Abstract
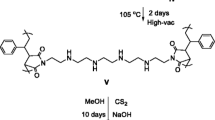
A methylene crosslinked calix[4]arene tetracarboxylic acid resin that exhibits high precious metal adsorption properties was prepared. The maximum loading capacity of this resin was compared with that of an unmodified methylene crosslinked calix[4]arene resin to verify its applicability for platinum and palladium adsorption. The introduction of multiple acetic acid groups improved the adsorption capacity for divalent palladium, while that for tetravalent platinum was slightly suppressed. The newly obtained capacity of palladium was 1.17 mmol/g, while that of platinum was 0.23 mmol/g. The stoichiometry of Pd(II) to the calix[4]arene tetracarboxylic acid monomer in the methylene crosslinked tetracarboxylic acid calix[4]arene resin was found to be 1:2, while that of Pt(IV) to the same monomer was 9:1. Linear and non-linear methods were applied to evaluate the fitting of the Langmuir and Freundlich models for the studied adsorption isotherm. The results showed that the adsorption isotherm was consistently fitted by the Langmuir model. Further, the non-linear method was found to be more suitable for obtaining the isotherm parameters.







Similar content being viewed by others
References
Yoon, S., Kim, S., Cho, C.-W., Yun, Y.-S.: The preparation of modified industrial waste polyacrylonitrile for the adsorptive recovery of Pt(IV) from acidic solutions. Materials 9, 988 (2016)
Bacelo, H.A.M., Santos, S.C.R., Botelho, C.M.S.: Tannin-based biosorbents for environmental applications—a review. Chem. Eng. J. 303, 575–587 (2016)
Iyer, N., Ruhela, R., Das, A., Yadav, M., Singh, A.K., Chakravartty, J.K.: Novel imino diacetamide grafted styrene divinylbenzene resin for separation and recovery of palladium from simulated high level liquid waste. Sep. Sci. Technol. 51, 1971–1978 (2016)
Xu, Y., Kim, S.-Y., Ito, T., Tokuda, H., Hitomi, K., Ishi, K.: Adsorption behavior of platinum group metals onto a Silica-based (Crea+Dodec)/SiO2-P extraction resin from simulated high level liquid waste. Sep. Sci. Technol. 50, 260–266 (2015)
Gutsche, C.D.: Calixarenes. Royal Society of Chemistry, Cambridge (1989)
Dumazet-Bonnamour, I., Halouani, H., Oueslati, F., Lamartine, R.: Calixarene for metal cations extraction. C. R. Chim. 8, 881–891 (2005)
Casnati, A., Barboso, S., Rouquette, H., Schwing-Weill, M.-J., Arnaud-Neu, F., Dozol, J.-F., Ungaro, R.: New efficient Calixarene Amide ionophores for the selective removal of strontium ion from nuclear waste: synthesis, complexation, and extraction properties. J. Am. Chem. Soc. 123, 12182–12190 (2001)
Nagasaki, T., Shinkai, S.: Solvent extraction of transition metal cations by calixarene-based cylic ligands. Bull. Chem. Soc. Jpn. 45, 471–475 (1992)
Konczyk, J., Nowik-Zajac, A., Kozlowski, C.A.: Calixarene-based extractants for heavy metal ions removal from aqueous solutions. Sep. Sci. Technol. 51, 2394–2410 (2016)
Yordanov, A.T., Roundhill, D.M.: Solution extraction of transition and post-transition heavy and precious metals by chelate and macrocyclic ligands. Coord. Chem. Rev. 170, 93 (1998)
Jumina, Priastomo, Y., Setiawan, H.R., Mutmainah, S., Kurniawan, Y.S., Ohto, K.: Simultaneous removal of Lead (II), Chromium (III) and Copper (II) heavy metal ions through an adsorption process using C-phenylcalix[4]pyrogalolena Material. J. Environ. Chem. Eng. 8(4), 103971 (2020)
