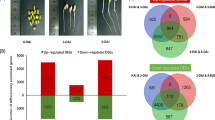
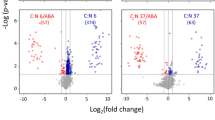
Abstract
Energy production plays an important role in seed germination. Elucidating the molecular mechanisms in the main pathways and the energy supply processes that control the germination of Phyllostachys edulis seeds could provide necessary information for the genomic-level improvement of seed crops. In this study, we performed ultrastructural observations and measured the respiration rate, total starch content, crude oil content, and enzymes related to respiratory activities. Transcriptome sequences at five different stages of germination were obtained and analyzed. First, carbohydrate degradation, including lipid and starch metabolism, plays an important role in providing energy during P. edulis seed germination. The total starch and crude oil contents decreased, and starch metabolism and lipid body degradation pathway genes were consistently upregulated during P. edulis seed germination. Second, the glycolytic pathway and citrate cycle in the respiration pathway supply energy during P. edulis seed germination. The respiration intensity increased significantly, with the 6-phosphofructokinase (FPK) enzyme peaking in stage II. Promitochondria broke down into smaller mitochondria, and respiration intensity increased in stages III to IV, with glucose-6-phosphate dehydrogenase and NAD-malate dehydrogenase enzymes reaching peak values in these stages. The AP2 and bHLH proteins were the most commonly upregulated transcription factors related to seed germination and may play significant roles in seed germination.










Similar content being viewed by others
Data Availability
The sequencing data of this study are available in the Sequence Read Archive (SRA) at the National Center for Biotechnology Information (NCBI) (accession number: PRJNA627339).
Abbreviations
- TCA:
-
Tricarboxylic acid
- TFs:
-
Transcription factors
- DEGs:
-
Differentially expressed genes
- GA:
-
Gibberellins
- TKW:
-
Thousand seed weight
- TEM:
-
Transmission electron microscopy
- G-6PDH:
-
6-Phosphate glucose dehydrogenase
- NAD-MDH:
-
NAD-malate dehydrogenase
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NADH:
-
Nicotinamide adenine dinucleotide
- SRA:
-
Sequence read archive
- GO :
-
Gene Ontology
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- qRT-PCR:
-
Quantitative real-time PCR
- PCC:
-
Principle component classifier
- FDR:
-
False discovery rate
- HK:
-
Hexokinase
- PK:
-
Pyruvate kinase
- GPI:
-
Glucose phosphate isomerase
- FPK:
-
6-Phosphofructokinase
- MDH:
-
Malate dehydrogenase
References
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11:R106. https://doi.org/10.1186/gb-2010-11-10-r106
Bao YZ, Yao ZQ, Cao XL, Peng JF, Xu Y, Chen MX, Zhao SF (2017) Transcriptome analysis of Phelipanche aegyptiaca seed germination mechanisms stimulated by fluridone, TIS108, and GR24. PLoS One 12:e0187539. https://doi.org/10.1371/journal.pone.0187539
Barrôco RM, Kris Van Poucke, Bergervoet JHW, De Veylder L, Groot SPC, Dirk Inzé, Engler G (2005) The role of the cell cycle machinery in resumption of postembryonic development. Plant Physiol 137:127–140. https://doi.org/10.1104/pp.104.049361
Bewley JD (1997) Seed germination and dormancy. Plant Cell 9:1055–1066. https://doi.org/10.1105/tpc.9.7.1055
Botha FC, Small JGC, Grobbelaar N (1984) The effect of water stress on the respiration and some aspects of respiratory metabolism of Citrullus lanatus seeds. S Afr J Bot 2:247
