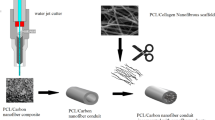
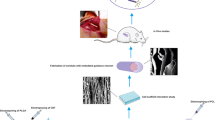
Abstract
Development of tissue-engineered alternatives to nerve autografts remains a serious challenge particularly considering the electroactive properties of native peripheral nerves. In this study, three-dimensional (3D) conductive nerve scaffolds composed of polycaprolactone (PCL) and carbon nanotubes (CNTs) were fabricated using an integration molding method. In vitro, the combination of PCL/CNTs scaffolds with electrical stimulation that mimic the endogenous electric fields could significantly promote Schwann cell proliferation and neurotrophic factor genes expression. In vivo, the effects of PCL/CNTs scaffolds on peripheral nerve regeneration were investigated using a 15-mm rat sciatic nerve defect model. Functional and histological tests indicated that PCL/CNTs scaffolds could effectively promote nerve regeneration and functional recovery. Therefore, PCL/CNTs scaffolds are promising candidates for peripheral nerve restoration.
Graphical abstract






Similar content being viewed by others
References
Jiang BG, Han N, Rao F et al (2017) Advance of peripheral nerve injury repair and reconstruction. Chin Med J (Engl) 130:2996–2998
Griffin JW, Hogan MV, Chhabra AB et al (2013) Peripheral nerve repair and reconstruction. J Bone Joint Surg Am 95:2144–2151
Riccio M, Marchesini A, Pugliese P et al (2019) Nerve repair and regeneration: Biological tubulization limits and future perspectives. J Cell Physiol 234:3362–3375
Vijayavenkataraman S (2020) Nerve guide conduits for peripheral nerve injury repair: a review on design, materials and fabrication methods. Acta Biomater 106:54–69
Gu X, Ding F, Williams DF (2014) Neural tissue engineering options for peripheral nerve regeneration. Biomaterials 35:6143–6156
Yi S, Xu L, Gu X (2019) Scaffolds for peripheral nerve repair and reconstruction. Exp Neurol 319:112761
Gordon T (2016) Electrical stimulation to enhance axon regeneration after peripheral nerve injuries in animal models and humans. Neurotherapeutics 13:295–310
Wang L, Lu C, Yang S et al (2020) A fully biodegradable and self-electrified device for neuroregenerative medicine. Sci Adv 6:eabc6686
Al-Majed AA, Neumann CM, Brushart TM et al (2000) Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J Neurosci 20:2602–2608
Elzinga K, Tyreman N, Ladak A et al (2015) Brief electrical stimulation improves nerve regeneration after delayed repair in Sprague Dawley rats. Exp Neurol 269:142–153
Guo B, Ma PX (2018) Conducting polymers for tissue engineering. Biomacromol 19:1764–1782
Fraczek-Szczypta A (2014) Carbon nanomaterials for nerve tissue stimulation and regeneration. Mater Sci Eng C Mater Biol Appl 34:35–49
Carvalho CR, Silva-Correia J, Oliveira JM et al (2019) Nanotechnology in peripheral nerve repair and reconstruction. Adv Drug Deliv Rev 148:308–343
Manousiouthakis E, Park J, Hardy JG et al. (2021) Towards the translation of electroconductive organic materials for regeneration of neural tissues. Acta Biomater
Fabbro A, Prato M, Ballerini L (2013) Carbon nanotubes in neuroregeneration and repair. Adv Drug Deliv Rev 65:2034–2044
Hwang JY, Shin US, Jang WC et al (2013) Biofunctionalized carbon nanotubes in neural regeneration: a mini-review. Nanoscale 5:487–497
Cellot G, Cilia E, Cipollone S et al (2009) Carbon nanotubes might improve neuronal performance by favouring electrical shortcuts. Nat Nanotechnol 4:126–133
Hu H, Ni Y, Montana V et al (2004) Chemically functionalized carbon nanotubes as substrates for neuronal growth. Nano Lett 4:507–511
Mattson MP, Haddon RC, Rao AM (2000) Molecular functionalization of carbon nanotubes and use as substrates for neuronal growth. J Mol Neurosci 14:175–182
Lovat V, Pantarotto D, Lagostena L et al (2005) Carbon nanotube substrates boost neuronal electrical signaling. Nano Lett 5:1107–1110
Wu S, Qi Y, Shi W et al. (2020) Electrospun conductive nanofiber yarns for accelerating mesenchymal stem cells differentiation and maturation into Schwann cell-like cells under a combination of electrical stimulation and chemical induction. Acta Biomater
Galvan-Garcia P, Keefer EW, Yang F et al (2007) Robust cell migration and neuronal growth on pristine carbon nanotube sheets and yarns. J Biomater Sci Polym Ed 18:1245–1261
Wang L, Wu Y, Hu T et al (2019) Aligned conductive core-shell biomimetic scaffolds based on nanofiber yarns/hydrogel for enhanced 3D neurite outgrowth alignment and elongation. Acta Biomater 96:175–187
Su WT, Shih YA (2015) Nanofiber containing carbon nanotubes enhanced PC12 cell proliferation and neuritogenesis by electrical stimulation. Biomed Mater Eng 26(Suppl 1):S189–S195
