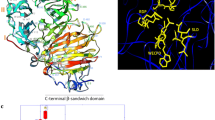
Abstract
Plant acid invertases are considered to be the key enzymes in sucrose unloading and carbohydrate supply to sink tissues. Acid cell-wall invertases control sucrose transport via the apoplastic pathway during sink initiation and expansion. In this study, we identified 12 LIN7 gene homologs encoding cell-wall invertases in red- and green-fruited tomato accessions (Solanum section Lycopersicon) of self-compatible and self-incompatible species. All genes consisted of six exons and five introns, including highly conserved 9-bp exon II. Identification of 226 exonic single nucleotide polymorphisms as well as extremely high intron variability indicates a significant interspecific divergence among the examined tomato accessions. Computational prediction revealed protein structure typical for the glycosyl hydrolase family 32 and conserved catalytic sites described for other plant cell-wall invertases. LIN7 expression in mature buds and flowers confirms LIN7 role in the development of pollen tubes and grains. The variability in gene and protein sequences and species-specific differences in LIN7 expression patterns may be responsible for putative functional divergence of invertases. Furthermore, we performed phylogenetic analysis of the Solanum section Lycopersicon species based on the LIN7 gene, which clearly divided the analyzed tomato accessions into two main clusters corresponding to self-compatible and self-incompatible species and was in agreement with the separation into red- and green-fruited plants. Given that LIN7 plays an essential role in tomato fertility and fruit ripening, the characterization of protein variability within species of section Lycopersicon may be useful to evaluate the potential application of the encoding genes for tomato breeding programs.






Similar content being viewed by others
Abbreviations
- CWIN:
-
Cell-wall invertase
- GH32:
-
Glycoside hydrolase family 32
- MG:
-
Mature green
- pI:
-
Isoelectric point
- qRT-PCR:
-
Quantitative RT-PCR
- RF:
-
Ripe fruit
- SC:
-
Self-compatible
- SI:
-
Self-incompatible
- SNP:
-
Single nucleotide polymorphism
- VIN:
-
Vacuolar invertase
References
Beckles DM (2012) Factors affecting the postharvest sugar content of tomato Solanum lycopersicum L. fruit. Postharvest Biol Technol 63(1):129–140
Beckles DM, Hong N, Stamova L, Luengwilai K (2012) Biochemical factors contributing to tomato fruit sugar content: a review. Fruits 67:49–64
Chen TH, Huang YC, Yang CS, Yang CC, Wang AY, Sung HY (2009) Insights into the catalytic properties of bamboo vacuolar invertase through mutational analysis of active site residues. Phytochemistry 70(1):25–31
Chen KY, Tanksley SD (2004) High-resolution map** and functional analysis of se2.1: a major stigma exertion quantitative trait locus associated with the evolution from allogamy to autogamy in the genus Lycopersicon. Genetics 168(3):1563–1573
Choi Y, Sims GE, Murphy S, Miller JR, Chan AP (2012) Predicting the functional effect of amino acid substitutions and indels. PLoS One 7(10):e46688. https://doi.org/10.1371/journal.pone.0046688
Clement C, Burrus M, Audran J-C (1996) Floral organ growth and carbohydrate content during pollen development in Lilium. Am J Bot 83:459–469
Davies JN (1966) Occurrence of sucrose in the fruit species of Lycopersicon. Nature 209(5023):640–641
Davies JN, Hobson GE (1981) The constituents of tomato fruit—the influence of environment, nutrition, and genotype. Crit Rev Food Sci Nutr 15(3):205–280
Dorion S, Lalonde S, Saini HS (1996) Induction of male sterility in wheat by meiotic-stage water deficit is preceded by a decline in invertase activity and changes in carbohydrate metabolism in anthers. Plant Physiol 111(1):137–145
Elliott KJ, Butler WO, Dickinson CD, Konno Y, Vedvick TS, Fitzmaurice L, Mirkov TE (1993) Isolation and characterization of fruit vacuolar invertase genes from two tomato species and temporal differences in mRNA levels during fruit ripening. Plant Mol Biol 21(3):515–524
Engelke T, Hirsche J, Roitsch T (2010) Anther-specific carbohydrate supply and restoration of metabolically engineered male sterility. J Exp Bot 61(10):2693–2706
Expósito-Rodríguez M, Borges A, Borges-Pérez A, Pérez A (2008) Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. BMC Plant Biol 8:131. https://doi.org/10.1186/1471-2229-8-131