Botha FC, Potgieter GP, Botha AM (1992) Respiratory metabolism and gene expression during seed germination. Plant Growth Regul 11:211–224. https://doi.org/10.1007/BF00024560
Dai MQ, Hu YF, Zhao Y, Liu HF, Zhou DX (2007) A WUSCHEL-LIKE HOMEOBOX gene represses a YABBY gene expression required for rice leaf development. Plant Physiol 144:380–390. https://doi.org/10.1104/pp.107.095737
Dai XY, Wang YY, Yang A, Zhang WH (2012) OsMYB2P-1, an R2R3 MYB transcription factor, is involved in the regulation of phosphate-starvation responses and root architecture in rice. Plant Physiol 159:169–183. https://doi.org/10.1104/pp.112.194217
Druka A, Muehlbauer G, Druka I, Caldo R, Baumann U, Rostoks N, Schreiber A, Wise R, Close T, Kleinhofs A, Graner A, Schulman A, Langridge P, Sato K, Hayes P, McNicol J, Marshall D, Waugh R (2006) An atlas of gene expression from seed to seed through barley development. Funct Integr Genomics 6:202–211. https://doi.org/10.1007/s10142-006-0025-4
Ehrenshaft M, Brambl R (1990) Respiration and mitochondrial biogenesis in germinating embryos of maize. Plant Physiol 93:295–304. https://doi.org/10.1104/pp.93.1.295
Evers T, Millar S (2002) Cereal grain structure and development: some implications for quality. J Cereal Sci 36:261–284. https://doi.org/10.1006/jcrs.2002.0435
Fan T, Li X, Yang W, **a K, Ouyang J, Zhang M (2015) Rice osa-miR171c mediates phase change from vegetative to reproductive development and shoot apical meristem maintenance by repressing four OsHAM transcription factors. PloS One 10:e0125833. https://doi.org/10.1371/journal.pone.0125833
Ge W, Hu T, Gao J, Li L, Cheng ZC, Peng ZH (2016) Difference of nutritional components between Phyllostachys edulis seeds and main grain crops. Turk J Biochem 41:331–337. https://doi.org/10.1515/tjb-2016-0048
Gimeno-Gilles C, Lelièvre E, Viau L, Malik-Ghulam M, Ricoult C, Niebel A, Limami AM (2009) ABA-mediated inhibition of germination is related to the inhibition of genes encoding cell-wall biosynthetic and architecture: modifying enzymes and structural proteins in Medicago truncatula embryo axis. Mol Plant 2:108–119. https://doi.org/10.1093/mp/ssn092
Gutierrez L, Van Wuytswinkel O, Castelain M, Bellini C (2007) Combined networks regulating seed maturation. Trends Plant Sci 12:294–300. https://doi.org/10.1016/j.tplants.2007.06.003
Han ZP, Wang B, Tian L, Wang SX, Zhang J, Guo SL, Zhang HC, Xu LR, Chen YH (2020) Comprehensive dynamic transcriptome analysis at two seed germination stages in maize (Zea mays L.). Physiol Plantarum 168:205–217. https://doi.org/10.1111/ppl.12944
Hirano K, Yoshida H, Aya K, Kawamura M, Hayashi M, Hobo T, Sato-Izawa K, Kitano H, Ueguchi-Tanaka M, Matsuoka M (2017) Small organ size 1 and SMALL small organ size 2/dwarf and low-tillering form a complex to integrate auxin and brassinosteroid signaling in rice. Mol Plant 10:590–604. https://doi.org/10.1016/j.molp.2016.12.013
Howell KA, Narsai R, Carroll A, Ivanova A, Lohse M, Usadel B, Millar AH, James W (2009) Map** metabolic and transcript temporal switches during germination in rice highlights specific transcription factors and the role of RNA instability in the germination process. Plant Physiol 149:961–980. https://doi.org/10.1104/pp.108.129874
Inukai Y, Sakamoto T, Ueguchi-Tanaka M, Shibata Y, Gomi K, Umemura I, Hasegawa Y, Ashikari M, Kitano H, Matsuoka M (2005) Crown rootless1, which is essential for crown root formation in rice, is a target of an AUXIN RESPONSE FACTOR in auxin signaling. Plant Cell 17:1387–1396. https://doi.org/10.1105/tpc.105.030981
