Abstract
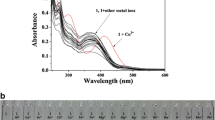
We designed and synthesized a fluorescent “turn-on” and colorimetric chemosensor ((E)-1-((p-tolylimino)methyl)naphthalen-2-ol) SB. The structure of the synthesized chemosensor was investigated by 1H NMR, FT-IR, and fluorescence spectroscopy, and its sensing properties were studied toward Mn2+, Cu2+, Pb2+, Cd2+, Na+, Ni2+, Al3+, K+, Ag+, Zn2+, Co2+, Cr3+, Hg2+, Ca2+, and Mg2+. SB showed an excellent colorimetric (yellow to yellowish brown) in MeOH and fluorescence “turn-on” sensing response to Cu2+ in MeOH/Water (10/90, v/v) media. The sensing mechanism of SB toward Cu2+ was investigated by FT-IR, 1H NMR titration, DFT studies, and Job’s plot analysis. The detection limit was calculated to be very low 0.0025 µg mL−1 (0.0025 ppm). Furthermore, the test strip containing SB also showed excellent selectivity and sensitivity toward Cu2+ in a solution medium and when supported on a solid medium.

















Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. All authors have read, understood, and have complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors.
References
Alharbi, K. H. (2022). A review on organic colorimetric and fluorescent chemosensors for the detection of Zn (II) ions. Taylor & Francis. https://doi.org/10.1080/10408347.2022.2033611. Accessed 11 February 2022.
Al-Saidi, H. M., & Khan, S. (2022a). Recent advances in thiourea based colorimetric and fluorescent chemosensors for detection of anions and neutral analytes: a review. https://doi.org/10.1080/10408347.2022.2063017, 1–17. https://doi.org/10.1080/10408347.2022.2063017
Al-Saidi, H. M., & Khan, S. (2022b). A review on organic fluorimetric and colorimetric chemosensors for the detection of Ag(I) ions. https://doi.org/10.1080/10408347.2022.2133561, 1–27. https://doi.org/10.1080/10408347.2022.2133561
Allen, R. O., & Brookhart, W. (2002). Determination of copper in complex matrices by neutron activation analysis using X-ray detection. Analytical Chemistry, 46(9), 1297–1299. https://doi.org/10.1021/AC60345A055
Ansari, T. M., Marr, I. L., & Tariq, N. (2003). Heavy metals in marine pollution perspective–a mini review. Journal of Applied Sciences, 4(1), 1–20. https://doi.org/10.3923/jas.2004.1.20
Aydin, D., Karuk Elmas, S. N., Savran, T., Arslan, F. N., Sadi, G., & Yilmaz, I. (2021). An ultrasensitive ″OFF–ON″ fluorogenic sensor based on thiazole derivative for Zn2+: food supplement, water and bio–imaging applications. Journal of Photochemistry and Photobiology A: Chemistry, 419. https://doi.org/10.1016/J.JPHOTOCHEM.2021.113459
Barceloux, D. G. (1999). Copper. Journal of Toxicology - Clinical Toxicology. Taylor & Francis. https://doi.org/10.1081/CLT-100102421
Delley, B. (2000). From molecules to solids with the DMol3 approach. The Journal of Chemical Physics, 113(18), 7756. https://doi.org/10.1063/1.1316015
Dubey, P., Thakur, V., & Chattopadhyay, M. (2020). Role of minerals and trace elements in diabetes and insulin resistance. Nutrients, 12(6), 1–17. https://doi.org/10.3390/NU12061864
Ganguly, A., Ghosh, S., Kar, S., & Guchhait, N. (2015). Selective fluorescence sensing of Cu(II) and Zn(II) using a simple Schiff base ligand: Naked eye detection and elucidation of photoinduced electron transfer (PET) mechanism. Spectrochimica Acta Part a: Molecular and Biomolecular Spectroscopy, 143, 72–80. https://doi.org/10.1016/J.SAA.2015.02.013
Ghaedi, M., Ahmadi, F., & Shokrollahi, A. (2007). Simultaneous preconcentration and determination of copper, nickel, cobalt and lead ions content by flame atomic absorption spectrometry. Journal of Hazardous Materials, 142(1–2), 272–278. https://doi.org/10.1016/J.JHAZMAT.2006.08.012
Huang, J. F., & Lin, B. T. (2009). Application of a nanoporous gold electrode for the sensitive detection of copperviamercury-free anodic strip** voltammetry. The Analyst, 134(11), 2306–2313. https://doi.org/10.1039/B910282E
Kargar, H., Forootan, P., Fallah-Mehrjardi, M., Behjatmanesh-Ardakani, R., Amiri Rudbari, H., Shahzad Munawar, K., et al. (2021). Novel oxovanadium and dioxomolybdenum complexes of tridentate ONO-donor Schiff base ligand: synthesis, characterization, crystal structures, Hirshfeld surface analysis, DFT computational studies and catalytic activity for the selective oxidation of benzylic alcohols. Inorganica Chimica Acta, 523, 120414. https://doi.org/10.1016/J.ICA.2021.120414
Khan, S., Chen, X., Almahri, A., Allehyani, E. S., Alhumaydhi, F. A., Ibrahim, M. M., & Ali, S. (2021). Recent developments in fluorescent and colorimetric chemosensors based on schiff bases for metallic cations detection: a review. Journal of Environmental Chemical Engineering, 9(6), 106381. https://doi.org/10.1016/J.JECE.2021.106381
