Abstract
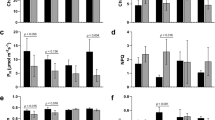
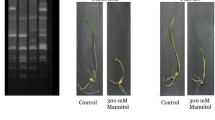
Sorghum is an important cereal in many parts of the world, but water stress is a major constraint to its yield. In response to water stress, plants can modify gene expression, leading to a variety of changes in biochemical and physiological pathways. In the present study, 32 accessions of sorghum were screened for drought tolerance at the seedling stage based on morphometric traits. After 14 days of water stress, the most-tolerant (NGB01435) and susceptible (AKZ-NrSOR1013) accessions were assessed for drought-related gene expression research. The result of the morphometric attributes showed variation in seedling growth among the accessions. Also, significant correlations exist among the evaluated attributes, and the clustering pattern revealed genetic diversity in the accessions. The analysis of Ct values across the samples revealed differential expression of the reference gene (ELF4α) and the target genes (SNAC1 and DREB1A). SNAC1 is a stress-responsive NAC superfamily transcription factor, while DREB1A is a dehydration-responsive element binding protein 1 (DREB1)/C-repeat-binding factor (CBF), both of which are involved in stress regulation in plants. Both target genes had lower expression under stress in the tolerant accession (NGB01435) and were upregulated in the susceptible accession (AKZ-NrSOR1013). This study concluded that accessions respond differentially to water stress in terms of growth and development; the downregulation of both SNAC1 and DREB1A is critical to the mechanism involved in the water stress modulation to confer resistance in sorghum seedlings under water deficit conditions.





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available with the corresponding author.
References
Abreha KB, Enyew M, Carlsson AS, Vetukuri RR, Feyissa T, Motlhaodi T, Nguni D, Geleta M (2022) Sorghum in dryland: morphological, physiological, and molecular responses of sorghum under drought stress. Planta 255:20. https://doi.org/10.1007/s00425-021-03799-7
Adeniyi A (2020) Trends analysis of temperature and humidity in Kwara State Nigeria. J Enviro Geog 13(3–4):44–50. https://doi.org/10.2478/jengeo-2020-0011
Ajadi BS, Adeniyi A, Afolabi MT (2011) Impact of climate on urban agriculture: case study of Ilorin City Nigeria. Global J Hum Soc Sci 11(1):24–30
Ajiboye TO, Iliasu GA, Adeleye AO, Abdussalam FA, Akinpelu SA, Ogunbode SM, Jimoh SO, Oloyede OB (2014) Nutritional and antioxidant dispositions of sorghum/millet-based beverages indigenous to Nigeria. Food Sci Nutr 2(5):597–604. https://doi.org/10.1002/fsn3.140
Ajum SA, **e XY, Wang LC, Saleem MF, Man C, Lei W (2011) Morphological, physiological, and biochemical responses of plants to drought stress. Afri J Agric Res 6(9):2026–2032. https://doi.org/10.5897/AJAR10.027
Akatwijuka R, Rubaihayo PR, Odong TL (2016) Genetic diversity among sorghum landraces of south-western highlands of Uganda. Afri Crop Sci J 24(2):179–190. https://doi.org/10.4314/acsj.v24i2.6
Ali N, Anjum MM (2016) Drought stress: major cause of low yield and productivity. Austin Environ Sci 1:10–12
Amelework B, Shimelis H, Tongoona P, Laing M (2015) Physiological mechanisms of drought tolerance in sorghum, genetic basis and breeding methods: a review. Afri J Agric Res 10(31):3029–3040. https://doi.org/10.5897/AJAR2015.9595
Animasaun DA, Morakinyo JA, Krishnamurthy R, Mustapha OT (2017) Genetic divergence of Nigerian and Indian pearl millet accessions based on agronomical and morphological traits. J Agric Sci 62(2):115–131. https://doi.org/10.2298/JAS1702115A
