Abstract
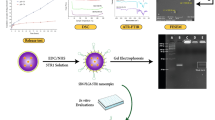
Doxorubicin (DOX) is an efficient chemotherapeutic agent widely used to treat different types of cancer; however, there is an inherent risk of adverse effects due to its unspecific action in healthy cells. In order to enhance the DOX arrival and accumulation inside the cancerous cells, we have developed DOX-loaded nanoparticles (NPs) using the biocompatible polymer poly(lactic-co-glycolic acid) (PLGA). pH sensitivity was achieved by incorporation of the surfactant, 77KS, while poloxamer was explored as stabilizer and chemosensitizer. The protein transferrin (Tf) was conjugated to the NPs with the role to actively targeting them to the cancerous cells. The nanoprecipitation method yielded NPs with size about 100 nm, with polydispersity index around 0.20 and a negative zeta potential. Transmission electron microscopy and infrared spectroscopy confirmed the shape and the functional groups presence. DOX release from the NPs followed a control and pH-sensitive pattern, allowing accelerated DOX release in acidic conditions. Through the hemolysis assay, using the erythrocyte as a model for the endosomal membrane, it was evidenced the pH-sensitive membranolytic behavior of the NPs. Furthermore, the NPs were safe and compatible with blood. Finally, the formulations were applied to tumor and non-tumor cell lines, HeLa and HaCaT, respectively. Over 72 h of incubation, the Tf-conjugated NPs induced a notable reduction in HeLa cell growth and were able to protect the HaCaT cells from the DOX unspecific cytotoxicity. The results suggest that the dual-active targeting promoted by 77KS and Tf is a promising platform to overcome the side effects of conventional chemotherapeutic drugs and nontargeted nanosystems.

Graphical







Similar content being viewed by others
References
AbuHammad S, Zihlif M (2013) Gene expression alterations in doxorubicin resistant MCF7 breast cancer cell line. Genomics 101:213–220. https://doi.org/10.1016/j.ygeno.2012.11.009
Baker EN, Baker HM, Kidd RD (2002) Lactoferrin and transferrin: functional variations on a common structural framework. Biochem Cell Biol 80:27–34. https://doi.org/10.1139/o01-153
Balasubramanian S, Girija AR, Nagaoka Y, Iwai S, Suzuki M, Kizhikkilot V, Yoshida Y, Maekawa T, Nair SD (2013) Curcumin and 5-fluorouracil-loaded, folate- and transferrin-decorated polymeric magnetic nanoformulation: a synergistic cancer therapeutic approach, accelerated by magnetic hyperthermia. Int J Nanomedicine 9:437–459. https://doi.org/10.2147/IJN.S49882
Barenholz Y (2012) Doxil® — the first FDA-approved nano-drug: lessons learned. J Control Release 160:117–134. https://doi.org/10.1016/j.jconrel.2012.03.020
Batrakova EV, Kabanov AV (2008) Pluronic block copolymers: evolution of drug delivery concept from inert nanocarriers to biological response modifiers. J Control Release 130:98–106. https://doi.org/10.1016/j.jconrel.2008.04.013
Betancourt T, Brown B, Branon-Peppas L (2007) Doxorubicin-loaded PLGA nanoparticles by nanoprecipitation: preparation, characterization and in vitro evaluation. Nanomedicine 2:219–232. https://doi.org/10.2217/17435889.2.2.219
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Chang J, Jallouli Y, Kroubi M, Yuan X, Feng W, Kang C, Pu P, Betbeder D (2009) Characterization of endocytosis of transferrin-coated PLGA nanoparticles by the blood–brain barrier. Int J Pharm 379:285–292. https://doi.org/10.1016/j.ijpharm.2009.04.035