Franklin-Tong VE, Franklin FCH (2003) The different mechanisms of gametophytic self-incompatibility. Phil Trans R Soc Lond B 358:1025–1032
Fridman E, Zamir D (2003) Functional divergence of a syntenic invertase gene family in tomato, potato, and Arabidopsis. Plant Physiol 131(2):603–609
Fotopoulos V (2005) Plant invertases: structure, function and regulation of a diverse enzyme family. J Biol Res 4:127–137
Gerszberg A, Hnatuszko-Konka KT, Kononowicz AK (2015) Tomato (Solanum lycopersicum L.) in the service of biotechnology. Plant Cell Tissue Organ Cult 120:881–902
Godt DE, Roitsch T (1997) Regulation and tissue-specific distribution of mRNAs for three extracellular invertase isoenzymes of tomato suggests an important function in establishing and maintaining sink metabolism. Plant Physiol 115(1):273–282
Goetz M, Godt DE, Guivarc'h A, Kahmann U, Chriqui D, Roitsch T (2001) Induction of male sterility in plants by metabolic engineering of the carbohydrate supply. Proc Natl Acad Sci U S A 98(11):6522–6527
Goetz M, Guivarch A, Hirsche J, Bauerfeind MA, González MC, Hyun TK, Eom SH, Chriqui D, Engelke T, Großkinsky DK, Roitsch T (2017) Metabolic control of tobacco pollination by sugars and invertases. Plant Physiol 173(2):984–997
Goetz M, Roitsch T (1999) The different pH optima and substrate specificities of extracellular and vacuolar invertases from plants are determined by a single amino-acid substitution. Plant J 20(6):707–711
Gramzow L, Theißen G (2015) Phylogenomics reveals surprising sets of essential and dispensable clades of MIKC(c)-group MADS-box genes in flowering plants. J Exp Zool B Mol Dev Evol 324(4):353–362
Hirsche J, Engelke T, Voller D, Gotz M, Roitsch T (2009) Interspecies compatibility of the anther specific cell wall invertase promoters from Arabidopsis and tobacco for generating male sterile plants. Theor Appl Genet 118(2):235–245
Igic B, Lande R, Kohn JR (2008) Loss of self incompatibility and its evolutionary consequences. Int J Plant Sci 169:93–104
Ji X, Van den Ende W, Van Laere A, Cheng S, Bennett J (2005) Structure, evolution, and expression of the two invertase gene families of rice. J Mol Evol 60(5):615–634
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJ (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10(6):845–858
Koch K (2004) Sucrose metabolism: regulatory mechanisms and pivotal roles in sugar sensing and plant development. Curr Opin Plant Biol 7(3):235–246
Laskowski RA, Watson JD, Thornton JM (2005) ProFunc: a server for predicting protein function from 3D structure. Nucleic Acids Res 33(Web Server):W89–W93. https://doi.org/10.1093/nar/gki414
Luckwill LC (1943) The genus Lycopersicon: an historical, biological, and taxonomical survey of the wild and cultivated tomatoes. Aberdeen Univ Stud 120:1–44
Maiti S, Kundu S, Chakraborty D, Paul S, Sengupta S, Das K, Pal A (2011) Developmentally regulated temporal expression and differential acid invertase activity in differentiating cotyledonary explants of mungbean [Vigna radiata (L.) Wilczek]. Plant Cell Tissue Organ Cult 107:417–425
Marshall JA, Knapp S, Davey MR, Power JB, Cocking EC, Bennett MD, Cox AV (2001) Molecular systematics of solanum section Lycopersicum (Lycopersicon) using the nuclear ITS rDNA region. Theor Appl Genet 103:1216–1222
Miller JS, Kostyun JL (2011) Functional gametophytic self-incompatibility in a peripheral population of Solanum peruvianum (Solanaceae). Heredity (Edinb) 107:30–39
Miron D, Petreikov M, Carmi N, Shen S, Levin I, Granot D, Zamski E, Schaffer AA (2002) Sucrose uptake, invertase localization and gene expression in develo** fruit of Lycopersicon esculentum and the sucrose-accumulating Lycopersicon hirsutum. Physiol Plant 115(1):35–47
Miron D, Schaffer AA (1991) Sucrose phosphate synthase, sucrose synthase, and Invertase activities in develo** fruit of Lycopersicon esculentum mill. and the sucrose accumulating Lycopersicon hirsutum Humb. and Bonpl. Plant Physiol 95(2):623–627
Müller CH (1940) A revision of the genus Lycopersicon. U S D A Misc Publ 382:1–28+10pl
Obata-Sasamoto H, Thorpe TA (1983) Cell wall invertase activity in cultured tobacco tissues. Plant Cell Tissue Organ Cult 2:3–9
