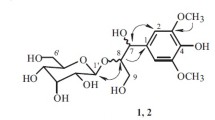
Two new phenolic glycosides, 2-(2-((1E,6Z)-3,8-dihydroxy-3,7-dimethylocta-1,6-dien-1-yl)-4-hydroxy-6-methylphenoxy)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol (1) and 2-(2-((E)-2-(2,6-dimethyl-2,3,4,7-tetrahydrooxepin-2-yl)vinyl)-4-hydroxy-6-methylphenoxy)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol (2) were isolated from Thalictrum scabrifolium. Their structures were elucidated by spectroscopic methods. Compounds 1 and 2 exhibited anti-TMV activities with inhibition rates of 34.1 and 35.9% at a concentration of 20 μM/mL, respectively, compared with 35.3% for the positive control ningnanmycin.

Similar content being viewed by others
References
D. C. Hao, Ranunculales Medicinal Plants, Biodiversity, Chapter 7, Biodiversity, Chemodiversity, and Pharmacotherapy of Thalictrum Medicinal Plants, Elsevier Science Publisher B V, Amsterdam, 2019, p. 261.
C. Y. Wang, Flora of China, Vol. 27, 2nd Section, Chinese Science Press, Bei**g, 2001, p. 282.
F. Wu, L. Liu, Y. N. Zhu, N. Lv, and Z. G. Li, Chem. Nat. Compd., 57, 339 (2021).
B. D. Komilov, M. A. Agzamova, I. M. Isaev, and K. A. Eshbakova, Chem. Nat. Compd., 56, 814 (2020).
F. C. Meng, X. D. Wei, Y. Sun, Q. H. Zeng, G. W. Wang, X. Z. Lan, Z. H. Liao, and M. Chen, Nat. Prod. Res., 35, 5757 (2020).
G. Y. Gao, S. B. Chen, and L. W. Wang, China J. Chin. Mat. Med.., 24, 60 (1999).
F. Z. Erdemgil, K. H. C. Baser, P. Akbay, O. Sticher, and I. Calis, Zeitschrift fur Naturforschung C., 58, 632 (2003).
Y. P. Lou, Y. Gu, and M. A. Ou Yang, Res. Developm. Nat. Prod., 19, 8 (2007).
Y. G. Gu, Foreign Pharm.·Bot. Med., 3, 1 (1982).
J. Ropivia, S. Derbre, C. Rouger, F. Pagniez, P. L. Pape, and P. Richomme, Molecules, 15, 6476 (2010).
S. Julia and V. Maria, Planta Med., 69, 153 (2003).
S. G. Li, Y. Q. Su, D. K. Ma, J. Y. Li, J. Guo, D. H. Li, Z. L. Li, and H. M. Hua, Modern Medicine Clinic., 029, 312 (2014).
T. Liu, J. North Pharm., 10, 85 (2013).
R. Badamjav, L. Zhang, D. Sonom, Y. H. Wu, J. P. Kou, B. Y. Yu, and F. Li, J. Ethnopharmacol., 264, 1 (2020).
Z. Tian, G. Y. Gao, and J. S. Yang, Chin. Tradit. Herb. Drugs, 31, 839 (2000).
R. S. Wang, J. F. Sun, M. **, C. Ye, J. M. Wang, L. **, Y. J. Ma, W. Zhou, and G. Li, Nat. Prod. Res., 35, 4423 (2021).
J. R. Ioset, A. Marston, M. P. Gupta, and K. Hostettmann, J. Nat. Prod., 63, 424 (2000).
X. Q. Zhang, J. Shi, S. X. Feng, L. Xue, and L. P. Tian, Nat. Prod. Res., 34, 398 (2020).
Q. F. Hu, B. Zhou, J. M. Huang, X. M. Gao, L. D. Shu, G. Y. Yang, and C. T. Che, J. Nat. Prod., 76, 292 (2013).
M. Zhou, M. M. Miao, G. Du, S. Z. Shang, W. Zhao, Z. H. Liu, G. Y. Yang, C. T. Che, Q. F. Hu, and X. M. Gao, Org. Lett., 16, 5016 (2014).
Acknowledgment
This project was supported by the Key Laboratory of Yunnan Provincial Department of Education (2022HD021), the Foundation of Yunnan Basic Research Youth Program (2019FD119), and the Foundation of Yunnan Innovative Research Team (2019HC020).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 4, July–August, 2023, pp. 568–571.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, JN., Yang, M., **ong, RF. et al. Two New Phenolic Glycosides from Thalictrum scabrifolium and Their Anti-TMV Activities. Chem Nat Compd 59, 675–679 (2023). https://doi.org/10.1007/s10600-023-04084-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-04084-7