Abstract
Background
Initial response to corticosteroids (CS) is recognized as a strong predictor of outcomes in ulcerative colitis (UC).
Aim
To compare outcomes of early poor responders (PR) versus good responders (GR) to initial CS at 1, 2, and 3 years from diagnosis.
Methods
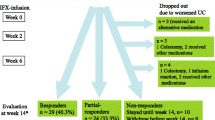
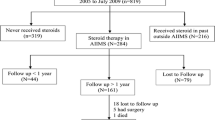
In this retrospective study, we report longitudinal outcomes of children with moderate–severe UC, initiating oral/IV CS < 1 month of diagnosis and a minimum follow-up (FU) of 1 year. CS resistance (CSR) and CS dependency (CSD) were combined as PR, and those with CS-free remission (CSFR) at 6 months were GR.
Results
Of 116 children with UC, 76 (33 males) fulfilled study criteria. Median age at diagnosis was 12 years (IQR 12–14), and a median FU was 48 months (IQR 27–65). Thirty-five (46%, CSR = 10, CSD = 25) were PR, and 41 (54%) were GR. Mean relapse (2.39 vs. 1.1, p = 0.0009), acute severe UC flare-up (40% vs. 9.7%, p = 0.002), and colectomy rates (34.2% vs. 2.4%) were greater in PR versus GR, despite frequent early (< 6 months) use of azathioprine (74% vs. 27%, p = 0.004) and anti-TNFs (43% vs. 2.4%, p = 0.0001). Cumulative colectomy at 3 years was lowest in those with GR versus CSD and CSR (2.4% vs. 28% and 50% p = 0.001). On univariate analysis, CRP > 20 mg/L at diagnosis, Mayo Clinical Score > 1 at 3 months, and PR predicted colectomy. On multivariate regression, only baseline CRP > 20 mg/L predicted colectomy (HR 4.9, p = 0.03).
Conclusions
Baseline CRP and poor response to initial CS are associated with unfavorable outcomes in children with UC.





Similar content being viewed by others
References
Ruemmele FM, Turner D. Differences in the management of pediatric and adult onset ulcerative colitis—lessons from the joint ECCO and ESPGHAN consensus guidelines for the management of pediatric ulcerative colitis. J Crohns Colitis. 2014;8:1–4.
Van Limbergen J, Russell R, Drummond H, et al. Definition of phenotypic characteristics of childhood-onset inflammatory bowel disease. Gastroenterology. 2008;135:1114–1122.
Jakobsen C, Bartek J Jr, Wewer V, et al. Differences in phenotype and disease course in adult and paediatric inflammatory bowel disease—a population-based study. Aliment Pharmacol Ther. 2011;34:1217–1224.
Jakobsen C, Munkholm P, Paerregaard A, et al. Steroid dependency and pediatric inflammatory bowel disease in the era of immunomodulators—a population-based study. Inflamm Bowel Dis. 2011;17:1731–1740.
Hyams J, Markowitz J, Lerer T, et al. The natural history of corticosteroid therapy for ulcerative colitis in children. Clin Gastroenterol Hepatol. 2006;4:1118–1123.
Duricova D, Pedersen N, Lenicek M, et al. The clinical implication of drug dependency in children and adults with inflammatory bowel disease: a review. J Crohns Colitis. 2011;5:81–90.
Yoon J, Cheon J, Park J, et al. Clinical outcomes and factors for response prediction after the first course of corticosteroid therapy in patients with active ulcerative colitis. J Gastroenterol Hepatol. 2011;26:1114–1122.
Sandborn WJ, Rutgeerts P, Feagan BG, et al. Colectomy comparison after treatment of ulcerative colitis with placebo or infliximab. Gastroenterology. 2009;137:1250–1260.
Turner D, Mack D, Leleiko N, et al. Severe pediatric ulcerative colitis: a prospective multicentre study of outcomes and predictors of response. Gastroenterology. 2010;138:2282–2291.
Turner D, Levine A, Escher J, et al. Management of pediatric ulcerative colitis: joint ECCO and ESPGHAN evidence-based consensus guidelines. J Pediatr Gastroenterol Nutr. 2012;55:340–361.
Peyrin-Biroulet L, Panés J, Sandborn W, et al. Defining disease severity in inflammatory bowel diseases: current and future directions. Clin Gastroenterol Hepatol. 2016;14:348–354.
Dignass A, Eliakim R, Magro F. Second European evidence-based consensus on the diagnosis and management of ulcerative colitis: current management. J Crohns Colitis. 2012;6:965–990.
Fumery M, Duricova D, Gower-Rousseau C, et al. Review article: the natural history of paediatric-onset ulcerative colitis in population-based studies. Aliment Pharmacol Ther. 2016;43:346–355.
