Abstract
Key message
A methyl jasmonate responsive 3-carene synthase (Li3CARS) gene was isolated from Lavandula x intermedia and functionally characterized in vitro.
Abstract
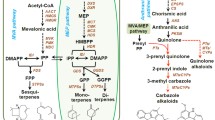
Lavenders produce essential oils consisting mainly of monoterpenes, including the potent antimicrobial and insecticidal monoterpene 3-carene. In this study we isolated and functionally characterized a leaf-specific, methyl jasmonate (MeJA)-responsive monoterpene synthase (Li3CARS) from Lavandula x intermedia. The ORF excluding transit peptides encoded a 64.9 kDa protein that was expressed in E. coli, and purified with Ni–NTA agarose affinity chromatography. The recombinant Li3CARS converted GPP into 3-carene as the major product, with K m and k cat of 3.69 ± 1.17 µM and 2.01 s−1 respectively. Li3CARS also accepted NPP as a substrate to produce multiple products including a small amount of 3-carene. The catalytic efficiency of Li3CARS to produce 3-carene was over ten fold higher for GPP (k cat /K m = 0.56 µM−1s−1) than NPP (k cat /K m = 0.044 µM−1s−1). Production of distinct end product profiles from different substrates (GPP versus NPP) by Li3CARS indicates that monoterpene metabolism may be controlled in part through substrate availability. Li3CARS transcripts were found to be highly abundant in leaves (16-fold) as compared to flower tissues. The transcriptional activity of Li3CARS correlated with 3-carene production, and was up-regulated (1.18- to 3.8-fold) with MeJA 8–72 h post-treatment. The results suggest that Li3CARS may have a defensive role in Lavandula.









Similar content being viewed by others
Abbreviations
- BPPS:
-
Bornyl pyrophosphate synthase
- EO:
-
Essential oil
- GPP:
-
Geranyl pyrophosphate
- MeJA:
-
Methyl jasmonate
- mTPS:
-
Monoterpene synthase
- Li3CARS:
-
L. x intermedia 3-carene synthase
- NPP:
-
Neryl pyrophosphate
- qPCR:
-
Quantitative real-time PCR
- TPS:
-
Terpene synthase
References
Bohlmann J, Meyer-Gauen G, Croteau R (1998) Plant terpenoid synthases: molecular biology and phylogenetic analysis. Proc Natl Acad Sci U S A 95:4126–4133. doi:10.1073/pnas.95.8.4126
Boone CK, Aukema BH, Bohlmann J, Carroll AL, Raffa KF (2011) Efficacy of tree defense physiology varies with bark beetle population density: a basis for positive feedback in eruptive species. Can J For Res 41:1174–1188. doi:10.1139/x11-041
Bordoli L, Kiefer F, Arnold K, Benkert P, Battey J, Schwede T (2009) Protein structure homology modeling using SWISS-MODEL workspace. Nat Protoc 4:1–13. doi:10.1038/nprot.2008.197
Burke CC, Wildung MR, Croteau R (1999) Geranyl diphosphate synthase: cloning, expression, and characterization of this prenyltransferase as a heterodimer. Proc Natl Acad Sci U S A 96:13062–13067. doi:10.1073/pnas.96.23.13062
Byun-McKay A, Godard K-A, Toudefallah M, Martin DM, Alfaro R, King J, Bohlmann J, Plant AL (2012) Wound-induced terpene synthase gene expression in Sitka spruce that exhibit resistance or susceptibility to attack by the white pine weevil. New Phytol 3:3043–3052. doi:10.1104/pp.105.071803
Chen F, Tholl D, Bohlmann J, Pichersky E (2011) The family of terpene synthases in plants: a mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J 66:212–229. doi:10.1111/j.1365-313X.2011.04520.x
