228:1011–1026. https://doi.org/10.1111/nph.16762
Xu Y, Li P, Zou C, Lu Y, **e C, Zhang X, Prasanna BM, Olsen MS (2017) Enhancing genetic gain in the era of molecular breeding. J Exp Bot 68:2641–2666. https://doi.org/10.1093/jxb/erx135
Zhan H, Wang Y, Zhang D, Du C, Zhang X, Liu X, Wang G, Zhang S (2021) RNA-seq bulked segregant analysis combined with KASP genoty** rapidly identified PmCH7087 as responsible for powdery mildew resistance in wheat. Plant Genome 14:e20120. https://doi.org/10.1002/tpg2.20120
Acknowledgements
R.K.J. is thankful to Dept. of Science Technology, Govt. of India for infrastructure support through the CURIE program. R.M. is also thankful to the President, Centurion University of Technology and Management for his encouragement and support.
Funding
This research work was carried out using financial assistance from Science and Engineering Research Board (SERB) (Grant no. EMR/2016/005234). JS is thankful for a Junior Research Fellowship from SERB, Govt. of India.
Author information
Authors and Affiliations
Contributions
R.K.J. conceived and designed the research work. J.S. conducted the research experiments including development of RIL population, BSR-sequencing, and SNP discovery. R.M. performed the SNP validation through KASP assay and data analysis. J.S. and R.M. wrote the manuscript. R.K.J. and R.M. critically reviewed the manuscript. All the authors have read, revised and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Message
• BSR-Seq and SNP genoty** identified a 7.3 Mb candidate PB resistance region in chromosome 6 of resistant onion.
• Twenty-three DEGs were reported in the candidate region, including ACCL_20794, a CC-NBS-LRR class R-gene.
• KASP assay revealed co-segregated expression of C6SNP1 marker with ACCL_20794 gene.
• C6SNP1 attributed a single amino acid mutation—histidine (H) to serine (S) in the resistance genotype Arka Kalyan.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sahoo, J., Mishra, R. & Joshi, R.K. Bulked Segregant RNA Sequencing (BSR-Seq) Combined with SNP Genoty** Towards Map** and Characterization of a Purple Blotch Resistance Gene in Onion (Allium cepa L.). Plant Mol Biol Rep (2024). https://doi.org/10.1007/s11105-024-01466-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11105-024-01466-1