Animasaun D, Adikwu V, Alex G, Akinsunlola T, Adekola O, Krishnamurthy R (2021) Morpho-agronomic traits variability, allelic polymorphism and diversity analysis of African yam bean: Towards improving utilization and germplasm conservation. Plant Genet Resour 19(3):216–228. https://doi.org/10.1017/S1479262121000253
Ashok-Kumar A, Reddy BVS, Sharma HC, Hash CT, Srinivasa-Rao P, Ramaiah B, Reddy PS (2011) Recent advances in sorghum genetic enhancement research at ICRISAT. Am J Plant Sci 2(4):589–600. https://doi.org/10.4236/ajps.2011.24070
Bhusal N, Pahuja SK, Vats AK, Srivastava A, Kumar RS (2017) Morphological characterization of forage sorghum genotypes for its various DUS traits. J Appl Natur Sci 9(2):912–919. https://doi.org/10.31018/jans.v9i2.1297
Blum A (2005) Drought resistance, water-use efficiency, and yield potential—are they compatible, dissonant, or mutually exclusive? Austr J Agric Res 56:1159–1168. https://doi.org/10.1071/AR05069
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant response to drought-from genes to the whole plant. Funct Plant Biol 30:239–264. https://doi.org/10.1071/FP02076
Chen X, Wu Q, Gao Y, Zhang J, Wang Y, Zhang R, Zhou Y, **ao M, Xu W, Huang R (2020) The role of deep roots in sorghum yield production under drought conditions. Agron 10:611. https://doi.org/10.3390/agronomy10040611
Dalton TJ, Zereyesus YA (2013) Economic impact assessment of sorghum, millet and other grains CRSP: Sorghum and millet germplasm development research. INTSORMIL Scientific Publications. 20. https://digitalcommons.unl.edu/intsormilpubs/20
de Morais Cardoso L, Pinheiro SS, Martino HS, Pinheiro-Sant’Ana HM (2017) Sorghum (Sorghum bicolor L.): nutrients, bioactive compounds, and potential impact on human health. Crit Rev Food Sci Nutr 57(2):372–390. https://doi.org/10.1080/10408398.2014.887057
de Souza AA, de Carvalho AJ, Bastos EA, Cardoso MJ, Julio MPM, Batista PSC, Julio BHM, Campolina CV, Portugal AF, de Menezes CB, de Oliveira SM (2021) Grain sorghum under pre- and post-flowering drought stress in a semiarid environment. Austr J Crop Sci 15(8): 1139–1145. https://doi.org/10.3316/informit.180231063416886
Derese SA, Shimelis H, Laing M, Mengistu F (2017) The impact of drought on sorghum production, and farmer’s varietal and trait preferences, in the north eastern Ethiopia: implication for breeding. Acta Agric Scandi Sect B Soil Plant Sci 68(5):424–436. https://doi.org/10.1080/09064710.2017.1418018
Djanaguiraman M, Narayanan S, Erdayani E, Prasad PVV (2020) Effects of high temperature stress during anthesis and grain filling periods on photosynthesis, lipids and grain yield in wheat. BMC Plant Biol 20:268. https://doi.org/10.1186/s12870-020-02479-0
Dube SP, Marais D, Mavengahama S, Van-Jaarsveld CM, Gerrano AS (2019) Characterisation of agro-morphological traits of Corchorus accessions. Acta Agric Scandin Sect B Soil Plant Sci 69(2):126–134. https://doi.org/10.1080/09064710.2018.1514419
Dugas DV, Monaco MK, Olsen A, Klein RR, Kumari S, Ware D, Klein PE (2011) Functional annotation of the transcriptome of Sorghum bicolor in response to osmotic stress and abscisic acid. BMC Genomics 12:514. https://doi.org/10.1186/1471-2164-12-514
Duncan DB (1955) Multiple range and multiple F tests. Biometrics 11(1):1–42. https://doi.org/10.2307/3001478
Dykes L, Rooney WL, Rooney LW (2013) Evaluation of phenolics and antioxidant activity of black sorghum hybrids. J Cereal Sci 58(2):278–283. https://doi.org/10.1016/j.jcs.2013.06.006
Eggen M, Ozdogan M, Zaitchick B, Ademe D, Foltz J, Simane B (2019) Vulnerability of sorghum production to extreme, sub-seasonal weather under climate change. Environ Res Lett 14:045005. https://doi.org/10.1088/1748-9326/aafe19