Jiang ZH (2002) The flowering and fruiting of bamboo and bamboo seedling breeding. In: Liu H, Luan SL (eds) Bamboo and rattan in the world, 1st edn. Liaoning Science and Technology publishing house, Shenyang, pp 124–125
Jiang FL, Bo LP, Xu JJ, Wu Z (2018) Changes in respiration and structure of non-heading Chinese cabbage seeds during gradual artificial aging. Sci Hortic-AMSTERDAM 238:14–22. https://doi.org/10.1016/j.scienta.2018.04.011
Jiang LY, Ma X, Zhao SS, Tang YY, Liu FX, Gu P, Fu YC, Zhu ZF, Cai HW, Sun CQ (2019) The APETALA2-like transcription factor SUPERNUMERARY BRACT controls rice seed shattering and seed size. Plant Cell 31:17–36. https://doi.org/10.1105/tpc.18.00304
Khan AA (1971) Cytokinins: permissive role in seed germination. Science 171:853–859. https://doi.org/10.1126/science.171.3974.853
Kitomi Y, Hiroko I, Tokunori H, Koichiro A, Hidemi K, Yoshiaki I (2011) The auxin responsive AP2/ERF transcription factor CROWN ROOTLESS5 is involved in crown root initiation in rice through the induction of OsRR1, a type-A response regulator of cytokinin signaling. Plant J 67:472–484. https://doi.org/10.1111/j.1365-313X.2011.04610.x
Laudencia-Chingcuanco DL, Stamova BS, You FM, Lazo GR, Beckles DM, Anderson OD (2007) Transcriptional profiling of wheat caryopsis development using cDNA microarrays. Plant Mol Biol 63:651–668. https://doi.org/10.1007/s11103-006-9114-y
Li HS (2000) Principles and techniques of plant physiological and biochemical experiments. Higher education press, Bei**g
Li J, Zhang BS, Gao HY, Mu SH, Gao J (2019) Anatomical structural changes and correlation between water content and germination characteristics during seed germination of Phyllostachys edulis (CARRIÈRE) J.HOUZ. Propag Ornam Plants 5:3–8
Li XY, **e LH, Zheng HF, Cai MM, Cheng ZC, Bai YC, Li J, Gao J (2019) Transcriptome profiling of postharvest shoots identifies PheNAP2-and PheNAP3-promoted shoot senescence. Tree Physiol 39:2027–2044. https://doi.org/10.1093/treephys/tpz100
Liu XH, Fu JJ, Gu D, Liu WX, Liu TS, Peng YL, Wang JH, Wang GY (2008) Genome-wide analysis of gene expression profiles during the kernel development of maize (Zea mays L.). Genomics 91:378–387. https://doi.org/10.1016/j.ygeno.2007.12.002
Liu W, Kohlen W, Lillo A, OP den Camp R, Sergey I, Hartog M, Limpens E, Jamil M, Smaczniak C, Kaufmann K (2011) Strigolactone biosynthesis in Medicago truncatula and rice requires the symbiotic GRAS-type transcription factors NSP1 and NSP2. Plant Cell 23:3853–3865. https://doi.org/10.1105/tpc.111.089771
Locascio A, Roig-Villanova I, Bernardi J, Varotto S (2014) Current perspectives on the hormonal control of seed development in Arabidopsis and maize: a focus on auxin. Front Plant Sci 5:412. https://doi.org/10.3389/fpls.2014.00412
Logan DC, Leaver CJ (2000) Mitochondria-targeted GFP high lights the heterogeneity of mitochondrial shape, size and movement within living plant cells. J Exp Bot 51:865–871
Love MI, Wolfgang H, Simon A (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Lu TC, Meng LB, Yang CP, Liu GF, Liu GJ, Ma W, Wang BC (2008) A shotgun phosphoproteomics analysis of embryos in germinated maize seeds. Planta 228:1029–1041. https://doi.org/10.1007/s00425-008-0805-2
Ma XS, Feng FJ, Zhang Y, Eid Elesawi I, Xu K, Li TF, Mei HW, Liu HY, Gao NN, Chen CL (2019) A novel rice grain size gene OsSNB was identified by genome-wide association study in natural population. PLoS Genet 15(5):e1008191. https://doi.org/10.1371/journal.pgen.1008191