Pease JB, Haak DC, Hahn MW, Moyle LC (2016) Phylogenomics reveals three sources of adaptive variation during a rapid radiation. PLoS Biol 14(2):e1002379
Peralta IE, Spooner DM (2001) Granule-bound starch synthase (GBSSI) gene phylogeny of wild tomatoes (solanum L. section Lycopersicon [Mill.] Wettst. subsection Lycopersicon). Am J Bot 88(10):1888–1902
Peralta IE, Spooner DM, Knapp S (2008) Taxonomy of wild tomatoes and their relatives (Solanum sect. Lycopersicoides, sect. Juglandifolia, sect. Lycopersicon; Solanaceae). Syst Bot Monogr 84:1–186
Pons T, Olmea O, Chinea G, Beldarraín A, Márquez G, Acosta N, Rodríguez L, Valencia A (1998) Structural model for family 32 of glycosyl-hydrolase enzymes. Proteins 33(3):383–395
Proels RK, González MC, Roitsch T (2006) Gibberellin-dependent induction of tomato extracellular invertase Lin7 is required for pollen development. Funct Plant Biol 33(6):547–554
Proels RK, Hause B, Berger S, Roitsch T (2003) Novel mode of hormone induction of tandem tomato invertase genes in floral tissues. Plant Mol Biol 52(1):191–201
Proels RK, Hückelhoven R (2014) Cell-wall invertases, key enzymes in the modulation of plant metabolism during defense responses. Mol Plant Pathol 15(8):858–864
Rao VR, Hodgkin T (2002) Genetic diversity and conservation and utilization of plant genetic resources. Plant Cell Tissue Organ Cult 68:1–19
Rick CM (1960) Hybridization between Lycopersicon esculentum and Solanum pennellii: phylogenetic and cytogenetic significance. Proc Natl Acad Sci U S A 46(1):78–82
Rick CM (1979) Biosystematic studies in Lycopersicon and closely related species of solanum. In: Hawkes JG, Lester RN, Skelding AD (eds) The biology and taxonomy of the Solanaceae. Academic Press, New York London, pp 667–677
Shima Y, Fujisawa M, Kitagawa M, Nakano T, Kimbara J, Nakamura N, Shiina T, Sugiyama J, Nakamura T, Kasumi T, Ito Y (2014) Tomato FRUITFULL homologs regulate fruit ripening via ethylene biosynthesis. Biosci Biotechnol Biochem 78(2):231–237
Slugina MA, Shchennikova AV, Kochieva EZ (2017) TAI vacuolar invertase orthologs: the interspecific variability in tomato plants (solanum section Lycopersicon). Mol Gen Genomics 292(5):1123–1138
Shimon-Kerner N, Mills D, Merchuk JC (2000) Sugar utilization and invertase activity in hairy-root and cell-suspension cultures of Symphytum officinale. Plant Cell Tissue Organ Cult 62:89–94
Steinhauser MC, Steinhauser D, Koehl K, Carrari F, Gibon Y, Fernie AR, Stitt M (2010) Enzyme activity profiles during fruit development in tomato cultivars and Solanum pennellii. Plant Physiol 153(1):80–98
Stone SL, Goring DR (2001) The molecular biology of self-incompatibility systems in flowering plants. Plant Cell Tissue Organ Cult 67:93–114
Sturm A (1996) Molecular characterization and functional analysis of sucrose-cleaving enzymes in carrot (Daucus carota L.) J Exp Bot 47:1187–1192
Sturm A (1999) Invertases. Primary structures, functions, and roles in plant development and sucrose partitioning. Plant Physiol 121(1):1–8
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30(12):2725–2729
Tang GQ, Lüscher M, Sturm A (1999) Antisense repression of vacuolar and cell wall invertase in transgenic carrot alters early plant development and sucrose partitioning. Plant Cell 11(2):177–189
Tanksley SD (2004) The genetic, developmental, and molecular bases of fruit size and shape variation in tomato. Plant Cell 16(Suppl):S181–S189
Techen N, Parveen I, Khan IA (2017) A single molecular marker to distinguish between species of Dioscorea. Genome 60(3):201–207
The 100 Tomato Genome Sequencing Consortium, Aflitos S, Schijlen E, de Jong H, de Ridder D, Smit S, Finkers R, Wang J, Zhang G, Li N, Mao L, Bakker F, Dirks R, Breit T, Gravendeel B, Huits H et al (2014) Exploring genetic variation in the tomato (solanum section Lycopersicon) clade by whole-genome sequencing. Plant J 80(1):136–148
Tomato Genome Consortium (2012) The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485(7400):635–641
Tymowska-Lalanne Z, Kreis M (1998) The plant invertases: physiology, biochemistry and molecular biology. In: Callow JA (ed) Advances in Botanical Research, vol 28, 1st edn. Academic Press, New York, pp 71–117
Unger C, Hardegger M, Lienhard S, Sturm A (1994) cDNA cloning of carrot (Daucus carota) soluble acid beta-fructofuranosidases and comparison with the cell wall isoenzyme. Plant Physiol 104(4):1351–1357