Fahad S, Bajwa AA, Nazir U, Anjum SA, Farooq A, Zohaib A, Sadia S, Nasim W, Adkins S, Saud S, Ihsan MZ, Alharby H, Wu C, WangHuang DJ (2017) Crop production under drought and heat stress: plant responses and management options. Front Plant Sci 8:11–47. https://doi.org/10.3389/fpls.2017.01147
Falconer DS, Mackay TFC, Frankham R (1996) Introduction to quantitative genetics, 4th edn. Longman Group, London, p 463
FAO (2015) Statistical database, Food and Agriculture Organization of the United Nations, Rome, Italy. http://faostat3.fao.org/home/E. Accessed 1 Feb 2022
FAOSTAT (2021) World Food and Agriculture - Statistical Yearbook 2021. Rome. https://doi.org/10.4060/cb4477en
Farooq M, Wahid A, Kobayashi N (2009) Plant drought stress: effects, mechanisms, and management. Agron Sustaine Dev 29:185–212. https://doi.org/10.1051/agro:2008021
Fenty J (2004) Analysing distances. Strata J 4:1–26
Fracasso A, Trindade LM, Amaducci S (2016) Drought stress tolerance strategies revealed by RNA-Seq in two sorghum genotypes with contrasting WUE. BMC Plant Biolo 16:115. https://doi.org/10.1186/s12870-016-0800-x
Hair JF, Black WC, Babin BJ, Anderson RE, Tatham R (2006) Multivariate data analysis, 6th edn. Pearson Prentice Hall, Upper Saddle River, NJ
Hao YJ, Wei W, Song QX, Chen HW, Zhang YQ, Wang F, Zou HF, Lei G, Tian AG, Zhang WK, Ma B, Zhang JS, Chen SY (2011) Soybean NAC transcription factors promote abiotic stress tolerance and lateral root formation in transgenic plants. Plant J 68(2):302–313. https://doi.org/10.1111/j.1365-313X.2011.04687.x. (Epub 2011 Jul 26 PMID: 21707801)
Hariprasanna K, Rakshit S (2016) Economic importance of sorghum. In: Rakshit S, Wang YH (eds) The sorghum genome, a compendium of plant genomes. Springer, Cham. https://doi.org/10.1007/978-3-319-47789-3_1
Hossain MS, Islam MN, Rahman MM, Mamunur M, Mostofa MG, Khan MAR (2022) Sorghum: a prospective crop for climatic vulnerability, food and nutritional security. J Agri Food Res 8:100300. https://doi.org/10.1016/j.jafr.2022.100300
Hu HH, Dai MQ, Yao JL, **ao BZ, Li XH et al (2006) Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Pro Natl Acad Sci USA 103:12987–12992
Hu H, You J, Fang Y, Zhu X, Qi Z, **ong L (2008) Characterization of transcription factor gene SNAC2 conferring cold and salt tolerance in rice. Plant Mol Biol 67(1–2):169–181. https://doi.org/10.1007/s11103-008-9309-5
IBPGR, ICRISAT (1993) Descriptor for sorghum [Sorghum bicolor (L.) Moench]. International Board for Plant Genetic Resources, Rome, Italy; International Crops Research Institute for the Semi-Arid Tropics, Patancheru. pp 1–35
International Crops Research Institute for the Semi-Arid Tropics. Socioeconomics, Policy Division, International Crops Research Institute for the Semi-Arid Tropics, Agriculture Organization of the United Nations. Basic Foodstuffs Service, Agriculture Organization of the United Nations. Commodities, & Trade Division (1996) The world sorghum and millet economies: facts, trends and outlook. Food & Agriculture Orgnisation
Ilyas M, Nisar M, Khan N, Hazrat A, Khan AH, Hayat K, Fahad S, Khan A, Ullah A (2021) Drought tolerance strategies in plants: a mechanistic approach. J Plant Growth Regul 40:926–944. https://doi.org/10.1007/s00344-020-10174-5
Jaegglia BG, Mortlockb MY, Borrell A (2017) Bigger is not always better: reducing leaf area helps stay-green sorghum use soil water more slowly. Environ Exp Bot 138:119–129. https://doi.org/10.1016/j.envexpbot.2017.03.002
Jia XX, Li YT, Qi EF, Ma S, Hu XY, Wen GH, Wang YH, Li JW, Zhang XH, Wang HM, Wang WT (2016) Overexpression of the Arabidopsis DREB1A gene enhances potato drought-resistance. Russ J Plant Physiol 63:523–531. https://doi.org/10.1134/S1021443716040063