Makita A (1998) The significance of the mode of clonal growth in the life history of bamboos. Plant Spec Biol 13:85–92. https://doi.org/10.1111/j.1442-1984.1998.tb00251.x
Mao C, He J, Liu L, Deng Q, Yao X, Liu C, Qiao Y, Li P, Ming F (2020) OsNAC2 integrates auxin and cytokinin pathways to modulate rice root development. Plant Biotechnol J 18:429–442. https://doi.org/10.1111/pbi.13209
Masubelele NH, Walter D, Margit M, Spencer M, Carl C, Rachael H, Jeroen N, Simon S, James AH (2005) Murray D-type cyclins activate division in the root apex to promote seed germination in Arabidopsis. Proc Natl Acad Sci USA 102:15694–15699. https://doi.org/10.1073/pnas.0507581102
Mihaela Pertea, Pertea GM, Antonescu CM, Tsung-Cheng C, Mendell JT, Salzberg SL (2015) String tie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33:290–295. https://doi.org/10.1038/nbt.3122
Møller IM (2001) Plant mitochondria and oxidative stress: electron transport, NADPH turnover, and metabolism of reactive oxygen species. Annu Rev Plant Physiol 52:561–591. https://doi.org/10.1134/S1028334X0901036X
Navrot N, Rouhier N, Gelhaye E, Jacquot JP (2007) Reactive oxygen species generation and antioxidant systems in plant mitochondria. Physiol Plant 129:185–195. https://doi.org/10.1111/j.1399-3054.2006.00777.x
Penfield S, Josse EM, Kannangara R, Gilday AD, Halliday KJ, Graham IA (2005) Cold and light control seed germination through the bHLH transcription factor SPATULA. Curr Biol 15:1998–2006. https://doi.org/10.1016/j.cub.2005.11.010
Peng ZH, Lu Y, Li LB, Zhao Q, Jiang ZH (2013) The draft genome of the fast-growing non-timber forest species moso bamboo (Phyllostachys heterocycla). Nat Genet 45:456–461. https://doi.org/10.1038/ng.2569
Pergo EM, Ishii-Iwamoto EL (2011) Changes in energy metabolism and antioxidant defense systems during seed germination of the weed species Ipomoea triloba L. and the responses to allelochemicals. J Chem Ecol 37:500–513. https://doi.org/10.1007/s10886-011-9945-0
Qin H, Wang J, Chen X, Wang F, Peng P, Zhou Y, Miao Y, Zhang Y, Gao Y, Qi Y (2019) Rice OsDOF15 contributes to ethylene-inhibited primary root elongation under salt stress. New Phytol 223:798–813. https://doi.org/10.1111/nph.15824
Rodríguez JL, De Diego JG, Rodríguez FD, Cervantes E (2015) Mitochondrial structures during seed germination and early seedling development in Arabidopsis thaliana. Biologia 70:1019–1025. https://doi.org/10.1515/biolog-2015-0130
Roxrud I, Lid SE, Fletcher JC, Schmidt ED, Opsahl-Sorteberg HG (2007) GASA4, one of the 14-member Arabidopsis GASA family of small polypeptides, regulates flowering and seed development. Plant Cell Physiol 48:471–483. https://doi.org/10.1093/pcp/pcm016
Sano N, Permana H, Kumada R, Shinozaki Y, Tanabata T, Ya-mada T, Hirasawa T, Kanekatsu M (2012) Proteomic analysis of embryonic proteins synthesized from long-lived m RNAs during germination of rice seeds. Plant Cell Physiol 53:687–698. https://doi.org/10.1093/pcp/pcs024
Sun L, Guo Q, Wang Q, Feng Y, Mu S (2012) Flower organs morph ology and structure of Phyllostachys edulis. Sci Silvae Sin 11:124–129. https://doi.org/10.1007/s11783-011-0280-z
Wang LJ, Pei ZY, Tian YC, He CZ (2005) OsLSD1, a rice zinc finger protein, regulates programmed cell death and callus differentiation. Mol Plant-Microbe Inreact 18(5):375–384. https://doi.org/10.1094/MPMI-18-0375
Wang C, Wang H, Zhang J, Chen SY (2008) A seed-specific AP2-domian transcription factor from soybean plays a certain role in regulation of seed germination. Chinese Sci Life Sci 51(4):336–345