Jumina, S., Siswanta, D., Nofiati, K., Imawan, A.C., Priastomo, Y., Ohto, K.: Synthesis of C-4-hydroxy-3-methoxyphenylcalix[4]resorcinarene and its application as adsorbent for lead(II), copper(II) and chromium(III). Bull. Chem. Soc. Jpn. 92, 825–831 (2019)
Yordanov, A.T., Mague, J.T., Roundhill, D.M.: Solvent extraction of divalent palladium and platinum from aqueous solutions of their chloro complexes using an N, N-dimethyldithiocarbamoylethoxy substituted calix[4]arene. Inorg. Chim. Acta 240, 441–446 (1995)
Ohto, K., Yano, M., Inoue, K., Yamamoto, T., Goto, M., Nakashio, F., Shinkai, S., Nagasaki, T.: Solvent extraction of trivalent rare earth metal ions with carboxylate derivatives of calixarenes. Anal. Sci. 11, 893–902 (1995)
Kim, J.Y., Priastomo, Y., Morisada, S., Kawakita, H., Ohto, K., Kim, Y.: Synthesis of a novel dibenzylamide derivative of p-tert-Octylcalix[4]arene and its extraction properties towards noble metal ions. Aus. J. Chem. 70(5), 450–455 (2017)
Liu, Y., Zhao, B.-T., Zhang, H.-Y., Ju, H.-F., Chen, L.-X., He, X.-W.: Molecular design of Calixarene part 4 synthesis of novel Double-Armed p-(tert-Butyl)calix[4]arene-Derived amides and their lead(II)(Pb2+)sSelective-electrode properties. Helv. Chim. Acta 84, 1969–1975 (2001)
Stankovic, V., Outarra, L., Zonnevijlle, F., Comninellis, C.H.: Solvent extraction of silver from nitric acid solutions by calix[4]arene amide derivatives. Sep. Purif. Technol. 61, 366–374 (2008)
Marcos, P.M., Felix, S., Ascenso, J.R., Segurado, M.A.P., Pereira, J.L., Khazaeli-Parsa, C.P., Hubscher-Bruderd, V., Arnaud-Neud, F., Khazaeli-Parsa, P.: Complexation and transport of alkali, alkaline earth, transition and heavy metal cations by p-tert-butyldihomooxacalix[4]arene tetra(diethyl)amide. New J. Chem. 28, 748–755 (2004)
Kim, J.Y., Morisada, S., Kawakita, H., Ohto, K., Kim, Y.: Relationship between chemical structure and extraction efficiency toward palladium with ketonic derivatives of p-tertoctylcalix[4]arene in nitric acid media. J. Incl. Phenom. Macrocycl. Chem. 85, 25–32 (2015)
de Namur, A.F.D., Goitia, M.T., Casal, A.R., Villanueva-Salas, J.A.: Extraction of silver (I) by calixarene amine derivatives: the medium effect on the stoichiometry of the phase transfer extraction process. PCCP 3, 5242 (2001)
Yamada, M., Gandhi, M.R., Kondo, Y., Hamada, F.: Synthesis and characterisation of pdiethylaminomethylthiacalix[4]arene for selective recovery of platinum from automotive catalyst residue. Supramol. Chem. 26(7–8), 620–630 (2014)
Yamada, M., Gandhi, M.R., Kaneta, Y., Hu, Y., Shibayama, A.: Calix[4]arene-based n-dialkylamino extractants form selective platinum group metal separation from automotive catalysts. ChemistrySelect 2, 1052–1057 (2017)
Yamada, M., Kaneta, Y., Gandhi, M.R., Kunda, U.M.R., Shibayama, A.: Calix[4]arene-Based amino extractants containing n-Alkyl moieties for separation of Pd(II) and Pt(IV) from leach liquors of automotive catalysts. Metals 8, 517 (2018)
Chetry, A.B., Matsufuji, T., Adhikari, B.B., Morisada, S., Kawakita, H., Ohto, K., Oshima, T., Jumina, S.: Intramolecular synergism for group separation extraction of trivalent rare earths with a cross type calix[4]arene with phosphonic and carboxylic acid bifunctionality. J. Incl. Phenom. Macrocycl. Chem. 81, 301–310 (2015)
Sadamatsu, H., Morisada, S., Kawakita, H., Ohto, K.: Allosteric coextraction of sodium and metal ions with Calix[4]Arene derivatives 3. Effect of propyl groups on size-discrimination for the second coextracted ion. Solvent Extr. Ion Exch. 33, 264–277 (2015)
