Abstract
Background
Increased SOX4 (SRY-related HMG-box) activity aids cellular transformation and metastasis. However, its specific functions and downstream targets remain to be completely elusive in colorectal cancer (CRC).
Aims
To investigate the role of SOX4 in CRC progression and the underlying mechanism.
Methods
In the current study, online available datasets of CRC patients were explored to check the expression status of SOX4. To investigate the further functions, SOX4 was overexpressed and knocked down in CRC cells. Colony formation assay, flowcytometry analysis, and MTT assay were used to check for proliferation and apoptosis. Acridine orange staining was done to check the role of SOX4 in autophagy induction. Furthermore, western blot, qRT-PCR, and bioinformatic analysis was done to elucidate the downstream molecular mechanism of SOX4.
Results
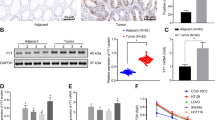
GEPIA database showed enhanced expression of SOX4 mRNA in CRC tumor, and the human protein atlas (HPA) showed strong staining of SOX4 protein in tumor when compared to the normal tissue. Ectopic expression of SOX4 enhanced colony formation ability as well as rescued cells from apoptosis. SOX4 overexpressed cells showed the formation of acidic vesicular organelles (AVOs) which indicated autophagy. Further results revealed the activation of p-AKT/MAPK molecules upon overexpression of SOX4. SOX4 expression was found to be positively correlated with histone deacetylase 2 (HDAC2). Knockdown of SOX4 or HDAC2 inhibition induced apoptosis, revealed by decrease in BCL2 and increase in BAX expression, and inactivated the p-AKT/MAPK signaling.
Conclusion
The study uncovers that SOX4/HDAC2 axis improves cell survivability and reduces apoptosis via activation of the p-AKT/MAPK pathway.
Graphical Abstract









Similar content being viewed by others
Data availability
The original data generated during the current study are available from the corresponding author in the supplementary information file. The dataset analysed during the current study are available in GEPIA (http://gepia.cancer-pku.cn/) and cBioPortal (https://www.cbioportal.org/).
References
Sung H, Ferlay J, Siegel RL et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209–249. https://doi.org/10.3322/CAAC.21660.
Chakraborty SA, Patnaik S. Clonal evolution and expansion associated with therapy resistance and relapse of colorectal cancer. Mutat Res 2022;790:108445. https://doi.org/10.1016/J.MRREV.2022.108445.
Vishnoi K, Viswakarma N, Rana A, Rana B. Transcription factors in cancer development and therapy. Cancers (Basel) 2020;12:1–32. https://doi.org/10.3390/CANCERS12082296.
Restivo A, Piacentini G, Placidi S et al. Cardiac outflow tract: a review of some embryogenetic aspects of the conotruncal region of the heart. Anat Rec A Discov Mol Cell Evol Biol 2006;288:936–943. https://doi.org/10.1002/AR.A.20367.
Xu F, Lv YM, Bin WH, Song YC. miR-31-5p/SOX4 axis affects autophagy and apoptosis of chondrocytes by regulating extracellular regulated protein kinase/mechanical target of rapamycin kinase signalling. Pathobiology 2022;89:63–73. https://doi.org/10.1159/000519006.
Krämer OH, Zhu P, Ostendorff HP et al. The histone deacetylase inhibitor valproic acid selectively induces proteasomal degradation of HDAC2. EMBO J 2003;22:3411–3420. https://doi.org/10.1093/EMBOJ/CDG315.
Zhu P, Martin E, Mengwasser J et al. Induction of HDAC2 expression upon loss of APC in colorectal tumorigenesis. Cancer Cell 2004;5:455–463. https://doi.org/10.1016/S1535-6108(04)00114-X.
Tang Z, Li C, Kang B et al. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res 2017;45:W98–W102. https://doi.org/10.1093/NAR/GKX247.
Wang Z, Jensen MA, Zenklusen JC. A practical guide to the cancer genome atlas (TCGA). Methods Mol Biol 2016;1418:111–141. https://doi.org/10.1007/978-1-4939-3578-9_6.