** X, Long Y, **ong S, Yang Z, Chen W, Hawar A, Chi X, Chen Y, Luo H, Qi J, Lu G, Dai L, Yang Y, Sun B (2021) SbNAC2 enhances abiotic stress tolerance by upregulating ROS scavenging activities and inducing stress-response genes in sorghum. Environ Exp Bot 192:104664. https://doi.org/10.1016/j.envexpbot.2021.104664
Jumrani K, Bhatia VS (2019) Identification of drought tolerant genotypes using physiological traits in soybean. Physiol Mol Biolo Plants 25(3):697–711. https://doi.org/10.1007/s12298-019-00665-5
Kamath VG, Chandrashekar A, Ra**i PS (2004) Antiradical properties of sorghum (Sorghum bicolor L. Moench) flour extracts. J Cereal Sci 40(3):283–288. https://doi.org/10.1016/j.jcs.2004.08.004
Kasuga M, Miura S, Shinozaki K, Yamaguchi-Shinozaki K (2004) A combination of the Arabidopsis DREB1A gene and stress-inducible rd29A promoter improved drought- and low-temperature stress tolerance in tobacco by gene transfer. Plant Cell Physiol 45(3):346–350. https://doi.org/10.1093/pcp/pch037
Kebede H, Subudhi PK, Rosenow DT, Nguyen HT (2001) Quantitative trait loci influencing drought tolerance in grain sorghum (Sorghum bicolor L. Moench). Theor Appl Genet 103(2–3):266–276. https://doi.org/10.1007/s001220100541
Khan A, Pan X, Najeeb U, Tan DKY, Fahad S, Zahoor R, Luo H (2018) Co** with drought: stress and adaptive mechanisms, and management through cultural and molecular alternatives in cotton as vital constituents for plant stress resilience and fitness. Biolo Res 51:47. https://doi.org/10.1186/s40659-018-0198-z
Lamptey S, Nyarko G, Falon A, Yeboah S (2014) Assessing the performance of sorghum varieties in the Guinea Savanna Zone of Ghana. Asian J Agric Food Sci 2(1):64–72. www.ajouronline.com
Li X, Chang Y, Ma S, Shen J, Hu H, **ong L (2019) Genome-wide identification of SNAC1-targeted genes involved in drought response in rice. Front Plant Sci 10:982. https://doi.org/10.3389/fpls.2019.00982
Liu G, Li X, ** S, Liu X, Zhu L, Nie Y, Zhang X (2014) Overexpression of rice NAC gene SNAC1 improves drought and salt tolerance by enhancing root development and reducing transpiration rate in transgenic cotton. PLoS ONE 9(1):e86895. https://doi.org/10.1371/journal.pone.0086895
Luo LJ (2010) Breeding for water-saving and drought-resistance rice (WDR) in China. J Expl Bot 61(13):3509–3517. https://doi.org/10.1093/jxb/erq185
Mahmood T, Khalid S, Abdullah M, Ahmed Z, Shah MKN, Ghafoor A, Du X (2019) Insights into drought stress signaling in plants and the molecular genetic basis of cotton drought tolerance. Cells 9(1):105. https://doi.org/10.3390/cells9010105
Makanda I, Tongoona P, Derera J, Sibiya J, Fato P (2010) Combining ability and cultivar superiority of sorghum germplasm for grain yield across tropical low- and mid-altitude environments. Field Crops Res 116:75–85. https://doi.org/10.1016/j.fcr.2009.11.015
Manavalan LP, Guttikonda SK, Phan Tran LS, Nguyen HT (2009) Physiological and molecular approaches to improve drought resistance in soybean. Plant Cell Physiol 50(7):1260–1276. https://doi.org/10.1093/pcp/pcp082
Murray SC, Sharma A, Rooney WL, Klein PE, Mullet JE, Mitchell SE, Kresovich S (2008) Genetic improvement of sorghum as a biofuel feedstock: I. QTL for stem sugar and grain nonstructural carbohydrates. Crop Sci 48(6): 2165–2179. https://doi.org/10.2135/cropsci2008.01.0068
Nagaraja GS, Varier A, Singh U, Sharma SP (2000) Characterization of inbreds and hybrids of sorghum by seed and seedling morphology and electrophoresis of seed proteins and isoenzymes. Plant Var Seeds 13:17–29