Chang J, Paillard A, Passirani C, Morille M, Benoit J, Betbeder D, Garcion E (2012) Transferrin adsorption onto PLGA nanoparticles governs their interaction with biological systems from blood circulation to brain Cancer cells. Pharm Res 29:1495–1505. https://doi.org/10.1007/s11095-011-0624-1
Chittasupho C, Lirdprapamongkol K, Kewsuwan P, Sarisuta N (2014) Targeted delivery of doxorubicin to A549 lung cancer cells by CXCR4 antagonist conjugated PLGA nanoparticles. Eur J Pharm Biopharm 88:529–538. https://doi.org/10.1016/j.ejpb.2014.06.020
Cryer AM, Thorley AJ (2019) Nanotechnology in the diagnosis and treatment of lung cancer. Pharm Ther, in press. https://doi.org/10.1016/j.pharmthera.2019.02.010
Cui Y, Xu Q, Chow PK, Wang D, Wang C (2013) Transferrin-conjugated magnetic silica PLGA nanoparticles loaded with doxorubicin and paclitaxel for brain glioma treatment. Biomaterials 34:8511–8520. https://doi.org/10.1016/j.biomaterials.2013.07.075
Danhier F, Feron O, Préat V (2010) To exploit the tumor microenvironment: passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. J Control Release 148:135–146. https://doi.org/10.1016/j.jconrel.2010.08.027
Daniels TR, Delgado T, Rodriguez JA, Helguera G, Penichet ML (2006) The transferrin receptor part I: biology and targeting with cytotoxic antibodies for the treatment of cancer. Clin Immunol 121:144–158. https://doi.org/10.1016/j.clim.2006.06.010
Fessi H, Puisieux F, Devissaguet JP, Ammoury N, Benita S (1989) Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int J Pharm 55:R1–R4. https://doi.org/10.1016/0378-5173(89)90281-0
Flatley M, Dodwell D (2019) Adjuvant treatment for breast cancer. Surgery (Oxford) 34:43–46. https://doi.org/10.1016/j.mpsur.2015.10.003
Fornaguera C, Calderó G, Mitjans M, Vinardell MP, Solansa C, Vauthier C (2015) Interactions of PLGA nanoparticles with blood components: protein adsorption, coagulation, activation of the complement system and hemolysis studies. Nanoscale 7:6045–6059. https://doi.org/10.1039/c5nr00733j
Frasco MF, Almeida GM, Santos-Silva F, Pereira M do C, Coelho MAN (2015).Transferrin surface-modified PLGA nanoparticles-mediated delivery of a proteasome inhibitor to human pancreatic cancer cells. J Biomed Mater Res A103A:1476–1484. https://doi.org/10.1002/jbm.a.35286
Gewirtz DA (1999) A critical evaluation of the mechanisms of action proposed for the antitumor effects of the anthracycline antibiotics adriamycin and daunorubicin. Biochem Pharmacol 57:727–741. https://doi.org/10.1016/s0006-2952(98)00307-4
Hare JI, Lammers T, Ashford MB, Puri S, Storm G, Barry ST (2017) Challenges and strategies in anti-cancer nanomedicine development: an industry perspective. Adv Drug Deliv Rev 108:25–38. https://doi.org/10.1016/j.addr.2016.04.025
He Y, **ng L, Cui P, Zhang J, Zhu Y, Qiao J, Lyu J, Zhang M, Luo C, Zhou Y, Lu N, Jiang H (2017) Transferrin-inspired vehicles based on pH-responsive coordination bond to combat multidrug-resistant breast cancer. Biomaterials 113:266–278. https://doi.org/10.1016/j.biomaterials.2016.11.001
ICH – International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use Q2 R1: Guideline on Validation of Analytical Procedure – Methodology. (2005)
INVITOX PROTOCOL (1992) Number 37: red blood cell test system
Kumari M, Purohit MP, Patnaik S, Shukla Y, Kumar P, Gupta KC (2018) Curcumin loaded selenium nanoparticles synergize the anticancer potential of doxorubicin contained in self-assembled, cell receptor targeted nanoparticles. Eur J Pharm Biopharm 130:185–199. https://doi.org/10.1016/j.ejpb.2018.06.030