Wei C, Yang C, Wang S et al. Crosstalk between cancer cells and tumor associated macrophages is required for mesenchymal circulating tumor cell-mediated colorectal cancer metastasis. Mol Cancer 2019;18:64–64. https://doi.org/10.1186/S12943-019-0976-4.
SenthilKumar G, Skiba JH, Kimple RJ. High-throughput quantitative detection of basal autophagy and autophagic flux using image cytometry. Biotechniques 2019;67:70–73. https://doi.org/10.2144/BTN-2019-0044.
Zhang H, Kong X, Kang J et al. Oxidative stress induces parallel autophagy and mitochondria dysfunction in human glioma U251 cells. Toxicol Sci 2009;110:376–388. https://doi.org/10.1093/TOXSCI/KFP101.
Jauch R, Ng CKL, Narasimhan K, Kolatkar PR. The crystal structure of the Sox4 HMG domain-DNA complex suggests a mechanism for positional interdependence in DNA recognition. Biochem J 2012;443:39–47. https://doi.org/10.1042/BJ20111768.
Lin CM, Fang CL, Hseu YC et al. Clinical and prognostic implications of transcription factor SOX4 in patients with colon cancer. PLoS One. 2013. https://doi.org/10.1371/JOURNAL.PONE.0067128.
Mourdjeva M, Kyurkchiev D, Mandinova A et al. Dynamics of membrane translocation of phosphatidylserine during apoptosis detected by a monoclonal antibody. Apoptosis 2005;10:209–217. https://doi.org/10.1007/S10495-005-6076-5.
Goumenos DS, Tsamandas AC, Kalliakmani P et al. Expression of apoptosis-related proteins bcl-2 and bax along with transforming growth factor (TGF-beta1) in the kidney of patients with glomerulonephritides. Ren Fail 2004;26:361–367. https://doi.org/10.1081/JDI-120039818.
Cheng BC, Chen JT, Yang ST et al. Cobalt chloride treatment induces autophagic apoptosis in human glioma cells via a p53-dependent pathway. Int J Oncol 2017;50:964–974. https://doi.org/10.3892/IJO.2017.3861.
Yun CW, Jeon J, Go G et al. The dual role of autophagy in cancer development and a therapeutic strategy for cancer by targeting autophagy. Int J Mol Sci 2020;22:1–22. https://doi.org/10.3390/IJMS22010179.
Tanida I, Ueno T, Kominami E. LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 2004;36:2503–2518. https://doi.org/10.1016/j.biocel.2004.05.009.
Yoshii SR, Mizushima N. Monitoring and measuring autophagy. Int J Mol Sci. 2017. https://doi.org/10.3390/IJMS18091865.
Wang C, Wang H, Zhang D et al. Phosphorylation of ULK1 affects autophagosome fusion and links chaperone-mediated autophagy to macroautophagy. Nat Commun. 2018. https://doi.org/10.1038/S41467-018-05449-1.
Mizushima N. The role of the Atg1/ULK1 complex in autophagy regulation. Curr Opin Cell Biol 2010;22:132–139. https://doi.org/10.1016/J.CEB.2009.12.004.
Moreno CS. SOX4: The unappreciated oncogene. Semin Cancer Biol 2020;67:57–64. https://doi.org/10.1016/J.SEMCANCER.2019.08.027.
Bilir B, Osunkoya AO, Wiles WG et al. SOX4 is essential for prostate tumorigenesis initiated by PTEN ablation. Cancer Res 2016;76:1112. https://doi.org/10.1158/0008-5472.CAN-15-1868.
Tazzari PL, Cappellini A, Bortul R et al. Flow cytometric detection of total and serine 473 phosphorylated Akt. J Cell Biochem 2002;86:704–715. https://doi.org/10.1002/JCB.10262.
Liu S, Gao F, Wen L et al. Osteocalcin induces proliferation via positive activation of the PI3K/Akt, P38 MAPK pathways and promotes differentiation through activation of the GPRC6A-ERK1/2 pathway in C2C12 myoblast cells. Cell Physiol Biochem 2017;43:1100–1112. https://doi.org/10.1159/000481752.