De Rapper S, Kamatou G, Viljoen A, Van Vuuren S (2013) The in vitro antimicrobial activity of Lavandula angustifolia essential oil in combination with other aroma-therapeutic oils. Evid Based Complement Altern Med. doi:10.1155/2013/852049
De Vega C, Herrera CM, Dotterl S (2014) Floral volatiles play a key role in specialized ant pollination. Perspect Plant Ecol Evol Syst 16:32–42. doi:10.1016/j.ppees.2013.11.002
Degenhardt J, Köllner TG, Gershenzon J (2009) Monoterpene and sesquiterpene synthases and the origin of terpene skeletal diversity in plants. Phytochemistry 70:1621–1637. doi:10.1016/j.phytochem.2009.07.030
Demissie ZA, Sarker LS, Mahmoud SS (2011) Cloning and functional characterization of β-phellandrene synthase from Lavandula angustifolia. Planta 233:685–696. doi:10.1007/s00425-010-1332-5
Demissie ZA, Cella MA, Sarker LS, Thompson TJ, Rheault MR, Mahmoud SS (2012) Cloning, functional characterization and genomic organization of 1,8-cineole synthases from Lavandula. Plant Mol Biol 79:393–411. doi:10.1007/s11103-012-9920-3
Demissie ZA, Erland LAE, Rheault MR, Mahmoud SS (2013) The biosynthetic origin of irregular monoterpenes in Lavandula: isolation and biochemical characterization of a novel cis-prenyl diphosphate synthase gene, lavandulyl diphosphate synthase. J Biol Chem 288:6333–6341. doi:10.1074/jbc.M112.431171
Dudareva N, Klempien A, Muhlemann JK, Kaplan I (2013) Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol 198:16–32. doi:10.1111/nph.12145
Erland LAE (2015) Enhancement of specialized metabolism, regeneration efficiency and biological activity in lavandin (Lavandula x intermedia cv ‘Grosso’). MSc Thesis, University of british Columbia, pp:40–41
Erland LAE, Rheault MR, Mahmoud SS (2015) Insecticidal and oviposition deterrent effects of essential oils and their constituents against the invasive pest Drosophila suzukii (Matsumura) (Diptera: Drosophilidae). Crop Prot 78:20–26. doi:10.1016/j.cropro.2015.08.013
Fäldt J, Martin D, Miller B, Rawat S, Bohlmann J (2003) Traumatic resin defense in Norway spruce (Picea abies): Methyljasmonate-induced terpene synthase gene expression, and cDNA cloningand functional characterization of (+)-3-carene synthase. Plant Mol Biol 51:119–133
Fontana A, Held M, Fantaye CA, Turlings TC, Degenhardt J, Gershenzon J (2011) Attractiveness of constitutive and Herbivore-induced Sesquiterpene blends of Maize to the Parasitic Wasp Cotesia marginiventris (Cresson). J Chem Ecol 37:582–591. doi:10.1007/s10886-011-9967-7
Frost CJ, Appel HM, Carlson JE, De Moraes CM, Mescher MC, Schulz JC (2007) Within-plant signalling via volatiles overcomes vascular constraints on systemic signalling and primes responses against herbivores. Ecol Lett 10:490–498. doi:10.1111/j.1461-0248.2007.01043.x
Fujimoto T, Tomitaka Y, Abe H, Tsuda S, Futai K, Mizukubo T (2011) Expression profile of jasmonic acid-induced genes and the induced resistance against the root-knot nematode (Meloidogyne incognita) in tomato plants (Solanum lycopersicum) after foliar treatment with methyl jasmonate. J Plant Physiol 168:1084–1097. doi:10.1016/j.jplph.2010.12.002
Guex N, Peitsch MC, Schwede T (2009) Automated comparative protein structure modeling with SWISS-MODEL and Swiss-PdbViewer: a historical perspective. Electrophoresis 30:162–173. doi:10.1002/elps.200900140