Gower-Rousseau C, Dauchet L, Vernier-Massouille G, et al. The natural history of pediatric ulcerative colitis: a population-based cohort study. Am J Gastroenterol. 2009;104:2080–2088.
Solberg IC, Lygren I, Jahnsen J, et al. Clinical course during the first 10 years of ulcerative colitis: results from a population-based inception cohort (IBSEN Study). Scand J Gastroenterol. 2009;44:431–440.
Lindgren SC, Flood LM, Kilander AF, et al. Early predictors of glucocorticosteroid treatment failure in severe and moderately severe attacks of ulcerative colitis. Eur J Gastroenterol Hepatol. 1998;10:831–835.
Ho GT, Mowat C, Goddard CJ, et al. Predicting the outcome of severe ulcerative colitis: development of a novel risk score to aid early selection of patients for second-line medical therapy or surgery. Aliment Pharmacol Ther. 2004;19:1079–1087.
Henriksen M, Jahnsen J, Lygren I, et al. C-reactive protein: a predictive factor and marker of inflammation in inflammatory bowel disease. Results from a prospective population-based study. Gut. 2008;57:1518–1523.
Schechter A, Griffiths C, Gana JC, et al. Early endoscopic, laboratory and clinical predictors of poor disease course in paediatric ulcerative colitis. Gut. 2015;64:580–588.
Sandborn W, Tremaine W, Schroeder K, et al. A placebo controlled trial of cyclosporine enema for mildly to moderately active ulcerative colitis. Gastroenterology. 1994;106:1429–1435.
Fell J, Muhammed R, Spray C, et al. Management of ulcerative colitis. Arch Dis Child. 2016;101:469–474.
Turner D, Otley A, Mack D, et al. Development, validation and evaluation of a pediatric ulcerative colitis activity index: a prospective multicentre study. Gastroenterology. 2007;133:423–432.
Rai T, Choudhury BN, Kedia S, et al. Short-term clinical response to corticosteroids can predict long-term natural history of ulcerative colitis: prospective study experience. Dig Dis Sci. 2017;62:1025–1034.
Barreiro-Alonso E, Saro-Gismera C, Sánchez M. Outcomes and prediction of corticosteroid therapy after successive courses of ulcerative colitis treatments. Expert Rev Gastroenterol Hepatol. 2018;12:733–741.
Khan N, Patel D, Shah Y, et al. A novel user-friendly model to predict corticosteroid utilization in newly diagnosed patients with ulcerative colitis. Inflamm Bowel Dis. 2017;23:991–997.
Turner D, Seow CH, Greenberg GR, et al. A systematic prospective comparison of noninvasive disease activity indices in ulcerative colitis. Clin Gastroenterol Hepatol. 2009;7:1081–1088.
Khan N, Almukhtar R, Cole E, et al. Early corticosteroids requirement after the diagnosis of ulcerative colitis diagnosis can predict a more severe long-term course of the disease—a nationwide study of 1035 patients. Aliment Pharmacol Ther. 2014;40:374–381.
Aloi M, D’Arcangelo G, Pofi F, et al. Presenting features and disease course of pediatric ulcerative colitis. J Crohns Colitis. 2013;7:e505–e515.
Hyams J, David S, Mack D, et al. Predicting response to pediatric colitis therapy: the PROTECT study. Gastroenterology. 2018;154:s667.
Hyams J, Lerer T, Mack D, et al. Outcome following thiopurine use in children with ulcerative colitis: a prospective multicenter registry study. Am J Gastroenterol. 2011;106:981–987.
Acknowledgments
We would like to acknowledge doctors Mews, Ravikumara, and Thacker for providing general support.
Author information
Authors and Affiliations
Contributions
GDR (10%), LT (10%), AL (0%), ZG (80%), and PL (0%) conceptualized and designed the study; GDR, (40%), LT (40%), AL (5%), ZG (15%), and PL (0%) took part in acquisition of data; GDR, (5%), LT (5%), AL (5%), ZG (85%), and PL (0%) analyzed and interpreted the data; and GDR, (5%), LT (5%), AL (5%), ZG (75%), and PL (10%) drafted the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors Gayetri Deva Rajoo, Lian Tan, Ainslie Lopez, and Zubin Grover declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deva Rajoo, G., Tan, L., Lopez, A. et al. Early Response to Corticosteroid and Baseline C-Reactive Protein Predicts Outcomes in Children with Moderate to Severe Ulcerative Colitis. Dig Dis Sci 64, 1929–1937 (2019). https://doi.org/10.1007/s10620-019-05486-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-05486-w