Halouani, H., Dumazet-Bonnamour, I., Perrin, M., Lamartine, R.: First synthesis and structure of â-Ketoimine Calix[4]arenes: complexation and extraction studies. J. Org. Chem. 69, 6521–6527 (2004)
Yamada, M., Gandhi, M.R., Sato, D., Kaneta, Y., Kimura, N.: Comparative study on palladium(II) extraction using Thioamide-Modified acyclic and cyclic extractants. Ind. Eng. Chem. Res. 55, 8914–8921 (2016)
Yamada, M., Gandhi, M.R., Shibayama, A.: Rapid and selective recovery of palladium from platinum group metals and base metals using a thioamide-modified calix[4]arene extractant in environmentally friendly hydrocarbon fluids. Sci. Rep. 8, 16909 (2018)
Ramesh, A., Hasegawa, H., Sugimoto, W., Maki, T., Ueda, K.: Adsorption of gold (III), platinum(IV) and palladium(II) onto glycine modified crosslinked chitosan resin. Bioresour. Technol. 99, 3801–3809 (2008)
Ohto, K., Tanaka, Y., Yano, M., Shinohara, T., Murakami, E., Inoue, K.: Selective adsorption of lead ion on Calix[4]arene carboxylate resin supported by Polyallylamine. Solvent Extr. Ion Exch. 19, 725–741 (2001)
Ohto, K., Inoue, S., Eguchi, N., Shinohara, T., Inoue, K.: Adsorption behavior of lead ion on calix[4]arene tetracarboxylic acid impregnated resin. Sep. Sci. Technol. 37, 1943–1958 (2002)
Shinohara, T., Wakisaka, S., Ohto, K., Inoue, K.: Synthesis of novel type resin based on Calix[4]arene carboxylate and selective separation of lead from zinc. Chem. Lett. 29, 640–641 (2000)
Ohto, K., Yamasaki, T., Wakisaka, S., Shinohara, T., Inoue, K.: Preparation of novel crosslinking type resins based on calix[4]arene tetracarboxylate with high selectivity and high loading capacity for lead ion. J. Ion Exch. 14(supplement), 301–304 (2003)
Adhikari, B.B., Gurung, M., Kawakita, H., Jumina, S., Ohto, K.: Methylene crosslinked calix[6]arene hexacaarboxylic acid resin: A highly efficient solid phase extractant for decontamination of lead bearing effluent. J. Hazard. Mater. 193, 200–208 (2011)
Adhikari, B.B., Kanemitsu, M., Kawakita, H., Jumina, S., Ohto, K.: Synthesis and application of a highly efficient polyvinylcalix[4]arene tetraacetic acid resin for adsorptive removal of lead from aqueous solutions. Chem. Eng. J. 172, 341–353 (2011)
Priastomo, Y., Morisada, S., Kawakita, H., Ohto, K., Jumina, S.: Synthesis of macrocyclic polypnenol resin by methylene crosslinked calix[4]arene for the adsorption of palladium and platinum ions. New J. Chem. 43, 8015–8023 (2019)
Nebgahe, K.C., Boundati, Y.E., Ziat, K., Naji, A., Rghioui, L., Saidi, M.: Comparison of linear and non-linear method for determination of optimum equilibrium isotherm for adsorption of copper(II) onto treated Martil sand. Fluid Phase Equilibr. 430, 188–194 (2016)
Kumar, K.V.: Optimum sorption isotherm by linear and non-linear methods for malachite green onto lemon peel. Dyes Pigments 74, 595–597 (2007)
Gutsche, C.D., Dhawan, B., No, K.H., Muthukrisnan, R.: Calixarenes. 4. The synthesis, characterization, and properties. J. Am. Chem. Soc. 103, 3782–3792 (1981)
Gutsche, C.D., Dhawan, B., Levine, J.A., No, K.H., Bauer, L.J.: Calixarenes 9: Conformational isomers of the ethers and esters of calix[4]arenes. Tetrahedron 39, 409 (1983)
Iwamoto, K., Yanagi, A., Araki, K., Shinkai, A.: Synthesis of new isomers from p-t-Butylcalix[4]arene. Strategies for regioselective alkylation on the lower rim. Chem. Lett. 16, 473–476 (1991)
