Abstract
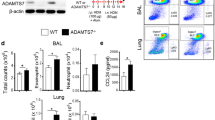
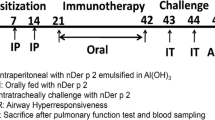
House dust mite (HDM) acts as an environmental antigen that might cause chronic allergic diseases. Neferine (NEF) shows anti-inflammation therapeutic effects. This study is to explore the protection role of NEF against HDM-induced allergic inflammation. HDM-induced allergic asthmatic C57BL/6J mice models were established. Differential histological staining was used to analyze lung tissue pathological scores. Flow cytometry was used to analyze subtypes and biomarker expression of immune cells. RT-PCR and ELISA were used to test cytokines-related gene and/or protein expression levels. Western blot was performed to investigate the signaling pathway that mediates allergic inflammation from mice lung tissue and bone marrow–derived dendritic cells (BMDCs). H&E and PAS staining results indicate NEF significantly attenuated inflammatory index and the percentage of goblet cells in the lung tissue induced by HDM. The HDM-elevated TH2 and TH17 cells were significantly decreased by NEF; inflammatory cytokines Il-4, Il-13 and Il-17 were dramatically downregulated in the NEF plus HDM group compared with HDM alone. CD40+ and CD86+ DCs, eosinophils and mast cells, and ILC2 cells were decreased by NEF which was elevated under HDM stimulation. In vivo and ex vivo investigations indicated NEF can attenuate the activated NF-κB signaling induced by HDM is involved in allergic inflammatory immune response and regulates cytokines-related gene expression. HDM-activated DCs promoted differentiation of TH2 and TH17 cells but were attenuated by NEF. This study suggests NEF interrupts the overexpression of some cytokines released by DCs, TH2, and TH17 cells; NEF attenuates HDM-induced allergic inflammation via inhibiting NF-κB signaling of DCs.







Similar content being viewed by others
Availability of Data and Materials
The data that support this study are available from the corresponding authors upon reasonable request.
Abbreviations
- APCs:
-
Antigen-presenting cells
- CD:
-
Cluster of differentiation
- DC:
-
Dendritic cell
- HDM:
-
House dust mite
- IgE:
-
Immunoglobulin E
- IL:
-
Interleukin
- ILC2:
-
Group 2 innate lymphoid cells
- i.p.:
-
Intraperitoneal injection
- i.n.:
-
Intranasal
- NEF:
-
Neferine
- NF-κB:
-
Nuclear factor-kappa B
- MHC-II:
-
Major histocompatibility complex class II
- MLN:
-
Mediastinal lymph node
- OX40L:
-
Ligand for OX40 or CD134
- TH:
-
T helper cell
- TNF-α:
-
Tumor necrosis factor α
References
Galli, S.J., M. Tsai, and A.M. Piliponsky. 2008. The development of allergic inflammation. Nature 454 (7203): 445–454.
Hu, H., et al. 2022. Mitochondrial VDAC1: A potential therapeutic target of inflammation-related diseases and clinical opportunities. Cells 11(19).
Miki, H., et al. 2020. Allergin-1 immunoreceptor suppresses house dust mite-induced allergic airway inflammation. The Journal of Immunology 204 (4): 753–762.
Kay, A.B., 2001 Allergy and allergic diseases. First of two parts. The New England Journal of Medicine 344(1): p. 30–7.
Joller, N., et al. 2014. Treg cells expressing the coinhibitory molecule TIGIT selectively inhibit proinflammatory Th1 and Th17 cell responses. Immunity 40 (4): 569–581.
Yao, C., et al. 2009. Prostaglandin E2-EP4 signaling promotes immune inflammation through Th1 cell differentiation and Th17 cell expansion. Nature Medicine 15 (6): 633–640.
Wu, Y., et al. 2020. Induction of ferroptosis-like cell death of eosinophils exerts synergistic effects with glucocorticoids in allergic airway inflammation. Thorax 75 (11): 918–927.
Iype, J. and M. Fux. 2021. Basophils orchestrating eosinophils’ chemotaxis and function in allergic inflammation. Cells 2021. 10(4).
Patel, D.F., et al. 2019. Neutrophils restrain allergic airway inflammation by limiting ILC2 function and monocyte-dendritic cell antigen presentation. Science Immunology 4(41).
Radermecker, C., et al. 2019. Locally instructed CXCR4(hi) neutrophils trigger environment-driven allergic asthma through the release of neutrophil extracellular traps. Nature Immunology 20 (11): 1444–1455.
Lambrecht, B.N., and H. Hammad. 2010. The role of dendritic and epithelial cells as master regulators of allergic airway inflammation. Lancet 376 (9743): 835–843.
Soumelis, V., et al. 2002. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nature Immunology 3 (7): 673–680.
