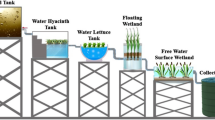
Abstract
The green seaweeds Derbesia tenuissima and Ulva ohnoi were assessed comparatively for yields of biomass and bioproducts (fatty acids, soluble fibres and amino acids) under controlled land-based culture over 6 months. The intensive cultivation of these seaweeds yielded an average biomass productivity of 15 g dry weight (dw) m−2 day−1 (56 t dw ha−1 year−1) for D. tenuissima and 38 g dw m−2 day−1 (138 t dw ha−1 year−1) for U. ohnoi. The production of D. tenuissima was comparatively consistent, ranging between 8 and 20 g dw m−2 day−1, while that of U. ohnoi was highly variable and stochastic, ranging between 16 and 77 g dw m−2 day−1. The major bioproducts were lipids (13 % dw) and fatty acids (5 % dw) for D. tenuissima and soluble fibres (ulvan, 12 % dw) for U. ohnoi. These concentrations were consistent over time, irrespective of the variation in environmental conditions and biomass productivity. In addition, D. tenuissima and U. ohnoi are potential bioresources for the extraction of proteins (amino acids). The amino acid content of D. tenuissima (24 % dw) was higher than that of U. ohnoi (13 % dw). However, the annual amino acid productivity of U. ohnoi (18 t ha−1 year−1) was higher than that of D. tenuissima (14 t ha−1 year−1) due to the higher annual productivity of biomass. Notably, both species offer niche opportunities to deliver multiple products through a biorefinery process.




Similar content being viewed by others
References
Adams JM, Ross AB, Anastasakis K, Hodgson EM, Gallagher JA, Jones JM, Donnison IS (2011) Seasonal variation in the chemical composition of the bioenergy feedstock Laminaria digitata for thermochemical conversion. Bioresour Technol 102:226–234
Ainsworth EA, Yendrek CR, Skoneczka JA, Long SP (2012) Accelerating yield potential in soybean: potential targets for biotechnological improvement. Plant Cell Environ 35:38–52
Angell AR, Mata L, de Nys R, Paul NA (2014) Variation in amino acid content and its relationship to nitrogen content and growth rate in Ulva ohnoi (Chlorophyta). J Phycol 50:216–226
Bidwell RGS, Mclachlan J, Lloyd NDH (1985) Tank cultivation of Irish moss, Chondrus crispus Stackh. Bot Mar 28:87–97
Boland MJ, Rae AN, Vereijken JM, Meuwissen MPM, Fischer ARH, van Boekel MAJS, Rutherfurd SM, Gruppen H, Moughan PJ, Hendriks WH (2013) The future supply of animal-derived protein for human consumption. Trends Food Sci Technol 29:62–73
Bolton JJ, Robertson-Andersson DV, Shuuluka D, Kandjengo L (2009) Growing Ulva (Chlorophyta) in integrated systems as a commercial crop for abalone feed in South Africa: a SWOT analysis. J Appl Phycol 21:575–583
Bruhn A, Dahl J, Nielsen HB, Nikolaisen L, Rasmussen MB, Markager S, Olesen B, Arias C, Jensen PD (2011) Bioenergy potential of Ulva lactuca: biomass yield, methane production and combustion. Bioresour Technol 102:2595–2604
Buschmann AH, Mora OA, Gómez P, Böttger M, Buitano S, Retamales C, Vergara PA, Gutierrez A (1994) Gracilaria chilensis outdoor tank cultivation in Chile: use of land-based salmon culture effluents. Aquacult Eng 13:283–300
Capo TR, Jaramillo JC, Boyd AE, Lapointe BE, Serafy JE (1999) Sustained high yields of Gracilaria (Rhodophyta) grown in intensive large-scale culture. J Appl Phycol 11:143–147
Castelar B, Reis R, Santos Calheiros A (2014) Ulva lactucaand U. flexuosa (Chlorophyta, Ulvophyceae) cultivation in Brazilian tropical waters: recruitment, growth, and ulvan yield. J Appl Phyco l26:1989–1999
Chiellini F, Morelli A (2011) Ulvan: a versatile platform of biomaterials from renewable resources. In: Pignatello R (ed) Biomaterials—physics and chemistry. InTech, Rijieka, pp 75–98