Ndlovu E, van Staden J, Maphosa M (2021) Morpho-physiological effects of moisture, heat and combined stresses on Sorghum bicolor [Moench (L.)] and its acclimation mechanisms. Plant Stress 2(2021):100018 https://doi.org/10.1016/j.stress.2021.100018
Nuruzzaman M, Sharoni AM, Satoh K et al (2012) Comprehensive gene expression analysis of the NAC gene family under normal growth conditions, hormone treatment, and drought stress conditions in rice using near-isogenic lines (NILs) generated from crossing Aday Selection (drought tolerant) and IR64. Mol Genet Genom 287:389–410. https://doi.org/10.1007/s00438-012-0686-8
Olorunmaiye KS, Joseph GG, Oyedeji ADAS (2019) Mutagenic components and dosage effects of ethyl methanesulphonate on Arachis hypogea (SAMNUT 24 VR.). Ife J Sci 21:309–322. https://doi.org/10.4314/ijs.v21i2.5
Palacios CE, Nagai A, Torres P, Rodrigues JA, Salatino A (2021) Contents of tannins of cultivars of sorghum cultivated in Brazil, as determined by four quantification methods. Food Chem 337:127970
Patil JV, Rakshit S, Khot KB (2013) Genetics of post-flowering drought tolerance traits in post-rainy sorghum [Sorghum bicolor (L.) Moench]. Indian J Genet Plant Breed 73(1):44–50. https://doi.org/10.5958/j.0019-5200.73.1.006
Payne RW, Murray DA, Harding SA, Baird DB, Soutar DM, Lane P (2007) GenStat for Windows, 10th Edition Introduction. VSN International, Hertfordshire
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nuc Acids Res 29(9):45–49. https://doi.org/10.1093/nar/29.9.e45
Prasad VBR, Govindaraj M, Djanaguiraman M, Djalovic I, Shailani A, Rawat N, Singla-Pareek SL, Pareek A, Prasad PVV (2021) Drought and high temperature stress in sorghum: physiological, genetic, and molecular insights and breeding approaches. Inter J Molecular Sci 22(18):9826. https://doi.org/10.3390/ijms22189826
Prasad PVV, Djanaguiraman M, Jagadish SVK, Ciampitti IA (2019) Drought and high temperature stress and traits associated with tolerance. In: Ciampitti IA, Prasad PVV (eds) Sorghum: a state of the art and future perspectives, vol 58. ASA, CSSA, SSSA; Madison, WI, pp 245–265
Quinby JR, Karper RE (1954) Inheritance of height in sorghum. Agron J 46:211–216. https://doi.org/10.2134/agronj1954.00021962004600050007x
Raghuvanshi S, Swamy D, Mahadevakumar S, Singh OP (2014) DUS discriptor of seventeen forage cultivars of sorghum [Sorghum bicolour (L.) Moench]. Glob J Biosc Biotechnol 3:100–108
Rajarajan K, Ganesamurthy K, Raveendran M et al (2021) Differential responses of sorghum genotypes to drought stress revealed by physio-chemical and transcriptional analysis. Mol Biolo Rep 48:2453–2462. https://doi.org/10.1007/s11033-021-06279-z
Rakshit S, Hariprasanna K, Gomashe S, Ganapathy KN, Das IK, Ramana OV ... Patil JV (2014) Changes in area, yield gains, and yield stability of sorghum in major sorghum‐producing countries, 1970 to 2009. Crop Sci 54(4):1571–1584. https://doi.org/10.2135/cropsci2012.12.0697
Ritter KB, Jordan DR, Chapman SC, Godwin ID, Mace ES, McIntyre CL (2008) Identification of QTL for sugar-related traits in a sweet × grain sorghum (Sorghum bicolor L. Moench) recombinant inbred population. Mol Breed 367–384. https://doi.org/10.1007/s11032-008-9182-6
Salas Fernandez MG, Becraft PW, Yin Y, Lübberstedt T (2009) From dwarves to giants? Plant height manipulation for biomass yield. Trends Plant Sci 14(8):454–461. https://doi.org/10.1016/j.tplants.2009.06.005
Selvaraj MG, Ishizaki T, Valencia M, Ogawa S, Dedicova B, Ogata T, Yoshiwara K, Maruyama K, Kusano M, Saito K, Takahashi F, Shinozaki K, Nakashima K, Ishitani M (2017) Overexpression of an Arabidopsis thaliana galactinol synthase gene improves drought tolerance in transgenic rice and increased grain yield in the field. Plant Biotechnol J 15(11):1465–1477. https://doi.org/10.1111/pbi.12731
