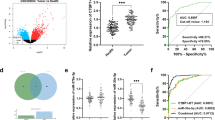
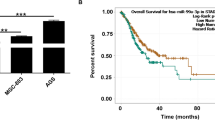
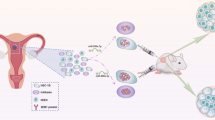
Abstract
Introduction
Colorectal cancer (CRC) is the second common cancer and the fourth major reason of cancer death worldwide. Dysregulation of intracellular pathways, such as TGF-β/SMAD signaling, contributes to CRC development. MicroRNAs (miRNAs) are post-transcriptional regulators that are involved in CRC pathogenesis. Here, we aimed to investigate the effect of miR-3613-3p on the TGF-β /SMAD signaling pathway in CRC.
Methods & results
Bioinformatics analysis suggested that miR-3613-3p is a regulator of TGF-Β signaling downstream genes. Then, miR-3613-3p overexpression was followed by downregulation of TGF-βR1, TGF-βR2, and SMAD2 expression levels, detected by RT-qPCR. Additionally, dual luciferase assay supported the direct interaction of miR-3613-3p with 3’UTR sequences of TGF-βR1 and TGF-βR2 genes. Furthermore, reduced SMAD3 protein level following the miR-3613-3p overexpression verified its suppressive effect against TGF-β signaling in HCT-116 cells, detected by western blot analysis. Finally, miR-3613-3p overexpression induced sub-G1 arrest in HCT116 cells, detected by flow cytometry, and promoted downregulation of cyclin D1 protein expression, which was detected by western blotting analysis.
Conclusion
Our findings indicated that miR-3613-3p plays an important role in CRC by targeting the TGF-β/SMAD signaling pathway and could be considered as a new candidate for further therapy investigations.





Similar content being viewed by others
Data availability
All of the related data including originals of the experiments will be available upon request.
Abbreviations
- CRC:
-
Colorectal cancer
- WNT:
-
Wingless/Integrated
- MAPK:
-
Mitogen-Activated Protein Kinase
- TGF-β:
-
Transforming Growth Factor Beta
- TGF-βR:
-
Transforming Growth Factor Beta Receptor
- SMAD:
-
Small mothers against decapentaplegic
- R-SMAD:
-
Receptor-regulated Smad
- CO-SMAD:
-
Common mediator Smad
- SMAD6/7:
-
Inhibitory Smad 6/7
- miRNAs:
-
MicroRNAs
- MREs:
-
miRNA recognition elements
- 3’UTR:
-
3’ Untranslated Region
References
Siegel R, Sandeep Wagle N, Cercek A, Smith R, Jemal A (2023) Colorectal cancer statistics,CA. Cancer J Clin 73:233–254
Cao Z (2015) N.J.A.j.o.u. Kyprianou, mechanisms navigating the TGF-β pathway in prostate cancer. 2(1):11–18
Nature C (2012) Comprehensive molecular characterization of human colon and rectal cancer. 487(7407):330
Pellatt AJ et al (2018) The TGF-Β-signaling pathway and colorectal cancer: associations between dysregulated genes and miRNAs. 16(1):191
Dandan Peng (2022) Targeting TGF-β signal transduction for fibrosis and cancer therapy. Mole Cancer 21(1):104
Ringuette Goulet, C., and Pouliot, F. TGF-Β signaling in the Tumor Microenvironment. Adv Exp Med Biol. (2021) :1270:89–105
Eulalio A, Huntzinger E, Izaurralde EJC (2008) Getting to the root of miRNA. -mediated gene Silencing 132(1):9–14
Chekulaeva M (2009) W.J.C.o.i.c.b. Filipowicz, mechanisms of miRNA-mediated post-transcriptional regulation in animal cells. 21(3):452–460
Pereira DM et al (2013) Delivering Promise miRNA cancer Ther 18(5–6):282–289
Svoronos AA, Engelman DM (2016) J.J.C.r. Slack, OncomiR or tumor suppressor? The duplicity of microRNAs in cancer. 76(13):3666–3670
Rustam Effendi-Ys., Colonoscopy, Biomarkers, and Targeted Therapy in Colorectal Cancer Acta Med Indones, (2022) Jul;54(3):476–486
Tanksley JP (2013) Tales of Three Signaling pathways: EGFR, TGF-Β, and WNT signaling in the GI Tract. Vanderbilt University
