Abstract
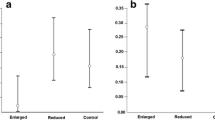
Rearing density has profound effects on life-history decisions for develo** organisms because, for example, it can foreshadow the sexual competitive environment potentially experienced in adulthood. Increased sexual competitiveness should favour greater investment in pre- or post-copulatory sexually selected traits such as weaponry or testes, while sexual-signaling traits that attract mates from a long distance should be favoured in low density populations. High density environments should also trigger density-dependent prophylaxis in which individuals increase their investment in immunity. In this study, we experimentally investigated the effect of rearing density on the adult expression of an immunological trait (i.e., melanization response) as well as pre- (i.e., mandible length, harp size) and post-copulatory (i.e., testis size) sexually selected traits in male Gryllus firmus crickets. We found that all morphological traits positively intercorrelated within each environment whereas melanization response was unrelated to any other measured trait. We also found that high-density males produced larger mandibles (weaponry), contrary to expectation, and stronger immunological responses (degree of melanization), in line with expectation, than their low-density counterparts. As predicted, low-density males produced larger harps, a trait involved in sexual signaling. Our results suggest that male G. firmus facultatively partition resources in a manner that maximizes fitness in the environment they expect to encounter as adults.

Similar content being viewed by others
Data availability
References
Anderson RM, May RM (1979) Population biology of infectious diseases: part I. Nature 280:361–367
Applebaum S, Heifetz Y (1999) Density-dependent physiological phase in insects. Ann Rev Entomol 44:317–341
Bailey NW, Gray B, Zuk M (2010) Acoustic experience shapes alternative mating tactics and reproductive investment in male field crickets. Curr Biol 20:845–849
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:105
Bennet-Clark HC (1989) Songs and the physics of sound production. In: Huber F, Moore LTE (eds) Cricket behavior and neurobiology. Cornell University Press, Ithaca, NY, USA, pp 227–261
Bretman A, Fricke C, Westmancoat J, Chapman T (2016) Effect of competitive cues on reproductive morphology and behavioral plasticity in male fruitflies. Behav Ecol 27:452–461
Brown CR, Brown MB (2003) Testis size increases with colony size in cliff swallows. Behav Ecol 14:569–575
Cotter SC, Hails RS, Cory JS, Wilson K (2004) Density-dependent prophylaxis and condition‐dependent immune function in Lepidopteran larvae: a multivariate approach. J Anim Ecol 73:283–293
Friesen OC, Li C-H, Sykes EME et al (2022) Density-dependent prophylaxis in freshwater snails driven by oxylipin chemical cues. Front Immunol 13:153
Gage MJ (1995) Continuous variation in reproductive strategy as an adaptive response to population density in the moth Plodia interpunctella. Proc Royal Soc Lond Ser B: Biol Sci 261:25–30
Halekoh U, Højsgaard S (2014) A kenward-roger approximation and parametric bootstrap methods for tests in linear mixed models – the R package pbkrtest. J Stat Softw 59:1–32
Hissmann K (1990) Strategies of mate finding in the european field cricket (Gryllus campestris) at different population densities: a field study. Ecol Entomol 15:281–291
Hosken D (2001) Sex and death: microevolutionary trade-offs between reproductive and immune investment in dung flies. Curr Biol 11:R379–R380
Hosken DJ, Ward PI (2001) Experimental evidence for testis size evolution via sperm competition. Ecol Lett 4:10–13
Iba M, Nagao T, Urano A (1995) Effects of population density on growth, behavior and levels of biogenic amines in the cricket, Gryllus bimaculatus. Zoolog Sci 12:695–702
Johnson T, Symonds M, Elgar M (2017) Anticipatory flexibility: larval population density in moths determines male investment in antennae, wings and testes. Proc Royal Soc Lond Ser B: Biol Sci 284:20172087
Judge K, Bonanno V (2008) Male weaponry in a fighting cricket. PLoS ONE 3:e3980
Kasumovic MM, Brooks RC (2011) It’s all who you know: the evolution of socially cued anticipatory plasticity as a mating strategy. Q Rev Biol 86:181–197
Kelly C (2018) The causes and evolutionary consequences of variation in female mate choice in insects: the effects of individual state, genotypes and environments. Curr Opin Insect Sci 27:1–8
Kelly CD, L’Heureux V (2021) Effect of diet and rearing density on contest outcome and settlement in a field cricket. Behav Ecol Sociobiol 75:1–14