Hall DE, Robert JA, Keeling CI, Domanski D, Quesada AI, Jancsik S, Kuzyk MA, Hamberger B, Borchers CH, Bohlmann J (2011) An integrated genomic, proteomic and biochemical analysis of (+)-3-carene biosynthesis in Sitka spruce (Picea sitchensis) genotypes that are resistant or susceptible to white pine weevil. Plant J 65:936–948. doi:10.1111/j.1365-313X.2010.04478.x
Hasegawa M, Mitsuhara I, Seo S, Imai T, Koga J, Okada K, Yamane H, Ohashi Y (2010) Phytoalexin accumulation in the interaction between rice and the blast fungus. Mol Plant Microbe Interact 23:1000–1011. doi:10.1094/MPMI-23-8-1000
Heiling S, Schuman MC, Schoettner M, Mukerjee P, Berger B, Schneider B, Jassbi AR, Baldwin IT (2010) Jasmonate and ppHsystemin regulate key Malonylation steps in the biosynthesis of 17-Hydroxygeranyllinalool Diterpene Glycosides, an abundant and effective direct defense against herbivores in Nicotiana attenuata. Plant Cell 22:273–292. doi:10.1105/tpc.109.071449
Hoelscher DJ, Williams DC, Wildung MR, Croteau R (2003) A cDNA clone for 3-carene synthase from Salvia stenophylla. Phytochemistry 62:1081–1086. doi:10.1016/S0031-9422(02)00674-X
Huang M, Abel C, Sohrabi R, Petri J, Haupt I, Cosimano J, Gershenzon J, Tholl D (2010) Variation of herbivore-induced volatile terpenes among Arabidopsis ecotypes depends on allelic differences and subcellular targeting of two terpene synthases, TPS02 and TPS03. Plant Physiol 153:1293–1310. doi:10.1104/pp.110.154864
Hyatt DC, Youn B, Zhao Y, Santhamma B, Coates RM, Croteau RB, Kang C (2007) Structure of limonene synthase, a simple model for terpenoid cyclase catalysis. Proc Natl Acad Sci U S A 104:5360–5365. doi:10.1073/pnas.0700915104
Jullien F, Moja S, Bony A, Legrand S, Petit C, Benabdelkader T, Poirot K, Fiorucci S, Guitton Y, Nicole F, Baudino S, Magnard JL (2014) Isolation and functional characterization of a τ-cadinol synthase, a new sesquiterpene synthase from Lavandula angustifolia. Plant Mol Biol 84:227–241. doi:10.1007/s11103-013-0131-3
Krieger E, Joo K, Lee J, Lee J, Raman S, Thompson J, Tyka M, Baker D, Karplus K (2009) Improving physical realism, stereochemistry, and side-chain accuracy in homology modeling: Four approaches that performed well in CASP8. Proteins Struct Funct Bioinforma 77:114–122. doi: 10.1002/prot.22570
Landmann C, Fink B, Festner M, Dregus M, Engel KH, Schwab W (2007) Cloning and functional characterization of three terpene synthases from lavender (Lavandula angustifolia). Arch Biochem Biophys 465:417–429. doi:10.1016/j.abb.2007.06.011
Lane A, Boecklemann A, Woronuk GN, Lukman S, Mahmoud SS (2010) A genomics resource for investigating regulation of essential oil production in Lavandula angustifolia. Planta 231:835–845. doi:10.1007/s00425-009-1090-4
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2^(-∆∆CT) method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Martin D, Tholl D, Gershenzon J (2002) Methyl jasmonate induces traumatic resin ducts, terpenoid resin biosynthesis, and terpenoid accumulation in develo** xylem of norway spruce stems 1. Society 129:1003–1018. doi:10.1104/pp.011001 McGarvey
Menzel TR, Weldegergis BT, David A, Boland W, Gols R, Van Loon JJA, Dicke M (2014) Synergism in the effect of prior jasmonic acid application on herbivore-induced volatile emission by Lima bean plants: transcription of a monoterpene synthase gene and volatile emission. J Exp Bot 65:4821–4831. doi:10.1093/jxb/eru242