Gonçalves C, Ribeiro J, Pereira T et al (2016) Preparation and characterization of electrical conductive PVA based materials for peripheral nerve tube-guides. J Biomed Mater Res A 104:1981–1987
Dash TK, Konkimalla VB (2012) Poly-є-caprolactone based formulations for drug delivery and tissue engineering: a review. J Control Release 158:15–33
Zhu L, Wang K, Ma T et al (2017) Noncovalent bonding of RGD and YIGSR to an electrospun poly(ε-caprolactone) conduit through peptide self-assembly to synergistically promote sciatic nerve regeneration in rats. Adv Healthc Mater 6:1600860
Reid AJ, de Luca AC, Faroni A et al (2013) Long term peripheral nerve regeneration using a novel PCL nerve conduit. Neurosci Lett 544:125–130
Jiang X, Mi R, Hoke A et al (2014) Nanofibrous nerve conduit-enhanced peripheral nerve regeneration. J Tissue Eng Regen Med 8:377–385
Wang J, Cheng Y, Chen L et al (2019) In vitro and in vivo studies of electroactive reduced graphene oxide-modified nanofiber scaffolds for peripheral nerve regeneration. Acta Biomater 84:98–113
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
de Medinaceli L, Freed WJ, Wyatt RJ (1982) An index of the functional condition of rat sciatic nerve based on measurements made from walking tracks. Exp Neurol 77:634–643
Jessen KR, Mirsky R, Lloyd AC (2015) Schwann cells: development and role in nerve repair. Cold Spring Harb Perspect Biol 7:a020487
Ghasemi-Mobarakeh L, Prabhakaran MP, Morshed M et al (2009) Electrical stimulation of nerve cells using conductive nanofibrous scaffolds for nerve tissue engineering. Tissue Eng Part A 15:3605–3619
Zhang J, Qiu K, Sun B et al (2014) The aligned core-sheath nanofibers with electrical conductivity for neural tissue engineering. J Mater Chem B 2:7945–7954
Frostick SP, Yin Q, Kemp GJ (1998) Schwann cells, neurotrophic factors, and peripheral nerve regeneration. Microsurgery 18:397–405
Hoyng SA, De Winter F, Gnavi S et al (2014) A comparative morphological, electrophysiological and functional analysis of axon regeneration through peripheral nerve autografts genetically modified to overexpress BDNF, CNTF, GDNF, NGF, NT3 or VEGF. Exp Neurol 261:578–593
Liu S, Liu Y, Zhou L et al (2021) XT-type DNA hydrogels loaded with VEGF and NGF promote peripheral nerve regeneration via a biphasic release profile. Biomater Sci 9:8221–8234
Wang B, Lu CF, Liu ZY et al (2022) Chitin scaffold combined with autologous small nerve repairs sciatic nerve defects. Neural Regen Res 17:1106–1114
Yang S, Zhu J, Lu C et al (2022) Aligned fibrin/functionalized self-assembling peptide interpenetrating nanofiber hydrogel presenting multi-cues promotes peripheral nerve functional recovery. Bioact Mater 8:529–544
Mata M, Alessi D, Fink DJ (1990) S100 is preferentially distributed in myelin-forming Schwann cells. J Neurocytol 19:432–442
Posmantur R, Hayes RL, Dixon CE et al (1994) Neurofilament 68 and neurofilament 200 protein levels decrease after traumatic brain injury. J Neurotrauma 11:533–545
Saffari TM, Bedar M, Hundepool CA et al (2020) The role of vascularization in nerve regeneration of nerve graft. Neural Regen Res 15:1573–1579
Tondnevis F, Keshvari H, Mohandesi JA (2020) Fabrication, characterization, and in vitro evaluation of electrospun polyurethane-gelatin-carbon nanotube scaffolds for cardiovascular tissue engineering applications. J Biomed Mater Res B Appl Biomater 108:2276–2293
Jiang C, Wang K, Liu Y et al (2021) Using wet electrospun PCL/Gelatin/CNT yarns to fabricate textile-based scaffolds for vascular tissue engineering. ACS Biomater Sci Eng 7:2627–2637
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (31771322); Bei**g Natural Science Foundation of China (7212121) and Shenzhen Science and Technology Plan Project of China (JCYJ20190806162205278).
Author information
Authors and Affiliations
Contributions
WP was involved in writing—original draft and visualization. WP and LZ were involved in conceptualization. WP, WZ and LZ were involved in methodology. WP and WZ were involved in data curation. SL, CL and MZ performed investigation. YW and PZ were involved in project administration. YW was involved in supervision. PZ was involved in writing—review & editing and funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Annela M. Seddon.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pi, W., Zhou, L., Zhang, W. et al. Three-dimensional conductive polycaprolactone/carbon nanotubes scaffolds for peripheral nerve regeneration. J Mater Sci 57, 11289–11299 (2022). https://doi.org/10.1007/s10853-022-07336-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-022-07336-z