Verhaest M, Lammens W, Le Roy K, De Coninck B, De Ranter CJ, Van Laere A, Van den Ende W, Rabijns A (2006) X-ray diffraction structure of a cell-wall invertase from Arabidopsis thaliana. Acta Crystallogr D Biol Crystallogr 62(12):1555–1563
Wang Y, Chen J, Feng J, Qin Q, Huang J (2015) Overexpression of a loquat (Eriobotrya japonica Lindl.) vacuolar invertase affects sucrose levels and growth. Plant Cell Tissue Organ Cult 123:99–108
Yao Y, Wu XH, Geng MT, Li RM, Liu J, Hu XW, Guo JC (2014) Cloning, 3D modeling and expression analysis of three vacuolar invertase genes from cassava (Manihot Esculenta Crantz). Molecules 19(5):6228–6245
Zhang W, Kan SL, Zhao H, Li ZY, Wang XQ (2014) Molecular phylogeny of tribe Theeae (Theaceae s.s.) and its implications for generic delimitation. PLoS One 9(5):e98133
Zhang YL, Zhang AH, Jiang J (2013) Gene expression patterns of invertase gene families and modulation of the inhibitor gene in tomato sucrose metabolism. Genet Mol Res 12(3):3412–3420
Zhao P, Pan Q, Yu W, Zhao L (2006) Dissect style response to pollination using metabolite profiling in self-compatible and self-incompatible tomato species. J Chromatogr B Analyt Technol Biomed Life Sci 1017-1018:153–162
Zuriaga E, Blanca J, Nuez F (2009) Classification and phylogenetic relationships in solanum section Lycopersicon based on AFLP and two nuclear gene sequences. Genet Resour Crop Evol 56:663–678
Acknowledgments
This work was financially supported by grants from the Federal Agency for Scientific Organizations (FASO Russia: no. 0104-2014-0210; CPSR “Potato breeding and seed production”) and was performed using the experimental climate control facility located in the Institute of Bioengineering, Research Center of Biotechnology of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Contributions
EZK conceived and designed the research. MAS conducted the experiments and performed gene cloning, phylogenetic reconstructions, variability analyses, and expression pattern determination. AVS contributed to in silico analyses. EZK, MAS, and AVS wrote the manuscript. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Key Message
Genes of LIN7 cell-wall invertase were identified in 12 Solanum section Lycopersicon accessions. Comparison of the LIN7 structure, polymorphism, phylogeny, and expression suggests conserved LIN7 role in pollen development.
Electronic Supplementary Material
ESM 1
Supplementary Figs. S1-S3 ‘Alignment of LIN7 sequences from analyzed wild and cultivated tomato accessions compared to S. lycopersicum cv. Heinz sequences: S1 – LIN7 genomic sequences alignment; S2 – LIN7 cDNA sequences alignment; S3 – LIN7 amino acid sequences alignment’. Supplementary Tables S1, S2 ‘List of primers used for LIN7 genes amplification, sequencing, and expression analysis (S1); The significance (p-value) of qRT-PCR results within tissue between pairs of tomato species (S2)’ (XLS) (XLS 1322 kb)
ESM 2
Supplementary Figs. S4–S8 ‘Evolutionary relationships of Solanum section Lycopersicon taxa: S4 The evolutionary history of Solanum section Lycopersicon taxa based on LIN7 homologous gene sequences was inferred using the Maximum Likelihood method based on the Jukes-Cantor model; S5 The evolutionary history of Solanum section Lycopersicon taxa based on LIN7 homologous gene sequences was inferred using the Minimum Evolution method. S6 The evolutionary history of Solanum section Lycopersicon taxa based on LIN7 orthologues sequences was inferred using the Neighbor-Joining method. S7 The evolutionary history of Solanum section Lycopersicon taxa based on LIN7 orthologues sequences was inferred using the Maximum Likelihood method based on the Jukes-Cantor model. S8 The evolutionary history of Solanum section Lycopersicon taxa based on LIN7 orthologues sequences was inferred using the Minimum Evolution method’ (PDF) (PDF 548 kb)
Rights and permissions
About this article
Cite this article
Slugina, M.A., Shchennikova, A.V. & Kochieva, E.Z. LIN7 Cell-Wall Invertase Orthologs in Cultivated and Wild Tomatoes (Solanum Section Lycopersicon). Plant Mol Biol Rep 36, 195–209 (2018). https://doi.org/10.1007/s11105-018-1071-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-018-1071-5