Miller B, Madilao LL, Ralph S, Bohlmann J (2005) Insect-induced conifer defense. White pine weevil and methyl jasmonate induce traumatic resinosis, de novo formed volatile emissions, and accumulation of terpenoid synthase and putative octadecanoid pathway transcripts in Sitka spruce. Plant Physiol 137:369–382. doi:10.1104/pp.104.050187
Moon T, Wilkinson JM, Cavanagh HMA (2006) Antibacterial activity of essential oils, hydrosols and plant extracts from Australian grown Lavandula spp. Int J Aromather 16:9–14. doi:10.1016/j.ijat.2006.01.007
Moreira X, Sampedro L, Zas R (2009) Defensive responses of Pinus pinaster seedlings to exogenous application of methyl jasmonate: Concentration effect and systemic response. Environ Exp Bot 67:94–100. doi:10.1016/j.envexpbot.2009.05.015
Navia-Giné WG, Gomez SK, Yuan J, Chen F, Korth KL (2009) Insect-induced gene expression at the core of volatile terpene release in Medicago truncatula. Plant Signal Behav 4:639–641. doi:10.1016/j.plaphy.2009.01.008
Ott DS, Yanchuk AD, Huber DPW, Wallin KF (2011) Genetic variation of lodgepole pine, Pinus contorta var. latifolia, chemical and physical defenses that affect mountain pine beetle, Dendroctonus ponderosae, attack and tree mortality. J Chem Ecol 37:1002–1012. doi:10.1007/s10886-011-0003-8
Rivera SB, Swedlund BD, King GJ, Bell RN, Hussey CE, Shattuck-Eidens DM, Wrobel WM, Peiser GD, Poulter CD (2001) Chrysanthemyl diphosphate synthase: isolation of the gene and characterization of the recombinant non-head-to-tail monoterpene synthase from Chrysanthemum cinerariaefolium. Proc Natl Acad Sci USA 98:4373–4378. doi:10.1073/pnas.071543598
Roach CR, Hall DE, Zerbe P, Bohlmann J (2014) Plasticity and evolution of (+)-3-carene synthase and (-)-sabinene synthase functions of a Sitka spruce monoterpene synthase gene family associated with weevil resistance. J Biol Chem 289:23859–23869. doi:10.1074/jbc.M114.571703
Robert JA, Madilao LL, White R, Yanchuk A, King JR, Bohlmann J (2010) Terpenoid metabolite profiling in Sitka spruce identifies association of dehydroabietic acid, (+)-3-carene, and terpinolene with resistance against white pine weevil. Botany-Botanique 88:810–820. doi:10.1139/B10-068
Ruijter JM, Ramakers C, Hoogaars WMH, Karlen Y, Bakker O, Van Den Hoff MJB, Moorman AFM (2009) Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucl Acids Res. doi:10.1093/nar/gkp045
Rynkiewicz MJ, Cane DE, Christianson DW (2001) Structure of trichodiene synthase from Fusarium sporotrichioides provides mechanistic inferences on the terpene cyclization cascade. Proc Natl Acad Sci U S A 98:13543–13548. doi:10.1073/pnas.231313098
Sarker LS, Galata M, Demissie ZA, Mahmoud SS (2012) Molecular cloning and functional characterization of borneol dehydrogenase from the glandular trichomes of Lavandula x intermedia. Arch Biochem Biophys 528:163–170. doi:10.1016/j.abb.2012.09.013
Savage TJ, Croteau R (1993) Biosynthesis of monoterpenes—regiochemistry and stereochemistry of (+)-3-carene biosynthesis. Arch Biochem Biophys 305:581–587
Schilmiller AL, Schauvinhold I, Larson M, Xu R, Charbonneau AL, Schmidt A, Wilkerson C, Last RT, Pichersky E (2009) Monoterpenes in the glandular trichomes of tomato are synthesized from a neryl diphosphate precursor rather than geranyl diphosphate. Proc Natl Acad Sci U S A 106:10865–10870. doi:10.1073/pnas.0904113106\r0904113106 [pii]
