Abstract
Background
Nonadherence to direct-acting agents (DAAs) for hepatitis C (HCV) decreases viral response. To measure nonadherence to DAAs, a reliable, valid, and easily implemented method is needed.
Aims
The goals of this study were to refine a previously validated (in patients with hypertension) self-report measure of extent of nonadherence and reasons for nonadherence in the context of DAAs and to obtain initial evidence of content validity and reliability.
Methods
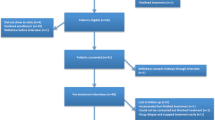
Phase I involved two focus groups with patients with HCV (n = 12) and one focus group with prescribers of HCV medications (n = 6) to establish content validity of reasons for nonadherence. Subsequent cognitive interviews with patients (n = 11) were conducted to refine items. Phase II was a prospective cohort study involving weekly administration of the refined measure by telephone to patients (n = 75) who are prescribed DAAs to evaluate reliability and consistency with viral response.
Results
In the cohort study, internal consistency ranged from acceptable (α = .69) to very high (α = 1.00) across time points and was quite high on average (α = .91). Across the 75 participants, there were 895 measurement occasions; of those, nonadherence was reported on only 27 occasions (3%), all of which occurred in the first 12 weeks. These 27 occasions represented 19 (26%) different individuals. At 12 weeks, 1 (1%) of patients had a detectable HCV viral load; at 12–24 weeks posttreatment, 4 (5%) had a sustained viral response. Nonadherent patients reported an average of 1.41 reasons for nonadherence.
Conclusions
This multi-method study established content validity of reasons for nonadherence and reliability of extent of nonadherence. High rates of adherence and viral response were consistent with previous studies using other nonadherence measurement methods.

Similar content being viewed by others
References
Petruzziello A, Marigliano S, Loquercio G, et al. Global epidemiology of hepatitis C virus infection: an up-date of the distribution and circulation of hepatitis C virus genotypes. World J Gastroenterol. 2016;22:7824–7840.
Kabiri M, Jazwinski AB, Roberts MS, et al. The changing burden of hepatitis C virus infection in the United States: model-based predictions. Ann Intern Med. 2014;161:170–180.
Razavi H, ElKhoury AC, Elbasha E, et al. Chronic hepatitis C virus (HCV) disease burden and cost in the United States. Hepatology. 2013;57:2164–2170.
van der Meer AJ, Veldt BJ, Feld JJ, et al. Association between sustained virological response and all-cause mortality among patients with chronic hepatitis C and advanced hepatic fibrosis. Jama. 2012;308:2584–2593.
Naggie S, Muir AJ. Oral combination therapies for hepatitis c virus infection: successes, challenges, and unmet needs. Annu Rev Med. 2017;68:345–358.
Lo Re V 3rd, Teal V, Localio AR, et al. Relationship between adherence to hepatitis C virus therapy and virologic outcomes: a cohort study. Ann Intern Med. 2011;155:353–360.
McHutchison JG, Manns M, Patel K, et al. Adherence to combination therapy enhances sustained response in genotype-1-infected patients with chronic hepatitis C. Gastroenterology. 2002;123:1061–1069.
Voils CI, Hoyle RH, Thorpe CT, et al. Improving the measurement of self-reported medication nonadherence. J Cin Epidemiol. 2011;64:250–254.
Voils CI, King HA, Neelon B, et al. Characterizing weekly self-reported antihypertensive medication nonadherence across repeated occasions. Patient Preference Adherence. 2014;8:643–650.
Voils CI, Maciejewski ML, Hoyle RH, et al. Initial validation of a self-report measure of the extent of and reasons for medication nonadherence. Med Care. 2012;50:1013–1019.
Blalock DV, Zullig LL, Bosworth HB, et al. Self-reported medication nonadherence predicts cholesterol levels over time. J Psychosom Res. 2019;118:49–55.
Weidenbacher HJ, Beadles CA, Maciejewski ML, et al. Extent and reasons for nonadherence to antihypertensive, cholesterol, and diabetes medications: the association with depressive symptom burden in a sample of American veterans. Patient Preference Adherence. 2015;9:327–336.
Cornelius T, Voils CI, Birk JL, et al. Identifying targets for cardiovascular medication adherence interventions through latent class analysis. Health Psychol. 2018;37:1006–1014.
Bonner JE, Esserman DA, Golin CE, et al. Self-efficacy and adherence to antiviral treatment for chronic hepatitis C. J Clin Gastroenterol. 2015;49:76–83.
Evon DM, Esserman DA, Bonner JE, et al. Adherence to PEG/ribavirin treatment for chronic hepatitis C: prevalence, patterns, and predictors of missed doses and nonpersistence. J Viral Hepatitis. 2013;20:536–549.
Evon DM, Golin CE, Bonner JE, et al. Adherence during antiviral treatment regimens for chronic hepatitis C: a qualitative study of patient-reported facilitators and barriers. J Clin Gastroenterol. 2015;49:e41–e50.
Guest G, Namey E, McKenna K. How many focus groups are enough? Building an evidence base for nonprobability sample sizes. Field Methods. 2017;29:3–22.