Kelly CD, Tawes B, Worthington A (2014) Evaluating indices of body condition in two cricket species. Ecol Evol 4:4476–4487
Knell RJ (2009) Population density and the evolution of male aggression. J Zool 278:83–90
Kokko H, Rankin DJ (2006) Lonely hearts or sex in the city? Density-dependent effects in mating systems. Philosophical Trans Royal Soc B: Biol Sci 361:319–334
Lemaître J, Ramm S, Hurst J, Stockley P (2011) Social cues of sperm competition influence accessory reproductive gland size in a promiscuous mammal. Proc Biol Sci 278:1171–1176
Lüdecke D, Ben-Shachar M, Patil I et al (2021) Performance: an R package for assessment, comparison and testing of statistical models. J Open Source Softw 6:3139
Mazurkiewicz A, Tumialis D, Pezowicz E et al The effect of density on the breeding optimization of the tropical house cricket Gryllodes sigillatus (Walker) (Orthoptera: Gryllidae)
Nylin S, Gotthard K (1998) Plasticity in life-history traits. Ann Rev Entomol 43:63–83
Parker G, Lessells C, Simmons L (2013) Sperm competition games: a general model for precopulatory male-male competition. Evolution 67:95–109
Peters TM, Barbosa P (1977) Influence of population density on size, fecundity, and developmental rate of insects in culture. Ann Rev Entomol 22:431–450
R Development Core Team (2023) R: A language and environment for statistical computing. Version: 4.2.3. Vienna (Austria): R Foundation for Statistical Computing
Saglam I, Roff D, Fairbairn D (2008) Male sand crickets trade-off flight capability for reproductive potential. J Evol Biol 21:997–1004
Scheuber H, Jacot A, Brinkhof M (2003) The effect of past condition on a multicomponent sexual signal. Proc Royal Soc Lond Ser B: Biol Sci 270:1779–1784
Simmons LW (1986) Inter-male competition and mating success in the field cricket, Gryllus bimaculatus (de Geer). Anim Behav 34:567–579
Simmons LW (2001) Sperm competition and its evolutionary Consequences in the insects. Princeton University Press
Simons AM, Roff DA (1994) The effect of environmental variability on the heritabilities of traits of a field cricket. Evolution 48:1637
Simons AM, Roff DA (1996) The effect of a variable environment on the genetic correlation structure in a field cricket. Evolution 50:267
Siva-Jothy MT, Moret Y, Rolff J (2005) Insect immunity: an Evolutionary Ecology Perspective. Adv Insect Physiol 32:1–48
Stockley P, Seal N (2001) Plasticity in reproductive effort of male dung flies (Scatophaga stercoraria) as a response to larval density. Funct Ecol 15:96–102
Tan GN, Govedich FR, Burd M (2004) Social group size, potential sperm competition and reproductive investment in a hermaphroditic leech, Helobdella papillornata (Euhirudinea: Glossiphoniidae). J Evol Biol 17:574–580
Walker SE, Roberts JA, Adame I et al (2008) Heads up: sexual dimorphism in house crickets (Acheta domesticus). Can J Zool 86:253–259
Webb KL, Roff DA (1992) The quantitative genetics of sound production in Gryllus firmus. Anim Behav 44:823–832
Wey TW, Réale D, Kelly CD (2019) Developmental and genetic effects on behavioral and life-history traits in a field cricket. Ecol Evol 9:3434–3445
Wilson K, Cotter SC (2009) Density-dependent prophylaxis in insects. In: Whitman DW, Ananthakrishnan TN (eds) Phenotypic plasticity of insects: mechanisms and consequences. Science Publishers Inc., Plymouth, UK, pp 381–420
Wilson K, Reeson AF (1998) Density-dependent prophylaxis: evidence from Lepidoptera-baculovirus interactions. Ecol Entomol 23:100–101
Wilson K, Thomas MB, Blanford S et al (2002) Co** with crowds: density-dependent disease resistance in desert locusts. Proc Natl Acad Sci 99:5471–5475
Acknowledgements
This study was supported by Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grants to CDK and DR as well as by funding from Canada Research Chairs (CDK).
Funding
CDK wishes to thank the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Canada Research Chairs program.
Author information
Authors and Affiliations
Contributions
TWW: Designed experiment and collected data. VL: Collected data. DR: Designed experiment. CDK: Designed experiment, analyzed data, wrote manuscript, dealt with all reviewer comments, and manuscript revisions.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kelly, C.D., L’Heureux, V., Wey, T.W. et al. Effect of rearing density on the expression of fitness-related traits in male sand field crickets (Gryllus firmus). Evol Ecol 37, 835–846 (2023). https://doi.org/10.1007/s10682-023-10251-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-023-10251-7