Iwamoto, K., Shinkai, A.: Syntheses and ion selectivity of all conformational isomers of Tetrakis(ethoxycarbony1)methoxy )calix[41arene. J. Org. Chem. 57, 7066–7073 (1992)
Freundlich, H.: Uber die adsorption in losungen. Z. Phys. Chem. 57, 387–470 (1906)
Langmuir, I.: The adsorption of gases on plane surface of glass, mica and platinum. J. Am. Chem. Soc. 40, 1361–1368 (1918)
Tarapcik, P.: Extraction of palladium from nitrate solutions with TBP in tetrachloromethane. Radiochem. Radioanal. Lett. 49, 353–359 (1981)
Wołowicz, A., Hubicki, Z.: Selective adsorption of palladium(II) complexes onto the chelating ion exchange Resin Dowex M4195—kinetic studies. Solvent Extr. Ion. Exch. 28, 124–159 (2010)
Spieker, W.A., Liu, J., Miller, J.T., Kropf, A.J., Regalbuto, J.R.: An EXAFS study of the co-ordination chemistry of hydrogen hexachloroplatinate(IV) 1. Speciation in aqueous solution. Appl. Catal. A. 232, 219–235 (2002)
Vasilchenko, D., Tkachev, S., Baidina, I., Korenev, S.: Speciation of platinum(IV) in nitric acid solutions. Inorg. Chem. 52, 10532–10541 (2013)
Wang, R., Liao, X., Shi, B.: Adsorption behaviors of Pt(II) and Pd(II) on collagen fiber immobilized bayberry tannin. Ind. Eng. Chem. Res. 44, 4221–4226 (2005)
Uheida, A., Iglesias, M., Fontas, C., Hidalgo, M., Salvado, V., Zhang, Y., Muhammed, M.: Sorption of palladium(II), rhodium(III), and platinum(IV) on Fe3O4 nanoparticles. J. Colloid Interf. Sci. 301, 402–408 (2006)
Parajuli, D., Kawakita, H., Inoue, K., Funaoka, M.: Recovery of gold(III), palladium(II), and platinum(IV) by aminated lignin derivatives. Ind. Eng. Chem. Res. 45, 6405–6412 (2006)
Park, C., Chung, J.S., Cha, K.W.: Separation and preconcentration method for palladium, platinum and gold from some heavy metals using Amberlite IRC 718 chelating Resin. Bull. Korean Chem. Soc. 21, 121–124 (2000)
Jermakowicz-Bartkowiak, D., Kolarz, B.N., Serwin, A.: Sorption of precious metals from acid solutions by functionalised vinylbenzyl chloride–acrylonitryle–divinylbenzene copolymers bearing amino and guanidine ligands. React. Funct. Polym. 65, 135–142 (2005)
Fujiwara, K., Ramesh, A., Maki, T., Hasegawa, H., Ueda, K.: Adsorption of platinum (IV), palladium (II) and gold (III) from aqueous solutions onto l-lysine modified crosslinked chitosan resin. J. Hazard. Mater. 146, 39–50 (2007)
Hall, K.R., Eagleton, L.C., Acrivos, A., Vermeulen, T.: Pore and solid diffusion kinetics fixed bed adsorption under constant pattern conditions. Ind. Eng. Chem. Fundam. 5, 212–223 (1966)
Acknowledgements
This research was financially supported in part by JSPS KAKENHI Grant Number (Grant-in-Aid for “Scientific Research (C) 19K12399) from JSPS ” and the authors deeply thank to Monbukagakusho Scholarship (MEXT) for providing a master degree fellowship to Yoga Priastomo (2015-2017) through Post-Graduated Program for Global Advancement (PPGA) in Environmental and Energy Science initiated by the Ministry of Education, Culture, Sports, Science and Technology of Japan. We would like to thank Editage (http://www.editage.com/ ) for English language editing.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Priastomo, Y., Morisada, S., Kawakita, H. et al. Improved precious metal adsorption by introduction of carboxylic acid groups on methylene crosslinked calix[4]arene resin matrix. J Incl Phenom Macrocycl Chem 101, 51–61 (2021). https://doi.org/10.1007/s10847-021-01079-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-021-01079-1