Del Campo E, García-Reina G, Correa JA (1998) Degradative disease in Ulva rigida (Chlorophyceae) associated with Acrochaete geniculata (Chlorophyceae). J Phycol 34:160
Domozych D, Ciancia M, Fangel JU, Mikkelsen MD, Ulvskov P, Willats WGT (2012) The cell walls of green algae: a journey through evolution and diversity. Front Plant Sci 3. doi:10.3389/fpls.2012.00082
Dubois V, Breton S, Linder M, Fanni J, Parmentier M (2007) Fatty acid profiles of 80 vegetable oils with regard to their nutritional potential. Eur J Lipid Sci Technol 109:710–732
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Friedlander M, Galai N, Farbstein H (1990) A model of seaweed growth in an outdoor culture in Israel. Hydrobiologia 204(205):367–373
Gellenbeck KW (2012) Utilization of algal materials for nutraceutical and cosmeceutical applications—what do manufacturers need to know? J Appl Phycol 24:309–313
Gosch B, Paul N, de Nys R, Magnusson M (2015) Seasonal and within-plant variation in fatty acid content and composition in the brown seaweed Spatoglossum macrodontum (Dictyotales, Phaeophyceae). J Appl Phycol 27:387–398
Gosch BJ, Magnusson M, Paul NA, de Nys R (2012) Total lipid and fatty acid composition of seaweeds for the selection of species for oil-based biofuel and bioproducts. GCB Bioenergy 4:919–930
Grieshop CM, Fahey GC (2001) Comparison of quality characteristics of soybeans from Brazil, China, and the United States. J Agric Food Chem 49:2669–2673
Grobbelaar JU (2009) Upper limits of photosynthetic productivity and problems of scaling. J Appl Phycol 21:519–522
Grobbelaar JU (2010) Microalgal biomass production: challenges and realities. Photosynth Res 106:135–144
Guil-Guerrero JL (2007) Stearidonic acid (18:4n-3): metabolism, nutritional importance, medical uses and natural sources. Eur J Lipid Sci Technol 109:1226–1236
Hafting J, Critchley A, Cornish ML, Hubley S, Archibald A (2012) On-land cultivation of functional seaweed products for human usage. J Appl Phycol 24:385–392
Huguenin JE (1976) An examination of problems and potentials for future large-scale intensive seaweed culture systems. Aquaculture 9:313–342
Ito K, Hori K (1989) Seaweed: chemical composition and potential food uses. Food Rev Int 5:101–144
Jaeger W, Siegel R (2008) Economics of oilseed crops and their biodiesel potential in Oregon’s Willamette Valley. Oregon State University, Department of Agricultural and Resource Economics
Kelly ME, Dworjanyn S (2008) The potential of marine biomass for anaerobic biogas production: a feasibility study with recommendations for further research. Crown Estate 103
Lahaye M, Gomez-Pinchetti J-L, del Rio MJ, Garcia-Reina G (1995) Natural decoloration, composition and increase in dietary fibre content of an edible marine algae, Ulva rigida (Chlorophyta), grown under different nitrogen conditions. J Sci Food Agric 68:99–104
Lahaye M, Jegou D (1993) Chemical and physical–chemical characteristics of dietary fibres from Ulva lactuca (L.) Thuret and Enteromorpha compressa (L.) Grev. J Appl Phycol 5:195–200
Lahaye M, Robic A (2007) Structure and functional properties of ulvan, a polysaccharide from green seaweeds. Biomacromolecules 8:1765–1774
Lapointe BE, Williams LD, Goldman JC, Ryther JH (1976) The mass outdoor culture of macroscopic marine algae. Aquaculture 8:9–21
Lawton RJ, Mata L, de Nys R, Paul NA (2013) Algal bioremediation of waste waters from land-based aquaculture using Ulva: selecting target species and strains. PLoS One 8(10):e77344
Li X, Rezaei R, Li P, Wu G (2011) Composition of amino acids in feed ingredients for animal diets. Amino Acids 40:1159–1168
Lobban CS, Harrison PJ (1996) Light and photosynthesis. In: Seaweed ecology and physiology. Cambridge University Press, Cambridge, pp 146–150