Lee ES, Oh KT, Kim D, Youn YS, Bae YH (2007) Tumor pH-responsive flower-like micelles of poly(L-lactic acid)-b-poly (ethylene glycol)-b-poly(L-histidine). J Control Release 123:19–26. https://doi.org/10.1016/j.jconrel.2007.08.006
Luu AZ, Chowdhury B, Al-Omran M, Teoh H, Hess DA, Verma S (2018) Role of endothelium in doxorubicin-induced cardiomyopathy. JACC Basic Transl Sci 3:861–871. https://doi.org/10.1016/j.jacbts.2018.06.005
Macedo LB, Nogueira-Librelotto DR, Vargas J, Scheeren LE, Vinardell MP, Rolim CMB (2019) Poly (ɛ-Caprolactone) nanoparticles with pH-responsive behavior improved the In Vitro antitumor activity of methotrexate. AAPS PharmSciTech 20:165–177. https://doi.org/10.1208/s12249-019-1372-5
Malinovskaya Y, Melnikov P, Baklaushev V, Gabashvili A, Osipova N, Mantrov S, Ermolenko Y, Maksimenko O, Gorshkova M, Balabanyan V, Kreuter J, Gelperina S (2017) Delivery of doxorubicin-loaded PLGA nanoparticles into U87 human glioblastoma cells. Int J Pharm 524:77–90. https://doi.org/10.1016/j.ijpharm.2017.03.049
Montha W, Maneeprakorn W, Buatong N, Tang I-M, Pon-On W (2016) Synthesis of doxorubicin-PLGA loaded chitosan stabilized (Mn, Zn)Fe2O4 nanoparticles: biological activity and pH-responsive drug release. Mater Sci Eng C 59:235–240. https://doi.org/10.1016/j.msec.2015.09.098
Muhamad N, Plengsuriyakarn T, Na-Bangchang K (2018) Application of active targeting nanoparticle delivery system for chemotherapeutic drugs and traditional/herbal medicines in cancer therapy: a systematic review. Int J Nanomedicine 13:3921–3935. https://doi.org/10.2147/IJN.S165210
Neun BW, Dobrovolskaia MA (2010) Method for in vitro analysis of nanoparticle thrombogenic properties. Characterization of Nanoparticles Intended for Drug Delivery. Methods Mol Biol 697 chapter 24
Nogueira DR, Mitjans M, Infante MR, Vinardell MP (2011a) The role of counterions in the membrane-disruptive properties of pH-sensitive lysine-based surfactants. Acta Biomater 7:2846–2856. https://doi.org/10.1016/j.actbio.2011.03.017
Nogueira DR, Mitjans M, Infante MR, Vinardell MP (2011b) Comparative sensitivity of tumor and non-tumor cell lines as a reliable approach for in vitro cytotoxicity screening of lysine-based surfactants with potential pharmaceutical applications. Int J Pharm 420:51–58. https://doi.org/10.1016/j.ijpharm.2011.08.020
Nogueira DR, Tavano L, Mitjans M, Pérez L, Infante MR, Vinardell MP (2013) In vitro antitumor activity of methotrexate via pH-sensitive chitosan nanoparticles. Biomaterials 34:2758–2772. https://doi.org/10.1016/j.biomaterials.2013.01.005
Nogueira DR, Scheeren LE, Vinardell MP, Mitjans M, Infante MR, Rolim CMB (2015) Nanoparticles incorporating pH-responsive surfactants as a viable approach to improve the intracellular drug delivery. Mater Sci Eng C 57:100–106. https://doi.org/10.1016/j.msec.2015.07.036
Nogueira DR, Scheeren LE, Macedo LB, Marcolino AIP, Vinardell MP, Mitjans M, Infante MR, Farooqi A, Rolim CMB (2016) Inclusion of a pH-responsive amino acid-based amphiphile in methotrexate-loaded chitosan nanoparticles as a delivery strategy in cancer therapy. Amino Acids 48:157–168. https://doi.org/10.1007/s00726-015-2075-1
Nogueira-Librelotto DR, Scheeren LE, Vinardell MP, Mitjans M, Rolim CMB (2016) Chitosan-tripolyphosphate nanoparticles functionalized with a pH-responsive amphiphile improved the in vitro antineoplastic effects of doxorubicin. Colloid Surface B 147:326–335. https://doi.org/10.1016/j.colsurfb.2016.08.014