Wang K, Ji W, Yu Y et al. FGFR1-ERK1/2-SOX2 axis promotes cell proliferation, epithelial-mesenchymal transition, and metastasis in FGFR1-amplified lung cancer. Oncogene 2018;37:5340–5354. https://doi.org/10.1038/S41388-018-0311-3.
Wang L, Zhang J, Yang X et al. SOX4 is associated with poor prognosis in prostate cancer and promotes epithelial-mesenchymal transition in vitro. Prostate Cancer Prostatic Dis 2013;16:301–307. https://doi.org/10.1038/PCAN.2013.25.
Zhang J, **ao C, Feng Z et al. SOX4 promotes the growth and metastasis of breast cancer. Cancer Cell Int. 2020. https://doi.org/10.1186/S12935-020-01568-2.
Sun J, Piao J, Li N et al. Valproic acid targets HDAC1/2 and HDAC1/PTEN/Akt signalling to inhibit cell proliferation via the induction of autophagy in gastric cancer. FEBS J 2020;287:2118–2133. https://doi.org/10.1111/FEBS.15122.
Song G, Ouyang G, Bao S. The activation of Akt/PKB signaling pathway and cell survival. J Cell Mol Med 2005;9:59–71. https://doi.org/10.1111/J.1582-4934.2005.TB00337.X.
Janssen E, Subtil B, Ortiz FJ et al. Combinatorial immunotherapies for metastatic colorectal cancer. Cancers (Basel) 2020;12:1–29. https://doi.org/10.3390/CANCERS12071875.
Wang C, Zhao H, Lu J et al. Clinicopathological significance of SOX4 expression in primary gallbladder carcinoma. Diagn Pathol. 2012. https://doi.org/10.1186/1746-1596-7-41.
Xu X, Zong K, Wang X et al. miR-30d suppresses proliferation and invasiveness of pancreatic cancer by targeting the SOX4/PI3K-AKT axis and predicts poor outcome. Cell Death Dis. 2021. https://doi.org/10.1038/S41419-021-03576-0.
Bache M, Kappler M, Said H et al. Detection and specific targeting of hypoxic regions within solid tumors: current preclinical and clinical strategies. Curr Med Chem 2008;15:322–338. https://doi.org/10.2174/092986708783497391.
Qureshi-Baig K, Kuhn D, Viry E et al. Hypoxia-induced autophagy drives colorectal cancer initiation and progression by activating the PRKC/PKC-EZR (ezrin) pathway. Autophagy 2020;16:1436–1452. https://doi.org/10.1080/15548627.2019.1687213.
Liao YL, Sun YM, Chau GY et al. (2008) Identification of SOX4 target genes using phylogenetic footprinting-based prediction from expression microarrays suggests that overexpression of SOX4 potentiates metastasis in hepatocellular carcinoma. Oncogene 2008;27(42):5578–5589. https://doi.org/10.1038/onc.2008.168.
Qi ZP, Yalikong A, Zhang JW et al. HDAC2 promotes the EMT of colorectal cancer cells and via the modular scaffold function of ENSG00000274093.1. J Cell Mol Med 2021;25:1190–1197. https://doi.org/10.1111/JCMM.16186.
Cui H, Yu W, Yu M et al. GPR126 regulates colorectal cancer cell proliferation by mediating HDAC2 and GLI2 expression. Cancer Sci 2021;112:1798–1810. https://doi.org/10.1111/CAS.14868.
Zhu X, Leboeuf M, Liu F et al. HDAC1/2 Control proliferation and survival in adult epidermis and pre-basal cell carcinoma through p16 and p53. J Invest Dermatol 2022;142:77-87.e10. https://doi.org/10.1016/J.JID.2021.05.026.
Hur W, Rhim H, Jung CK et al. SOX4 overexpression regulates the p53-mediated apoptosis in hepatocellular carcinoma: clinical implication and functional analysis in vitro. Carcinogenesis 2010;31:1298–1307. https://doi.org/10.1093/CARCIN/BGQ072.