Selvaraju P, Sivasubramaniam K (2000) Identification keys for sorghum varieties. Madras Agric J 87(10/12):660
Sharma JR (1998) Statistical and biometrical techniques in plant breeding. New Age International Pvt Limited, New Delhi
Shih CH, Siu SO, Ng R, Wong E, Chiu LC, Chu IK, Lo C (2007) Quantitative analysis of anticancer 3-deoxyanthocyanidins in infected sorghum seedlings. J Agric Food Chem 55(2):254–259. https://doi.org/10.1021/jf062516t
Sirany T, Tadele E, Aregahegn H, Wale D (2022) Economic potentials and use dynamics of sorghum food system in Ethiopia: Its implications to resolve food deficit. Adv Agric Article ID 4580643:11 https://doi.org/10.1155/2022/4580643
Sokal RR, Michener CD (1958) A statistical method for evaluating relationships. Univ Kans Sci Bull 38:1409–1448
Sudhakar Reddy P, Srinivas Reddy D, Sivasakthi K, Bhatnagar-Mathur P, Vadez V, Sharma KK (2016) Evaluation of Sorghum [Sorghum bicolor (L.)] reference genes in various tissues and under abiotic stress conditions for quantitative real-time PCR data normalization. Front Plant Sci 7:529. https://doi.org/10.3389/fpls.2016.00529
Tyagi AK, Sharma MK, Mishra SK, Kerkhi SA, Chand P (2014) Estimates of genetic variability, heritability and genetic advance in linseed (Linum usitatissinum L.) germplasm. Progres Agric 14(1): 37–48.
Waniska RD, Rooney LW, McDonough CM (2016) Sorghum: utilization. Encycl Food Grains Second Ed 6(3–4):116–123. https://doi.org/10.1016/B978-0-12-394437-5.00128-5
Wu Y, Li X, **ang W, Zhu C, Lin Z, Wu Y, Li J, Pandravada S, Ridder DD, Bai G, Wang ML, Trick HN, Bean SR, Tuinstra MR, Tesso TT, Yu J (2012) Presence of tannins in sorghum grains is conditioned by different natural alleles of Tannin1. Proc Nat Acad Sci USA 109(26):10281–10286. https://doi.org/10.1073/pnas.1201700109
Xu Z, Zhou G, Shimizu H (2018) Plant responses to drought and rewatering. Plant Signal Behav 5:649–654. https://doi.org/10.4161/psb.5.6.11398
Yu Y, Zhang G, Chen Y, Bai Q, Gao C, Zeng L., Li Z., Cheng Y, Chen J, Sun X, Guo L, Xu J, Yan Z (2019) Selection of reference genes for qPCR analyses of gene expression in ramie leaves and roots across eleven abiotic/biotic treatments. Sci Rep 9:20004. https://doi.org/10.1038/s41598-019-56640-3
Zhao X, Zhang X, Guo X, Li S, Han L, Song Z, Wang Y, Li J, Li M (2016) Identification and validation of reference genes for qRT-PCR studies of gene expression in Dioscorea opposita. Biomed Res Inter 2016:3089584. https://doi.org/10.1155/2016/3089584
Acknowledgements
We thank Dr Andreas Boerner of the Genetic Resource Unit, Institute of Plant Genetics and Crop Plant Research (IPK), Germany, and The Seed GenBank staff at the National Centre for Genetic Resources and Biotechnology (NACGRAB), Ibadan Nigeria for graciously providing the accessions used for the study.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
All the authors contributed significantly to the study. DAA and KAA conceptualise and design the experiments. DAA, KAA, ATA, AKA, KAA, FKA and JTO planned, conducted, and analysed the drought field experiments. DAA, KAA, ATA, AKA, and KAA conducted gene expression experiments and analyses. DAA and KAA wrote the manuscript, and all the authors checked it and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Animasaun, D.A., Adedoyin, K.A., Bakare, A.T. et al. Phenotypic characterization of sorghum seedlings and expression patterns of SNAC1 and DREB1A genes under water-stress: an insight towards develo** resilient cultivars. Genet Resour Crop Evol (2024). https://doi.org/10.1007/s10722-024-01956-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10722-024-01956-x