Hsu, C.L., et al. 2020. Allergic inflammation is initiated by IL-33-dependent crosstalk between mast cells and basophils. PLoS ONE 15 (1): e0226701.
McNeil, B.D., et al. 2015. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 519 (7542): 237–241.
Siracusa, M.C., et al. 2013. Basophils and allergic inflammation. Journal of Allergy and Clinical Immunology 132(4): p. 789–801; quiz 788.
Fu, L., et al. 2022. A mitochondrial STAT3-methionine metabolism axis promotes ILC2-driven allergic lung inflammation. The Journal of Allergy and Clinical Immunology 149 (6): 2091–2104.
Wallrapp, A., et al. 2017. The neuropeptide NMU amplifies ILC2-driven allergic lung inflammation. Nature 549 (7672): 351–356.
Dougan, M., G. Dranoff, and S.K. Dougan. 2019. GM-CSF, IL-3, and IL-5 family of cytokines: Regulators of inflammation. Immunity 50 (4): 796–811.
Schröder, A., et al. 2022. IL-37 regulates allergic inflammation by counterbalancing pro-inflammatory IL-1 and IL-33. Allergy 77 (3): 856–869.
Del Prete, G., M. Ricci, and S. Romagnani. 1991. T cells, IgE antibodies, cytokines and allergic inflammation. Allergy Immunology (Paris) 23 (6): 239–243.
Pelaia, G., et al. 2015. Cellular mechanisms underlying eosinophilic and neutrophilic airway inflammation in asthma. Mediators of Inflammation 2015: 879783.
Izumi, G., et al. 2021. CD11b(+) lung dendritic cells at different stages of maturation induce Th17 or Th2 differentiation. Nature Communications 12 (1): 5029.
Shao, Z., et al. 2009. Fms-like tyrosine kinase 3 ligand increases a lung DC subset with regulatory properties in allergic airway inflammation. The Journal of Allergy and Clinical Immunology 123 (4): 917-924.e2.
Hwang, Y.H., S.J. Kim, and S.T. Yee. 2020. Physcion-matured dendritic cells induce the differentiation of Th1 cells. International Journal of Molecular Sciences 21(5).
Besnard, A.G., et al. 2011. IL-33-activated dendritic cells are critical for allergic airway inflammation. European Journal of Immunology 41 (6): 1675–1686.
Marthandam Asokan, S., et al. 2018. Pharmacological benefits of neferine - a comprehensive review. Life Sciences 199: 60–70.
Chiu, K.M., et al. 2021. Anti-allergic and anti-inflammatory effects of neferine on RBL-2H3 cells. International Journal of Molecular Science 22(20).
Dasari, S., et al. 2020. Neferine, an alkaloid from lotus seed embryo targets HeLa and SiHa cervical cancer cells via pro-oxidant anticancer mechanism. Phytotherapy Research 34 (9): 2366–2384.
Deng, J., et al. 2023. House dust mite-induced endoplasmic reticulum stress mediates MUC5AC hypersecretion via TBK1 in airway epithelium. Experimental Lung Research p. 1–14.
Zhang, Y., et al. 2020. Resveratrol decreases cell apoptosis through inhibiting DNA damage in bronchial epithelial cells. International Journal of Molecular Medicine 45 (6): 1673–1684.
Guo, L., et al. 2022. A newly discovered PD-L1 B-cell epitope peptide vaccine (PDL1-Vaxx) exhibits potent immune responses and effective anti-tumor immunity in multiple syngeneic mice models and (synergizes) in combination with a dual HER-2 B-cell vaccine (B-Vaxx). Oncoimmunology 11 (1): 2127691.
**ong, A., et al. 2016. Ornithine decarboxylase antizyme 1 upregulate LOR to promote differentiation of SCC 15 cells by binding CBP / p 300 in promoter region. International Journal of Clinical and Experimental Medicine 9 (2): 2359–2366.
Cosmi, L., et al. 2011. Th17 cells: New players in asthma pathogenesis. Allergy 66 (8): 989–998.
Toussaint, M., et al. 2017. Host DNA released by NETosis promotes rhinovirus-induced type-2 allergic asthma exacerbation. Nature Medicine 23 (6): 681–691.
Plantinga, M., et al. 2013. Conventional and monocyte-derived CD11b(+) dendritic cells initiate and maintain T helper 2 cell-mediated immunity to house dust mite allergen. Immunity 38 (2): 322–335.
Mendez-Enriquez, E., et al. 2021. Mast cell-derived serotonin enhances methacholine-induced airway hyperresponsiveness in house dust mite-induced experimental asthma. Allergy 76 (7): 2057–2069.
Maun, H.R., et al. 2019. An allosteric anti-tryptase antibody for the treatment of mast cell-mediated severe asthma. Cell 179 (2): 417-431.e19.