Nogueira-Librelotto DR, Codevilla CF, Farooqi A, Rolim CMB (2017) Transferrin-conjugated Nanocarriers as active-targeted drug delivery platforms for cancer therapy. Cur Pharm Des 23:454–466. https://doi.org/10.2174/1381612822666161026162347
Nothnagel L, Wacker MG (2018) How to measure release from nanosized carriers? Eur J Pharm Sci 120:199–211. https://doi.org/10.1016/j.ejps.2018.05.004
Pandey SK, Patel DK, Maurya AK, Thakur R, Mishra DP, Vinayak M, Haldar C, Maiti P (2016) Controlled release of drug and better bioavailability using poly(lacticacid-co-glycolic acid) nanoparticles. Int J Biol Macromol:8999–8110. https://doi.org/10.1016/j.ijbiomac.2016.04.065
Pereverzeva E, Treschalin I, Treschalin M, Arantseva D, Ermolenko Y, Kumskova N, Maksimenko O, Balabanyan V, Kreuter J, Gelperina S (2019) Toxicological study of doxorubicin-loaded PLGA nanoparticles for the treatment of glioblastoma. Int J Pharm 554:161–178. https://doi.org/10.1016/j.ijpharm.2018.11.014
Pillai GJ, Greeshma MM, Menon D (2015) Impact of poly(lactic-co-glycolic acid) nanoparticle surface charge on protein, cellular and haematological interactions. Colloid Surface B 136:1058–1066. https://doi.org/10.1016/j.colsurfb.2015.10.047
Ritger PL, Peppas NA (1987) A simple equation for description of solute release I. Fickian and non-Fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J Control Release 5:23–36. https://doi.org/10.1016/0168-3659(87)90034-4
Sahoo SK, Labhasetwar V (2005) Enhanced antiproliferative activity of tranferrin-conjugated paclitaxel-loaded nanoparticles is mediated via sustained intracellular drug retention. Mol Pharm 2:373–383. https://doi.org/10.1021/mp050032z
Sanchez L, Mitjans M, Infante MR, Vinardell MP (2006a) Potential irritation of lysine derivative surfactants by hemolysis and HaCaT cell viability. Toxicol Lett 161:53–60. https://doi.org/10.1016/j.toxlet.2005.07.015
Sanchez L, Mitjans M, Infante MR, Vinardell MP (2006b) Determination of interleukin-1in human NCTC 2544 keratinocyte cells as a predictor of skin irritation from lysine-based surfactants. Toxicol Lett 167:40–46. https://doi.org/10.1016/j.toxlet.2006.08.006
Scheeren LE, Nogueira DR, Macedo LB, Vinardell MP, Mitjans M, Infante MR, Rolim CMB (2016) PEGylated and poloxamer-modified chitosan nanoparticles incorporating a lysine-based surfactant for pH-triggered doxorubicin release. Colloid Surface B 138:117–127. https://doi.org/10.1016/j.colsurfb.2015.11.049
Scheeren LE, Nogueira DR, Fernandes JR, Marcolino AIP, Macedo LB, Vinardell MP, Rolim CMB (2017) Comparative study of reversed-phase high-performance liquid chromatography and ultraviolet–visible spectrophotometry to determine doxorubicin in pH-sensitive nanoparticles. Anal Lett 51:1445–1463. https://doi.org/10.1080/00032719.2017.1380034
Sharma S, Parmar A, Kori S, Sandhir R (2016) PLGA-based nanoparticles: a new paradigm in biomedical applications. Trends Anal Chem 80:30–40. https://doi.org/10.1016/j.trac.2015.06.014
Shen ZM, Yang JT, Feng Y, Wu CC (1992) Conformational stability of porcine serum transferrin. Prot Sci I:1477–1484. https://doi.org/10.1002/pro.5560011109
Shindelman JE, Ortmeyer AE, Sussman HH (1981) Demonstration of the transferrin receptor in human breast cancer tissue. Potential marker for identifying dividing cells. Int J Cancer 27:329–334. https://doi.org/10.1002/ijc.2910270311
Singhal JP, Ray AR (2002) Synthesis of blood compatible polyamide block copolymers. Biomaterials 23:1139–1145. https://doi.org/10.1016/S0142-9612(01)00228-9