Hsieh H-F, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277–1288.
Namey E, Guest G, McKenna K, et al. Evaluating bang for the buck: a cost-effectiveness comparison between individual interviews and focus groups based on thematic saturation levels. Am J Eval. 2016;37:425–440.
Willis G. Cognitive Interviewing: A Tool for Improving Questionnaire Design. Thousand Oaks: Sage; 2005.
Lincoln YS, Guba E. Naturalistic Inquiry. Beverly Hills, CA: Sage; 1985.
Baker DW, Williams MV, Parker RM, et al. Development of a brief test to measure functional health literacy. Patient Educ Couns. 1999;38:33–42.
Fagerlin A, Zikmund-Fisher BJ, Ubel PA, et al. Measuring numeracy without a math test: development of the/Numeracy Scale. Med Decis Mak. 2007;27:672–680.
Callahan CM, Unverzagt FW, Hui SL, et al. Six-item screener to identify cognitive impairment among potential subjects for clinical research. Med Care. 2002;40:771–781.
Horne R, Weinman J, Hankins M. The beliefs about medicines questionnaire: the development and evaluation of a new method for assessing the cognitive representation of medication. Psychol Health. 1999;14:1–24.
Fernandez S, Chaplin W, Schoenthaler AM, et al. Revision and validation of the medication adherence self-efficacy scale (MASES) in hypertensive African Americans. J Behav Med. 2008;31:453–462.
Walker DR, Juday TR, Manthena SR, et al. The impact of ribavirin on real-world adherence rates in hepatitis C patients treated with sofosbuvir plus simeprevir. ClinicoEcon Outcomes Res. 2015;7:637–642.
Petersen T, Townsend K, Gordon LA, et al. High adherence to all-oral directly acting antiviral HCV therapy among an inner-city patient population in a phase 2a study. Hepatol Int. 2016;10:310–319.
Cunningham EB, Amin J, Feld JJ, et al. Adherence to sofosbuvir and velpatasvir among people with chronic HCV infection and recent injection drug use: the SIMPLIFY study. Int J Drug Policy. 2018;62:14–23.
Rogal SS, McCarthy R, Reid A, et al. Primary care and hepatology provider-perceived barriers to and facilitators of hepatitis CtTreatment candidacy and adherence. Dig Dis Sci. 2017;62:1933–1943. https://doi.org/10.1007/s10620-017-4608-9.
Phillips FH, Barnes D. Social support and adherence for military veterans with hepatitis C. Clin Spec. 2016;30:38–44.
Zacks S, Beavers K, Theodore D, et al. Social stigmatization and hepatitis C virus infection. J Clin Gastroenterol. 2006;40:220–224.
Zickmund S, Ho EY, Masuda M, et al. “They treated me like a leper”. Stigmatization and the quality of life of patients with hepatitis C. J Gen Intern Med. 2003;18:835–844.
Spitzer RL, Williams JB, Kroenke K, et al. Utility of a new procedure for diagnosing mental disorders in primary care: the PRIME-MD 1000 study. J Am Med Assoc. 1994;272:1749–1756.
Gellad WF, Thorpe CT, Steiner JF, et al. The myths of medication adherence. Pharmacoepidemiol Drug Saf. 2017;26:1437–1441.
Dominitz JA, Boyko EJ, Koepsell TD, et al. Elevated prevalence of hepatitis C infection in users of United States veterans medical centers. Hepatology. 2005;41:88–96.
Falade-Nwulia O, Suarez-Cuervo C, Nelson DR, et al. Oral direct-acting agent therapy for hepatitis c virus infection: a systematic review. Ann Intern Med. 2017;166:637–648.
Acknowledgments
The authors would like to extend special thanks to Andrew Muir, MD MHS, and Christine Hunt, MD, for their clinical input and Jamiyla Bolton, Jahdai Dawes, Leslie Gaillard, Tina Lucas, Elizabeth Strawbridge, and Cherisa Williams for their assistance with recruitment and data collection. The views represented in this article represent those of the authors and not those of the VA or the United States Government.
Funding
Phase I of this study was supported by an investigator-initiated grant from Vertex Pharmaceuticals to Dr. Voils. Phase II of this study was funded by the Department of Medicine at Duke University Medical Center. Effort on this paper was supported by resources and facilities at the Veterans Affairs Medical Centers in Madison and Durham and by a Research Career Scientist award to Dr. Voils from the Health Services Research and Development (HSR&D) service of the Department of Veterans Affairs (VA; RCS 14-443) and a VA HSR&D Career Development Award to Dr. Gellad (CDA 14-158).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Gellad is cofounder and holds equity in Higgs Boson Inc. No other author declares a conflict of interest.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Voils, C.I., King, H.A., Thorpe, C.T. et al. Content Validity and Reliability of a Self-Report Measure of Medication Nonadherence in Hepatitis C Treatment. Dig Dis Sci 64, 2784–2797 (2019). https://doi.org/10.1007/s10620-019-05621-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-05621-7