Iordanskaia T, Nawshad A (2011) Mechanisms of transforming growth factor β induced cell cycle arrest in palate development. J Cell Physiol 226(5):1415–1424
Huang X et al Dissecting miRNA signature in colorectal cancer progression and metastasis Cancer Lett., 2021. Mar 31;501:66–82
- Sheng K, Pong M, Gullerova (2018) Noncanonical functions of microRNA pathway enzymes - Drosha, DGCR8, Dicer and Ago proteins. FEBS Lett., Sep;592(17):2973–2986
Rusk N (2008) When microRNAs activate translation. Nat Methods 5(2):122
Sebastian-delaCruz M et al (2021) The role of lncRNAs in gene expression regulation through mRNA stabilization. Non-coding RNA 7(1):3
**ao B et al (2012) Increased miR-146a in gastric cancer directly targets SMAD4 and is involved in modulating cell proliferation and apoptosis. Oncol Rep 27(2):559–566
Jahangirimoez M et al (2020) Hsa-miR-587 regulates TGF-Β/SMAD signaling and promotes cell cycle progression. Cell J (Yakhteh) 22(2):158
Jafarzadeh M, Soltani BM (2016) Hsa-Mir-590-5p Interaction with SMAD3 transcript supports its Regulatory Effect on the TGF-Β signaling pathway. Cell J (Yakhteh) 18(1):7
Abedini Bakhshmand E et al (2018) Hsa-miR‐5582‐3P regulatory effect on TGF-Β signaling through targeting of TGF-Β‐R1, TGF-Β‐R2, SMAD3, and SMAD4 transcripts. J Cell Biochem 119(12):9921–9930
Dokanehiifard S, Soltani BM (2019) TrkC-miR2 regulates TGF-Β signaling pathway through targeting of SMAD3 transcript. J Cell Biochem 120(2):2634–2641
Mori Y, Chen S-J, Varga J (2000) Modulation of endogenous smad expression in normal skin fibroblasts by transforming growth factor-β. Exp Cell Res 258(2):374–383
Wu W, Wang X, Yu X, Lan HY Smad3 signatures in renal inflammation and fibrosis. Int J Biol Sci 2022 Mar 28;18(7):2795–2806
Gordan JD, Thompson CB, Simon MC (2007) HIF and c-Myc: sibling rivals for control of cancer cell metabolism and proliferation. Cancer Cell 12(2):108–113
Zhang K et al (2020) The dichotomous role of TGF-β in controlling liver cancer cell survival and proliferation. J Genet Genomics 47(9):497–512
Chen C, Pan Y, Bai L, Chen H, Duan Z, Si Q, Zhu R, Chuang T, Luo Y MicroRNA-3613-3p functions as a tumor suppressor and represents a novel therapeutic target in breast cancer. Breast Cancer Res. 2021 Jan 25;23(1):12
Hu Y, He J, He L, Xu B, Wang QJ (2021) Expression and function of Smad7 in autoimmune and inflammatory diseases. Mol Med (Berl) 99(9):1209–1220
Acknowledgements
We thank 4402 Lab mates for their cooperation’s.
Funding
This work was supported by Tarbiat Modares University, National Science Foundation (INSF# 96012116) financial aids.
Author information
Authors and Affiliations
Contributions
MJ & TH; performed the experiments, TH & PB; Helped the experiments BMS; designed and supervised the experiments, MB; advised the research.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest with any financial organization regarding the material discussed in the manuscript. Also, none of the authors have any non-financial conflict of interest.
Ethical approval
This study was carried out in accordance with the recommendations of the Research Ethics Committee of Tarbiat Modares University. All subjects gave written informed consent in accordance with the Declaration of Helsinki. Permission from the responsible authorities was granted. (Ethical code: IR.MODARES.REC.1400.136)
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jafarian, M., Hasannia, T., Badameh, P. et al. Introduction of miR-3613-3p as a regulator of transforming growth factor-β (TGF-β) signaling pathway in colorectal cancer. Mol Biol Rep 51, 728 (2024). https://doi.org/10.1007/s11033-024-09419-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09419-3