Khan, S., Muhammad, M., Al-Saidi, H. M., Hassanian, A. A., Alharbi, W., & Alharbi, K. H. (2022). Synthesis, characterization and applications of Schiff base chemosensor for determination of Cu2+ ions. Journal of Saudi Chemical Society, 26(4), 101503. https://doi.org/10.1016/J.JSCS.2022.101503
Kim, J. J., Kim, Y. S., & Kumar, V. (2019, July 1). Heavy Metal Toxicity: An Update of Chelating Therapeutic Strategies. Journal of Trace Elements in Medicine and Biology. Elsevier GmbH. https://doi.org/10.1016/j.jtemb.2019.05.003
Liu, P., & Rodriguez, J. A. (2005). Catalysts for hydrogen evolution from the [NiFe] hydrogenase to the Ni 2P(001) surface: The importance of ensemble effect. Journal of the American Chemical Society, 127(42), 14871–14878. https://doi.org/10.1021/JA0540019/ASSET/IMAGES/JA0540019.SOCIAL.JPEG_V03
Mergu, N., & Gupta, V. K. (2015). A novel colorimetric detection probe for copper(II) ions based on a Schiff base. Sensors and Actuators B: Chemical, 210, 408–417. https://doi.org/10.1016/J.SNB.2014.12.130
Muhammad, M., Khan, S., & Fayaz, H. (2021). Charge-transfer complex–based spectrophotometric method for the determination of mesotrione in environmental samples. Environmental Monitoring and Assessment, 193(10). https://doi.org/10.1007/S10661-021-09432-0
Muhammad, M., Khan, S., Rahim, G., Alharbi, W., & Alharbi, K. H. (2022). Highly selective and sensitive spectrofluorimetric method for determination of cypermethrin in different environmental samples. Environmental Monitoring and Assessment, 194(12), 890. https://doi.org/10.1007/S10661-022-10640-5
Myint, Z. W., Oo, T. H., Thein, K. Z., Tun, A. M., & Saeed, H. (2018, September 1). Copper Deficiency Anemia: Review Article Annals of Hematology. Springer Verlag. https://doi.org/10.1007/s00277-018-3407-5
Otero-Romaní, J., Moreda-Piñeiro, A., Bermejo-Barrera, P., & Martin-Esteban, A. (2009). Inductively coupled plasma–optical emission spectrometry/mass spectrometry for the determination of Cu, Ni, Pb and Zn in seawater after ionic imprinted polymer based solid phase extraction. Talanta, 79(3), 723–729. https://doi.org/10.1016/J.TALANTA.2009.04.066
Perdew, J. P., Burke, K., & Ernzerhof, M. (1996). Generalized gradient approximation made simple. Physical Review Letters, 77(18), 3865. https://doi.org/10.1103/PhysRevLett.77.3865
Royer, A., & Sharman, T. (2021). Copper toxicity. StatPearls. StatPearls Publishing. http://www.ncbi.nlm.nih.gov/pubmed/32491388. Accessed 17 April 2021.
Shah, R., Katouah, H., Sedayo, A. A., Abualnaja, M., Aljohani, M. M., Saad, F., et al. (2020). Practical and computational studies on novel Schiff base complexes derived from green synthesis approach: conductometry as well as in-vitro screening supported by in-silico study. Journal of Molecular Liquids, 319, 114116. https://doi.org/10.1016/J.MOLLIQ.2020.114116
Tong, L., Sun, S., Qiao, M., Fan, J., & Ding, L. (2019). Selective turn-on sensing of Cu2+ and Cl− by a ferrocene-modified pyrene derivative. Journal of Photochemistry and Photobiology a: Chemistry, 374, 131–137. https://doi.org/10.1016/j.jphotochem.2019.01.036
Uauy, R., Olivares, M., & Gonzalez, M. (1998). Essentiality of copper in humans. The American Journal of Clinical Nutrition, 67(5), 952S-959S. https://doi.org/10.1093/ajcn/67.5.952S
Udhayakumari, D., Naha, S., & Velmathi, S. (2017). Colorimetric and fluorescent chemosensors for Cu2+. A comprehensive review from the years 2013–15. Analytical Methods, 9(4), 552–578. https://doi.org/10.1039/C6AY02416E
Uriu-Adams, J. Y., & Keen, C. L. (2005, August 1). Copper, oxidative stress, and human health. Molecular Aspects of Medicine. Pergamon. https://doi.org/10.1016/j.mam.2005.07.015
**ng, G., Sardar, M. R., Lin, B., & Lin, J. M. (2019). Analysis of trace metals in water samples using NOBIAS chelate resins by HPLC and ICP-MS. Talanta, 204, 50–56. https://doi.org/10.1016/J.TALANTA.2019.05.041
Acknowledgements
The researchers would like to acknowledge Deanship of Scientific Research, Taif University for funding this work.
Author information
Authors and Affiliations
Contributions
Sikandar Khan: conceptualization, data curation and writing—original draft; Mian Muhammad: supervision and project administration; Waheed Kamran: formal analysis and conceptualization; Hamed M. Al-Saidi: investigation, resources, and data curation; Salman S. Alharthi: editing and visualization; Jari S. Algethami: validation and investigation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khan, S., Muhammad, M., Kamran, A.W. et al. An ultrasensitive colorimetric and fluorescent “turn-on” chemosensor based on Schiff base for the detection of Cu2+ in the aqueous medium. Environ Monit Assess 195, 633 (2023). https://doi.org/10.1007/s10661-023-11260-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-11260-3