Moresi V, Carrer M, Grueter CE et al. Histone deacetylases 1 and 2 regulate autophagy flux and skeletal muscle homeostasis in mice. Proc Natl Acad Sci USA 2012;109:1649–1654. https://doi.org/10.1073/PNAS.1121159109/SUPPL_FILE/PNAS.201121159SI.PDF.
Punpai S, Saenkham A, Jarintanan F et al. HDAC inhibitor cowanin extracted from G. fusca induces apoptosis and autophagy via inhibition of the PI3K/Akt/mTOR pathways in Jurkat cells. Biomed Pharmacother. 2022. https://doi.org/10.1016/J.BIOPHA.2021.112577.
Sun Y, Liu WZ, Liu T et al. Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J Recept Signal Transduct Res 2015;35:600–604. https://doi.org/10.3109/10799893.2015.1030412.
Cui H, Darmanin S, Natsuisaka M et al. Enhanced expression of asparagine synthetase under glucose-deprived conditions protects pancreatic cancer cells from apoptosis induced by glucose deprivation and cisplatin. Cancer Res 2007;67:3345–3355. https://doi.org/10.1158/0008-5472.CAN-06-2519.
Ramezani-Rad P, Geng H, Hurtz C et al. SOX4 enables oncogenic survival signals in acute lymphoblastic leukemia. Blood 2013;121:148–155. https://doi.org/10.1182/BLOOD-2012-05-428938.
Liang F, Ren C, Wang J et al. The crosstalk between STAT3 and p53/RAS signaling controls cancer cell metastasis and cisplatin resistance via the Slug/MAPK/PI3K/AKT-mediated regulation of EMT and autophagy. Oncogenesis. 2019. https://doi.org/10.1038/S41389-019-0165-8.
Lee S, Rauch J, Kolch W. Targeting MAPK signaling in cancer: mechanisms of drug resistance and sensitivity. Int J Mol Sci. 2020. https://doi.org/10.3390/IJMS21031102.
Chen JH, Zheng YL, Xu CQ et al. Valproic acid (VPA) enhances cisplatin sensitivity of non-small cell lung cancer cells via HDAC2 mediated down regulation of ABCA1. Biol Chem 2017;398:785–792. https://doi.org/10.1515/HSZ-2016-0307.
Carey N, La Thangue NB. Histone deacetylase inhibitors: gathering pace. Curr Opin Pharmacol 2006;6:369–375. https://doi.org/10.1016/J.COPH.2006.03.010.
Suzuki T, Yokozaki H, Kuniyasu H et al. Effect of trichostatin A on cell growth and expression of cell cycle-and apoptosis-related molecules in human gastric and oral carcinoma cell lines. Int J Cancer. 2000. https://doi.org/10.1002/1097-0215.
Acknowledgments
We are thankful to the KIIT School of Biotechnology, KIIT Deemed to be University and all the participants who made this study possible.
Funding
We acknowledge Department of Biotechnology (DBT), Ministry of Science and Technology, India (Grant No. BT/INF/22/SP42155/2021), Government of India for the funding. AS acknowledges the CSIR for research fellowship support under the CSIR-NET-SRF program (09/1035(0010)/2017-EMR-1).
Author information
Authors and Affiliations
Contributions
AS conceived the idea of the study, drafted the protocol, contributed to data generation, analysis, and manuscript writing. AC made contribution to data generation and manuscript writing. SP analyzed and interpreted the data, and made contributions to the conceptualization, supervision, review, and editing of the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest to disclose. The research was conducted in the absence of any commercial or financial relationship that could be constructed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
S, A., Chakraborty, A. & Patnaik, S. SOX4/HDAC2 Axis Enhances Cell Survivability and Reduces Apoptosis by Activating AKT/MAPK Signaling in Colorectal Cancer. Dig Dis Sci 69, 835–850 (2024). https://doi.org/10.1007/s10620-023-08215-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-08215-6