Maggi, L., et al. 2021. The dual function of ILC2: From host protection to pathogenic players in type 2 asthma. Molecular Aspects of Medicine 80: 100981.
Pivniouk, V., et al. 2022. Airway administration of OM-85, a bacterial lysate, blocks experimental asthma by targeting dendritic cells and the epithelium/IL-33/ILC2 axis. The Journal of Allergy and Clinical Immunology 149 (3): 943–956.
Rank, M.A., et al. 2009. IL-33-activated dendritic cells induce an atypical TH2-type response. The Journal of Allergy and Clinical Immunology 123 (5): 1047–1054.
Tchekneva, E.E., et al. 2019. Determinant roles of dendritic cell-expressed notch delta-like and jagged ligands on anti-tumor T cell immunity. Journal for Immunotherapy of Cancer 7 (1): 95.
Parola, C., et al. 2013. Selective activation of human dendritic cells by OM-85 through a NF-kB and MAPK dependent pathway. PLoS ONE 8 (12): e82867.
Arimilli, S., et al. 2007. TLR-4 and -6 agonists reverse apoptosis and promote maturation of simian virus 5-infected human dendritic cells through NFkB-dependent pathways. Virology 365 (1): 144–156.
Barnes, P.J. 2011. Pathophysiology of allergic inflammation. Immunological Reviews 242 (1): 31–50.
Lu, Y., et al. 2021. Eosinophil extracellular traps drive asthma progression through neuro-immune signals. Nature Cell Biology 23 (10): 1060–1072.
Laanesoo, A., et al. 2021. Dual role of the miR-146 family in rhinovirus-induced airway inflammation and allergic asthma exacerbation. Clinical and Translational Medicine 11 (6): e427.
Iwaszko, M., S. Biały, and K. Bogunia-Kubik. 2021. Significance of interleukin (IL)-4 and IL-13 in inflammatory arthritis. Cells 10(11).
Sehra, S., et al. 2010. IL-4 regulates skin homeostasis and the predisposition toward allergic skin inflammation. The Journal of Immunology 184 (6): 3186–3190.
Foray, A.P., et al. 2020. IL-4 and IL-17 are required for house dust mite-driven airway hyperresponsiveness in autoimmune diabetes-prone non-obese diabetic mice. Frontiers in Immunology 11: 595003.
Mark, D.A., et al. 2000. B7–1 (CD80) and B7–2 (CD86) have complementary roles in mediating allergic pulmonary inflammation and airway hyperresponsiveness. American Journal of Respiratory Cell and Molecular Biology 22 (3): 265–271.
Miura, K., et al. 2020. Role of CD4(+) T cells in allergic airway diseases: Learning from murine models. Internation Journal of Molecular Science 21(20).
Hamey, F.K., et al. 2021. Single-cell molecular profiling provides a high-resolution map of basophil and mast cell development. Allergy 76 (6): 1731–1742.
Toki, S., et al. 2020. TSLP and IL-33 reciprocally promote each other’s lung protein expression and ILC2 receptor expression to enhance innate type-2 airway inflammation. Allergy 75 (7): 1606–1617.
Cayrol, C., and J.P. Girard. 2022. Interleukin-33 (IL-33): A critical review of its biology and the mechanisms involved in its release as a potent extracellular cytokine. Cytokine 156: 155891.
Funding
This work was supported by the National Natural Science Foundation of China (no. 81900028 to Yun Zhang, no. 82100021 to Hongmei Tang), the Natural Science Foundation of Sichuan Province (no. 2022NSFSC1306 to Yun Zhang), the Foundation of Luzhou Science and Technology Program and Southwest Medical University (no. 2020LZXNYDJ24 to **ng Wang, no. 2021LZXNYD-J19 to Yun Zhang), and the Sichuan Science and Technology Program (no. 2022YFS0629 to Gang Qin).
Author information
Authors and Affiliations
Contributions
X.W., Y.Z., and L.L.G conceived and designed the research. L.L.G and X.W. drafted the manuscript. W.Q., Z.L.Z., Y.R.H., H.M.T., H.H., X.F.Y., J.D., G.Q., and X.W. performed the experiments. W.Q., Y.Z., and X.W. analyzed the data. L.L.G and X.W. edited the article. All authors read and approved the final version of the article. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
All animal experiments (including euthanasia) were in compliance with the regulations and guidelines of the Southwest Medical University Institutional Animal Care Committee (approval no. 201903-117) and were conducted according to the AAALAC and IACUC guidelines.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Q., Guo, L., Zeng, Z. et al. Neferine Attenuates HDM-Induced Allergic Inflammation by Inhibiting the Activation of Dendritic Cell. Inflammation 46, 2433–2448 (2023). https://doi.org/10.1007/s10753-023-01891-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01891-6