Wu JH, Zhu CF, Pang JH, Zhang XG, Yang CL, **a GX, Tian YC, He CZ (2014) OsLOL1, aC2C2-type zinc finger protein, interacts with OsbZIP 58 to promote seed germination through the modulation of gibberellin biosynthesis in Oryza sativa. Plant J 80:1118–1130
Xu QT, Yang L, Zhou ZQ, Mei FZ, Qu LH, Zhou GS (2013) Process of aerenchyma formation and reactive oxygen species induced by water logging in wheat seminal roots. Planta 238:969–982. https://doi.org/10.1007/s00425-013-1947-4
Xue LJ, Zhang JJ, Xue HW (2012) Genome-wide analysis of the complex transcriptional networks of rice develo** seeds. PloS One 7:e31081. https://doi.org/10.1371/journal.pone.0031081
Yang PF, Li XJ, Wang XQ, Chen H, Chen F, Shen SH (2007) Proteomic analysis of rice (Oryza sativa) seeds during germination. Proteomics 7:3358–3368. https://doi.org/10.1002/pmic.200700207
Yang CH, Li DY, Liu X, Ji CJ, Hao LL, Zhao XF, Li XB, Chen CY, Cheng ZK, Zhu LH (2014) OsMYB103L, an R2R3-MYB transcription factor, influences leaf rolling and mechanical strength in rice (Oryza sativa L.). BMC Plant Biol 14:158. https://doi.org/10.1186/1471-2229-14-158
Yang WF, Zhang JL, Lü WZ, Cao GC, Chen JY (2014b) Study on the differential genes expression in maize embryo treated by a controlled deterioration treatment. Sci Agric Sin 47:1878–1893
Yao Z, Tian F, Cao X, Xu Y, Chen M, **ang B, Zhao S (2016) Global transcriptomic analysis reveals the mechanism of Phelipanche aegyptiaca seed germination. Int J Mol Sci 17:1139. https://doi.org/10.3390/ijms17071139
Yu YL, Guo GF, Lv DW, Hu YK, Li JR, Li XH, Yan YM (2014) Transcriptome analysis during seed germination of elite Chinese bread wheat cultivar Jimai 20. BMC Plant Biol 14:20. https://doi.org/10.1186/1471-2229-14-20
Zaynab M, Pan D, Fatima M, Chen S, Chen W (2018) Transcriptomic approach to address low germination rate in Cyclobalnopsis gilva seeds. S Afr J Bot 119:286–294. https://doi.org/10.1016/j.sajb.2018.09.024
Zhou FC (1998) Bamboo cultivation. Nan**g Forestry University Printing Company, Nan**g
Zhu N, Cheng SF, Liu XY, Hao D, Dai MQ, Zhou DX, Yang WJ, Zhao Y (2015) The R2R3-type MYB gene OsMYB91 has a function in coordinating plant growth and salt stress tolerance in rice. Plant Sci 236:146–156. https://doi.org/10.1016/j.plantsci.2015.03.023
Funding
This work was supported by the Basic Scientific Research Fund Program of ICBR (Grant No. 1632019024), the National Natural Science Foundation of China (32001379), and the Basic Scientific Research Fund Program of ICBR (Grant No. 1632021016).
Author information
Authors and Affiliations
Contributions
JL and JG conceived and designed the study. JL, YX, YB, HZ, and SM conducted experiments. JL, YB, XL, and LL analyzed the data. JL and XL wrote the manuscript; all authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Message
• Carbohydrate degradation including lipid and starch metabolism plays an important role in providing energy.
• The glycolytic pathway and citrate cycle in the respiration pathway supplied energy.
• AP2 and bHLH proteins are the most commonly upregulated transcription factors related to seed germination and may play significant roles.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Li, J., Li, X., Bai, Y. et al. Transcriptome Analysis of Energy Supply Process During Seed Germination in Phyllostachys edulis. Plant Mol Biol Rep 41, 489–511 (2023). https://doi.org/10.1007/s11105-023-01378-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-023-01378-6