Soe ZC, Kwon JB, Thapa RK, Ou W, Nguyen HT, Gautam M, Oh KT, Choi H, Ku SK, Yong CS, Kim JO (2019) Transferrin-conjugated polymeric nanoparticle for receptor-mediated delivery of doxorubicin in doxorubicin-resistant breast cancer cells. Pharmaceutics 11:63–80. https://doi.org/10.3390/pharmaceutics11020063
Sriraman SK, Salzano G, Sarisozen C, Torchilin V (2016) Anti-cancer activity of doxorubicin-loaded liposomes co-modified with transferrin and folic acid. Eur J Pharm Biopharm 105:40–49. https://doi.org/10.1016/j.ejpb.2016.05.023
Swider E, Koshkina O, Tel J, Cruz LJ, de Vries IJM, Srinivas M (2018) Customizing poly(lactic-co-glycolic acid) particles for biomedical applications. Acta Biomater 73:38–51. https://doi.org/10.1016/j.actbio.2018.04.006
Tavano L, Aiello R, Ioele G, Picci N, Muzzalupo R (2014) Niosomes from glucuronic acid-based surfactant as new carriers for cancer therapy: preparation, characterization and biological properties. Colloid Surface B 118:7–13. https://doi.org/10.1016/j.colsurfb.2014.03.016
Tewes F, Munnier E, Antoon B, Okassa LN, Cohen-Jonathan S, Marchais H, Douziech-Eyrolles L, Souce M, Dubois P, Chourpa I (2007) Comparative study of doxorubicin-loaded poly(lactide-co-glycolide) nanoparticles prepared by single and double emulsion methods. Eur J Pharm Biopharm 66:488–492. https://doi.org/10.1016/j.ejpb.2007.02.016
Thorn CF, Oshiro C, Marsh S, Hernandez-Boussard T, McLeod H, Klein TE, Altman RB (2011) Doxorubicin pathways: pharmacodynamics and adverse effects. Pharmacogenet Genomics 21(7):440–446. https://doi.org/10.1097/FPC.0b013e32833ffb56
Tian L, Bae YH (2012) Cancer nanomedicines targeting tumor extracellular pH. Colloid Surface B 99:116–126. https://doi.org/10.1016/j.colsurfb.2011.10.039
Tsuji T, Yoshitomi Y, Usukura J (2013) Endocytic mechanism of transferrin-conjugated nanoparticles and the effects of their size and ligand number on the efficiency of drug delivery. Microscopy 62:341–352. https://doi.org/10.1093/jmicro/dfs080
Vives MA, Infante MR, Garcia E, Selve C, Maugras M, Vinardell MP (1999) Erythrocyte hemolysis and shape changes induced by new lysine-derivate surfactants. Chem-Biol Interac 118:1–18. https://doi.org/10.1016/s0009-2797(98)00111-2
Wang H, Zhao Y, Wang H, Gong J, He H, Shin MC, Yang VC, Huang Y (2014) Low-molecular-weight protamine-modified PLGA nanoparticles for overcoming drug-resistant breast cancer. J Control Release 192:47–56. https://doi.org/10.1016/j.jconrel.2014.06.051
Zhang X, Li J, Yan M (2016) Targeted hepatocellular carcinoma therapy: transferrin modified, self-assembled polymeric nanomedicine for co-delivery of cisplatin and doxorubicin. Drug Dev Ind Pharm 40:1590–1599. https://doi.org/10.3109/03639045.2016.1160103
Funding
This research was supported by Projects 447548/2014-0 and 401069/2014-1 of the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq - Brazil). L.E.S thanks the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the PhD fellowship at Universidade Federal de Santa Maria and CNPq for the PhD internship at Universitat de Barcelona (grant number 204255/2018-0). D.R.N-L. thanks CNPq-Brazil for the Postdoctoral grant (grant number 150920/2018-0).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Scheeren, L.E., Nogueira-Librelotto, D.R., Macedo, L.B. et al. Transferrin-conjugated doxorubicin-loaded PLGA nanoparticles with pH-responsive behavior: a synergistic approach for cancer therapy. J Nanopart Res 22, 72 (2020). https://doi.org/10.1007/s11051-020-04798-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-020-04798-7