Magnusson M, Mata L, Nys R, Paul N (2014) Biomass, lipid and fatty acid production in large-scale cultures of the marine macroalga Derbesia tenuissima (Chlorophyta). Mar Biotech 16:456–464
Mata L, Schuenhoff A, Santos R (2010) A direct comparison of the performance of the seaweed biofilters, Asparagopsis armata and Ulva rigida. J Appl Phycol 22:639–644
McKendry P (2002) Energy production from biomass (part 1): overview of biomass. Bioresour Technol 83:37–46
Neveux N, Magnusson M, Maschmeyer T, de Nys R, Paul NA (2014) Comparing the potential production and value of high-energy liquid fuels and protein from marine and freshwater macroalgae. GCB Bioenergy:1–17. doi:10.1111/gcbb.12171
Pereira R, Yarish C, Critchley A (2013) Seaweed aquaculture for human foods in land-based and IMTA systems. In: Christou P, Savin R, Costa-Pierce B, Misztal I, Whitelaw CB (eds) Sustainable food production. Springer, New York, pp 1405–1424
Poudyal H, Panchal SK, Waanders J, Ward L, Brown L (2012) Lipid redistribution by α-linolenic acid-rich chia seed inhibits stearoyl-CoA desaturase-1 and induces cardiac and hepatic protection in diet-induced obese rats. J Nutr Biochem 23:153–162
Poudyal H, Panchal SK, Ward LC, Brown L (2013) Effects of ALA, EPA and DHA in high-carbohydrate, high-fat diet-induced metabolic syndrome in rats. J Nutr Biochem 24:1041–1052
Robertson-Andersson DV, Potgieter M, Hansen J, Bolton JJ, Troell M, Anderson RJ, Halling C, Probyn T (2008) Integrated seaweed cultivation on an abalone farm in South Africa. J Appl Phycol 20:579–595
Robic A, Sassi J-F, Dion P, Lerat Y, Lahaye M (2009) Seasonal variability of physicochemical and rheological properties of ulvan in two Ulva species (Chlorophyta) from the Brittany coast. J Phycol 45:962–973
Russo GL (2009) Dietary n-6 and n-3 polyunsaturated fatty acids: from biochemistry to clinical implications in cardiovascular prevention. Biochem Pharmacol 77:937–946
Ryther JH, Goldman JC, Gifford CE, Huguenin JE, Wing AS, Clarner JP, Williams LD, Lapointe BE (1975) Physical models of integrated waste recycling-marine polyculture systems. Aquaculture 5:163–177
Ryther JH, Debusk TA, Blakeslee M (1984) Cultivation and conversion of marine macroalgae (Gracilaria and Ulva). SERI/STR-231-2360, pp. 1–88
Saravanan P, Davidson NC, Schmidt EB, Calder PC (2010) Cardiovascular effects of marine omega-3 fatty acids. Lancet 376:540–550
Schmid M, Guihéneuf F, Stengel D (2014) Fatty acid contents and profiles of 16 macroalgae collected from the Irish Coast at two seasons. J Appl Phycol 26:451–463
Shuuluka D, Bolton J, Anderson R (2013) Protein content, amino acid composition and nitrogen-to-protein conversion factors of Ulva rigida and Ulva capensis from natural populations and Ulva lactuca from an aquaculture system, in South Africa. J Appl Phycol 25:677–685
Acknowledgments
We thank Ana Wegner, Elien Boogaerts, Sophie Raillardand Scott Seymour for assistance with the management, maintenance and data collection of the cultivation system. We also thank Veronique Mocellin and Kerri-Lee Dyer for assistance with the biochemical analysis. This research is part of the MBD Energy Research and Development programme for Biological Carbon Capture and Storage. This project is supported by the Advanced Manufacturing Cooperative Research Centre (AMCRC), funded through the Australian Government’s Cooperative Research Centre Scheme, and the Australian Renewable Energy Agency (ARENA).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 421 kb)
Rights and permissions
About this article
Cite this article
Mata, L., Magnusson, M., Paul, N.A. et al. The intensive land-based production of the green seaweeds Derbesia tenuissima and Ulva ohnoi: biomass and bioproducts. J Appl Phycol 28, 365–375 (2016). https://doi.org/10.1007/s10811-015-0561-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0561-1