Schüttelkopf AW, Van Aalten DMF (2004) PRODRG: A tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr Sect D Biol Crystallogr 60:1355–1363. doi: 10.1107/S0907444904011679
Shi J, Ma C, Qi D, Lv H, Yang T, Peng Q, Chen Z, Lin Z (2015) Transcriptional responses and flavor volatiles biosynthesis in methyl jasmonate-treated tea leaves. BMC Plant Biol 15:233. doi:10.1186/s12870-015-0609-z
Taniguchi S, Hosokawa-Shinonaga Y, Tamaoki D, Yamada S, Akimitsu K, Gomi K (2014) Jasmonate induction of the monoterpene linalool confers resistance to rice bacterial blight and its biosynthesis is regulated by JAZ protein in rice. Plant Cell Environ 37:451–461. doi:10.1111/pce.12169
Tholl D, Lee S (2011) Terpene Specialized Metabolism in Arabidopsis thaliana. Arabidopsis Book 9:e0143. doi:10.1199/tab.0143
Van Schie CCN, Haring MA, Schuurink RC (2007) Tomato linalool synthase is induced in trichomes by jasmonic acid. Plant Mol Biol 64:251–263. doi:10.1007/s11103-007-9149-8
Vickers CE, Gershenzon J, Lerdau MT, Loreto F (2009) A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat Chem Biol 5:283–291. doi:10.1038/nchembio.158
War A, Sharma HC, Paulraj MG, War MY, Ignacimuthu S (2011) Herbivore induced plant volatiles their role in plant defense for pest management. Plant Signal Behav 6:1973–1978. doi:10.4161/psb/6.12.18053
Whittington DA, Wise ML, Urbansky M, Urbansky M, Coates RM, Croteau RB, Christianson DW (2002) Bornyl diphosphate synthase: structure and strategy for carbocation manipulation by a terpenoid cyclase. Proc Natl Acad Sci USA 99:15375–15380. doi:10.1073/pnas.232591099
Williams DC, Mcgarvey DJ, Katahira EJ, Croteau R (1998) Truncation of limonene synthase preprotein provides a fully active “pseudomature” form of this monoterpene cyclase and reveals the function of the amino-terminal arginine pair. BioChemistry 37:12213–12220
Wise M, Savage T, Katahira E (1998) Monoterpene synthases from common sage (Salvia officinalis). J Biol 273:14891–14899. doi:10.1074/jbc.273.24.14891
Wittstock U, Gershenzon J (2002) Constitutive plant toxins and their role in defense against herbivores and pathogens. Curr Opin Plant Biol 5:300–307. doi:10.1016/S1369-5266(02)00264-9
Xu Y-H, Liao Y-C, Zhang Z, Liu J, Sun P-W, Gao Z-H, Sui C, Wei J-H (2016) Jasmonic acid is a crucial signal transducer in heat shock induced sesquiterpene formation in Aquilaria sinensis. Sci Rep 6:21843. doi:10.1038/srep21843
Zeng G (1998) Sticky-end PCR: new method for subcloning. Biotechniques 25:206–208
Acknowledgements
This work was supported through grants and/or in-kind contributions to SSM by UBC, Genome British Columbia, Natural Sciences and Engineering Research Council of Canada, Agriculture and Agri-Food Canada and the BC Ministry of Agriculture (through programs delivered by the Investment Agriculture Foundation of BC).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Adal, A.M., Sarker, L.S., Lemke, A.D. et al. Isolation and functional characterization of a methyl jasmonate-responsive 3-carene synthase from Lavandula x intermedia . Plant Mol Biol 93, 641–657 (2017). https://doi.org/10.1007/s11103-017-0588-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-017-0588-6